2776
Views & Citations1776
Likes & Shares
Background: Accumulating evidence suggests that Toll-like receptors (TLRs) are important players in the pathophysiology of ischemic stroke (IS). In most cases of IS, endogenous ligands binding to TLRs activate monocytes to secrete pro inflammatory cytokines. It remains elusive as to whether there is any correlation between a National Institutes of Health Stroke Scale (NIHSS) score with TLR2/TLR4 expression on the blood monocytes and pro-inflammatory/anti-inflammatory cytokines in IS.
Methods: We used flow cytometry in 68 consecutives IS patients and 78 controls without IS to assess the TLR2/TLR4 expression on peripheral blood monocytes. Serum IL-1β, IL-6, IL-10 cytokine levels were measured using ELISA.
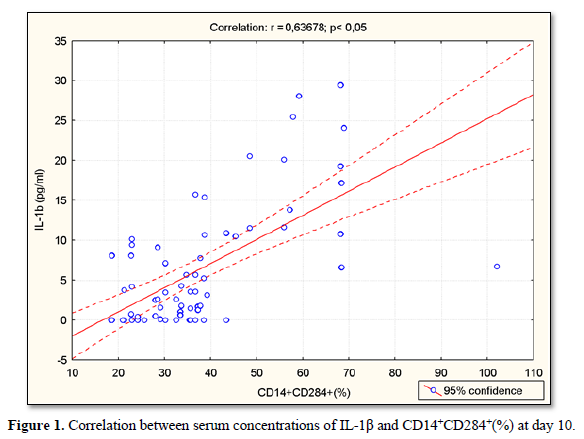
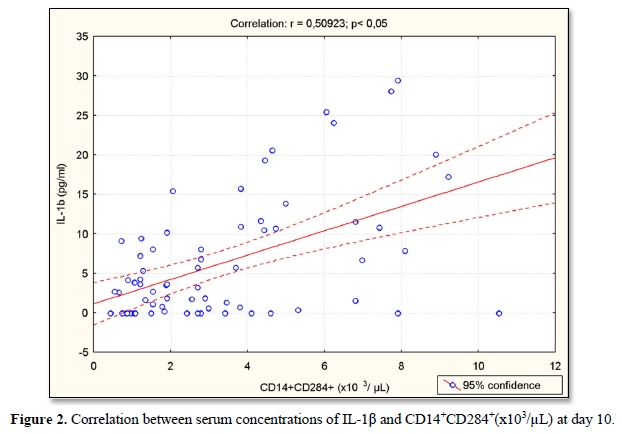
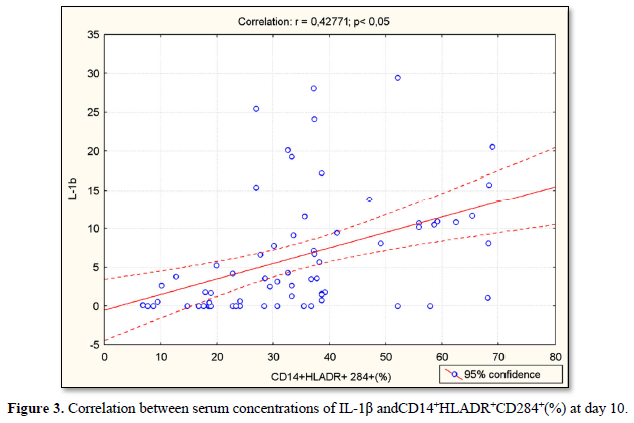
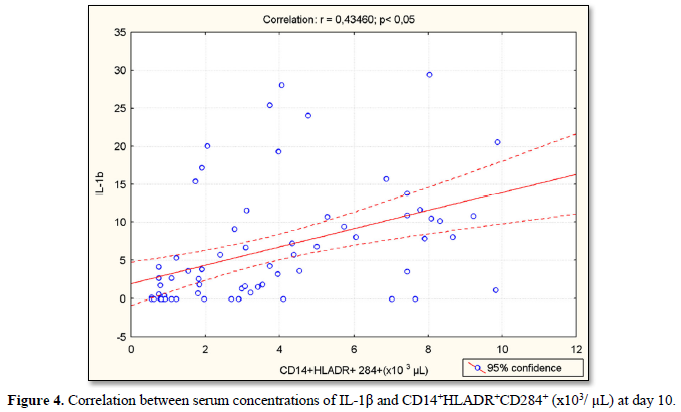
Results: At baseline, there were no differences in baseline blood monocytes counts, serum IL-1β and IL-10 levels between IS patients and controls. Serum concentrations of IL-1β and IL-6 were significantly increased in IS patients after 10 days. Serum concentrations of IL-1β were positively correlated with CD14+CD284+(%) (r=0.64; p<0.05), CD14+CD284+(х103/µL) (r=0.51; p<0.05), CD14+HLADR+CD284+ (%) (r=0.43; p<0.05) and CD14+HLADR+CD 284+ (х103/ µL) (r=0.43; p<0.05) at day 10. NIHSS at day 10 was positively correlated with serum concentrations of IL-1β (r=0.69; p<0.05), with CD14+CD284+(%) (r=0.63; p<0.05) and CD14+CD284+(х103/µL) (r=0.51; p<0.05) in IS patients. There were no significant correlations between NIHSS with IL-6 and IL-10 levels and TLR2 expression on the monocytes.
