3278
Views & Citations2278
Likes & Shares
Lead is toxic for virtually all organs of the body and has significant debilitating effects on the nervous, renal, hepatic and hematopoietic systems. The present work aimed to determine the effects of lead acetate (Pb) on the histopathological changes of kidney of Coturnix sp. and the ameliorative effect of vitamin C supplementation (VC) on Pb toxicity.
Twenty-four mature male quails (Coturnix sp.) were used and divided into four groups (6 bird/ group). The control one received only basal diet without any supplementation while other groups received treatment. The first treatment group received oral gavage (600 mg/kg) of Pb for 10 days, then received VC (60 mgl-1) for 11 days. The second group received a single dose of VC (60 mgl-1) and the third group was given single dose of Pb for 21 days. The histopathological alterations of kidney of quail domestic was detected using digital camera of light microscope.
Our results showed that Control and VC-supplemented groups have a normal histopathological kidney shape. However, the administration of Pb for 21days (group II) induced histopathological alterations in kidney like, tubular necrosis, cellular infiltration, mild fibrosis in interstitial tissue congestion, degeneration and vacuolization. Furthermore, the glomerular alterations were glomerular hyalinization when compared with control one. On the other hand, supplementation of VC on Pb intoxicated birds showed some ameliorative effects to Pb toxicity. Meanwhile, supplementation with vitamin C could ameliorate the negative impacts of Pb on quails.
Keywords: Lead acetate, Histological alterations, Vitamin C, Quails
INTRODUCTION
For many years, lead has been considered to be the toxicant that poses the greatest risk [1]. Moreover, lead is a dangerous heavy metal with negative effects on birds in natural and laboratory environment. Lead is toxic for virtually all organs of the body and has significant debilitating effects on the nervous, renal, hepatic and hematopoietic systems [2]. Lead has been linked with cancers of stomach, small intestine, large intestine, ovary, kidney, lungs, myeloma, all lymphomas, and all leukemia [3]. All sources of lead such as the manufacture of batteries, painting, printing, pottery glazing and lead smelting processes contribute to an increase in permissible exposure limit for metallic lead, lead oxide and lead salts and soaps that has set by WHO and other health organizations [4]. The long-term effects of lead chronic exposure have been documented by several investigations with special attention to the exposure of developing animals and young children [5].
The kidney is a vital organ in health and disease. Many environmental contaminants and chemical variables, including drugs, alter the functions of the kidney [6]. The absorbed Pb is conjugated in the liver and passed to the kidney, where a small quantity is excreted in urine and the rest accumulates in various body organs and interferes with their function specially the kidney as a target site for Pb toxicity [7]. Continuous environmental and occupational Pb exposure can lead to chronic nephropathy [8].
Vitamin C also known as ascorbic acid is a water-soluble vitamin required for physiological growth and development. It is needed for collagen synthesis which is necessary for the formation of tendons, ligaments, skin, and blood vessels. Vitamin C aids in wound healing and scar tissue formation. It restores and sustains bones, teeth and cartilage acting as a major antioxidant [9].
Quail’s meat with its unique capabilities, quick digestion, low in cholesterol, high in protein, is best for children, elderly and hypertensive peoples [10]. The histological and the histopathological characterizations in the kidney tissues due to lead intoxication are not documented and have not yet been identified. Therefore, this study was designed basically to evaluate the toxic impact of lead acetate on histopathological alterations of kidney of male local quail (Coturnix sp.) and the ameliorative role of Vitamin C to detoxification of Pb toxicity.
MATERIALS AND METHODS
Chemicals
Lead acetate (≥99.0%) and ascorbic acid (Vitamin C) (98%).
Sample collection and chemicals
Twenty-four young male (40 days old) local quails (Coturnix sp.) weighted average (250.0 ± 70.35 g) were used in the present study. Birds maintained and adapted for 7 days under lab conditions. After the acclimatization period in animal husbandry unit of faculty of science, Misurata University. Birds were randomly selected for formation of 4 groups with 6birds for each. Furthermore, animals in all groups were weighed every day.
EXPERIMENTAL DESIGN
Birds were weighed and classified randomly into 4 groups (6 birds/groups) according to doses of Pb and VC. Stock solution (1000 ppm) of Pb was prepared and stored in clean glass bottles and diluted to concentration of (600 mg/kg body weight. Similarly, for Vitamin C with dose of (60mg/kg bw). First group was the Control one that had given distilled water by orogastric tube. The second group of treated groups (Group A) were given vitamin C in a dose of 60mg/kg b.w. by orogastric tube for 21 days. The third (Group B), were given Pb in a dose of 600mg/kg b.w. by orogastric tube for 21 days. The last one (Group C) were given Pb (600mg/kg bw) for 10 days then were given vitamin C (60mg/kg bw) for 11 days by the same.
HISTOPATHOLOGICAL INVESTIGATIONS
For the histological investigations, small parts of the liver from the control and the treated animals were fixed in neutral formalin (10%). After the fixation period, the tissues were washed in saline solution 0.9% NaCl, dehydrated through a graded series of ethyl alcohol, cleared in xylene, embedded in the parables, sectioned at a thickness 5-6 microns, mounted on the glass slides and stained with haematoxylin and eosin for general morphological studies. Sections were studied and the tissues were compared with the control [11,12].
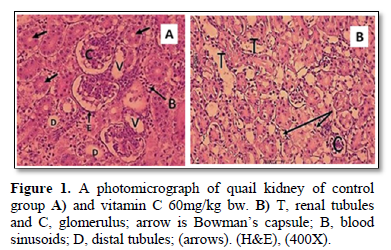
RESULTS
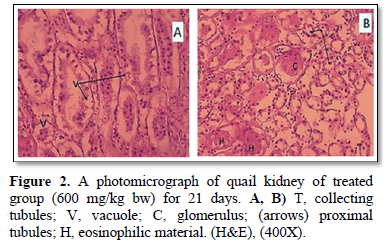
The present study showed that the transfer sections of kidney of Coturnix sp. in control and 60 mg/kg bw of VC treatment quail’s groups were appeared as a normal, with well-formed tubular and glomerular structures (Figure 1A, 1B). However, Figure 2, of group B that exposed to Pb showed histopathological alterations of kidney of quails treated when compared with control one. These histopathological examination of kidney of treatment group with 600mg/kg bw of Pb (Figure 2A, 2B) showed tubular necrosis, cellular infiltration, mild fibrosis in interstitial tissue congestion, degeneration and vacuolization. As period of exposure extended, vacuolated cytoplasm and degeneration of bowman’s capsules were noticed. Furthermore, eosinophilic material and observed necrosis of epithelium of proximal convoluted tubules of kidney (Figure 2B).
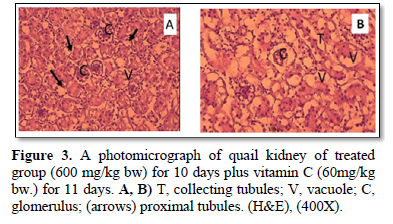
On the other hand, the present study reported that the histopathological changes in kidney tissue of treated group with Pb for the first 10 days and VC for the remaining 11 days (Figure 3A, 3B), were more improved than that of Pb group (Figure 2A, 2B).
DISCUSSION
The results of the present investigation showed that the subtoxic chronic lead acetate (600 mg/kg bw.) exposure causes slow progressive tubular, glomerular and interstitial damages. Some of these findings are in agreement with some results of previous investigations [13]. The varieties in the histopathological alterations of the renal tissues due to Pb intoxication obtained by different investigators [14] could be due to the variations in the level of exposure, duration, route of administration and animal species used in the experiments.
Severe and extensive histopathological alterations in the renal tissue induced by lead acetate as seen in the present work suggest the potential of lead acetate to cause oxidative damage to cellular organelles. Similar results were recorded by Jarrar and Mahmoud [10,15] lead toxicity and characterized by tubular, interstitial and glomerular damages, including renal lesions, tumors and cancer risk. The current study also agreed with other studies have shown that, after exposure animals to lead acetate causes of kidney necrosis, hypertrophy of glomerulus, degeneration and dissociation of renal tubules and Bowman's capsule, proliferation in the renal tubule and haemopoieatic tissue, shrinkage of glomerulus, dilated blood vessel, rupture of Bowman's capsule, and obliterated Bowman’s space [16,17].
Our result confirmed that using of vitamin C reduced the toxic impact of lead acetate on kidney of male quails to some extent. Abbasand Tanveer [18,19] reported that oxidative stress-induced tissue damage can be prevented or ameliorated by favoring the balance towards a lower oxidative stress status. Furthermore, it seems that the ameliorative effect of vitamin C involves the maintenance of antioxidant capacity in ameliorating the tissue against oxidative stress.
CONCLUSION
Lead supplementation in diet resulted in alteration of normal structure of kidney tissue. Histopathological changes of variable degree in kidney were also evident. Addition of higher doses of vitamin C to the diet may be more efficacious in fully reversing the negative effect of Pb on histological parameters.
ACKNOWLEDGEMENT
The authors extend their appreciation to the Animal husbandry unit and Zoology Researches LabatFaculty of science (Misurata University) and to Laboratory Attendants, FathyaHenish and MeladEshkab for their technical assistance.
1. EPA US (1989) Evaluation of the potential carcinogenicity of lead and lead compounds. Office of Health and Environmental Assessment.
2. Patrick L (2003) Toxic metals and antioxidants: Part II. The role of antioxidants in arsenic and cadmium toxicity. Altern Med Rev 8: 106-128.
3. Reddy SB, Charles MJ, Raju GJ, Deddy BS, Reddy TS, et al. (2004) Trace elemental analysis of cancer-afflicted intestine by PIXE technique. Biol Trace Elem Res 102: 265-281.
4. Harbison RD (1998) Hamilton & Hardy’s Industrial Toxicology (ed. R.D. Harbison), 5th ed, Mosby, Philadelphia, pp: 70-76.
5. El-Neweshy SM, El-Sayed SY (2011) Influence of vitamin C supplementation on lead-induced histopathological alterations in male rats. Exp Toxicol Pathol 63: 221-227.
6. Begg EJ, Barclay M L (1995) Aminoglycisides-50 years on. Br J Clin Pharmacol 39: 597-603.
7. Taib NT, Jarrar BM, Mubarak M (2004) Ultrastructural alterations in hepatic tissues of white rats (Rattusnorvegicus) induced by lead experimental toxicity. Saudi J Biol Sci 11: 11-20.
8. Wynee J, Stringfield C (2007) Treatment of lead toxicity and crop stasis in a California condor (Gymnogypscalifornianus). J Zoo Wildl Med 38: 588-590.
9. Ganesh HG, Arun JP, Jyotsna AP, Mandakini SK, Ajit S (2016) Biochemical effects of lead exposure on oxidative stress and antioxidant status of battery manufacturing workers of Western Maharashtra, India. J Basic Clin Physiol Pharmacol 27: 141-146.
10. Jarrar BM (2001) Ultrastructural alterations in proximal tubule cells induced by lead. Pak J Biol Sci 4: 1281-1284.
11. Presnell JK, Schreibman MP (1997) Humason’s Animal Tissue Techniques. Univ Press Balfi more.
12. Bancroft JD, Gamble M (2007) Theory and practice of histological techniques. 5th Ed. Churchill Livingstone London 800.
13. Rodrigo AF, Hernandez JJ, Lopez-Caballero F, Gill AP (2001) Immunohistochemical evidence for the expression and induction of paraxonase in rat liver, kidney, lung and brain tissue, implications for its physiological role. Chem Biol Interact 137: 123-137.
14. Alarifi SA, Al-Doaiss A, Alkahtani S, Al-Farraj SA, Al-Eissa MS (2011) Blood chemical changes and renal histological alterations induced by gentamicin in rats. Saudi. J Biol Sci 19: 103-110.
15. Mahmoud HM, Haggag AMH, El-Gebaly HS (2003) Toxicological studies of malathion on Japanese Quail (Coturnix Japonica). Life Sci J 9.
16. Alkahtani S, AlarifiSaud A, Al-Doaiss AA (2009) Detection of apoptosis induced by gentamicin in rat hepatocytes. Int J Zoolog Res 5:161-170.
17. Kandeel M, Abdelaziz I, Elhabashy N, Hegazy H, Tolba Y (2011) Nephrotoxicity and oxidative stress of single large dose or two divided doses of gentamicin in rats. Pak J Biol Sci 1: 1-7.
18. Abbas M T (2014) The protective effect of quercetin on Diazinon-induced oxidative stress in rats. Iraqi Nat J Chem 53: 96-122.
19. Tanveer A, Shagufta A, Ahmad DS, Ovais A (2015) Ameliorative effect of vitamin-C on lead-induced biochemical alterations in the serum of broilers. Appl Biol Res 17: 219-223.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Astronomy and Space Research
- Proteomics and Bioinformatics (ISSN:2641-7561)
- Food and Nutrition-Current Research (ISSN:2638-1095)
- Journal of Genomic Medicine and Pharmacogenomics (ISSN:2474-4670)
- Advances in Nanomedicine and Nanotechnology Research (ISSN: 2688-5476)
- Journal of Agriculture and Forest Meteorology Research (ISSN:2642-0449)
- Journal of Genetics and Cell Biology (ISSN:2639-3360)