Conclusions: In human IS, upregulation of TLR4 expression on peripheral blood monocytes and IL-1β production could be one of the peripheral mechanisms of brain inflammatory injury.
Keywords: TLR4, TLR2, Monocytes, Ischemic, Stroke
INTRODUCTION
Ischemic stroke (IS) is one of the leading causes of morbidity and mortality around the world. The mechanism underlying brain injury caused by cerebral ischemia and reperfusion remains very complex, but an inflammatory response has been found to be one of the main factors leading to brain damage in IS [1,2]. Accumulating evidence suggests that Toll-like receptors (TLRs) TLR2 and TLR4 are more important than other TLRs in the pathologic progression of cerebral ischemia and reperfusion [3,4]. TLR2 and TLR4 play crucial roles in modulating inflammatory response caused by cerebral ischemia and reperfusion via linking to their endogenous ligands, respectively [5,6]. Moreover, TLR4 polymorphisms were found to be associated with IS [7] and TLR2 and TLR4 expression on monocytes was associated with poor outcome in IS patients [8,9]. Early studies in rats described the arrival of circulating monocytes to capillaries and venules of brain ischemic areas as early as 4 h after IS onset [10]. These monocytes are of supreme immunologic relevance because they are the main effectors of innate immunity through the expression of TLRs [11]. Existing studies in mice have shown a remarkably rapid activation of peripheral immunity after experimental IS [12]. Interestingly,
as early as 6 h after middle cerebral artery occlusion, activated splenocytes secreted significantly enhanced levels of the pro-inflammatory cytokines (TNF-α, Il-1, IL-6) and also the anti-inflammatory IL-10 [12]. In most cases of IS, endogenous ligands binding to TLRs activate monocytes/macrophages to secrete IL-1𝛽 and the level of IL-1𝛽mRNA was found to be decreased in the TLR2 and TLR4 deficient mice which suffered from thromboembolic IS [13]. IL-6 has been mainly secreted by astrocyte and microglia during the course of cerebral ischemia/reperfusion and it was demonstrated that the neurological impairments and the level of IL-6 were both improved more apparently in TLR4 deficient mice than in wild-type mice [14].
It remains elusive as to whether there is any correlation between monocyte numbers in peripheral blood, TLR2/TLR4 expression on the monocytes and pro-inflammatory/anti-inflammatory cytokines in IS. Elucidation of this question is required to judge the need of immunomodulatory therapies in IS and to define specific immune targets that might translate into a better clinical outcome.
MATERIALS & METHODS
Study subject
This study was a hospital-based case-control study conducted in patients attending the affiliated hospitals of Chita Medical Academy (Chita, Russia) from January 2013 to December 2017. We prospectively studied 68 consecutives IS patients with a National Institutes of Health Stroke Scale (NIHSS) score on admission by a certified neurologist. Patients were evaluated at a median of 180 min (interquartile range, 120-350) of stroke onset. Age and sex matched subjects without a history of cerebrovascular disease (n=78) served as control. They were recruited from community samples or hospital employees.
A full physical examination of all the subjects was carried out, and data involving smoking habits, body mass index (BMI), hypertension, diabetes mellitus, and dyslipidemia were recorded. The BMI was calculated by using the formula: body weight in kilograms divided by height in meters squared. Systolic and diastolic blood pressure was measured twice at an interval of 30 min using the automated oscillometric device. An average value of the two readings was considered.
Exclusion criteria included a history of infection, use of steroids or immune suppressants within the preceding 3 months. We excluded those patients with possible concomitant infections by carefully looking for signs and symptoms of infectious diseases at the admission.
All patients underwent a brain CT scan or MRI on admission and were transferred and managed in the Stroke Unit according to the 2013 “Guidelines for the Early Management of Patients with Acute Ischemic Stroke” [15]. Neurological deficiency was assessed daily using the NIHSS until day 7. Patients were categorized according to NIHSS severity in mild (NIHSS ≤ 8), moderate or severe (NIHSS ≥ 9) stroke [15].
The study was approved by the local ethics committee at Chita Medical Academy, Russia, and all participants or their legal representatives signed a written informed consent.
Laboratory analyses
Blood samples were collected between 8:00 AM and 9:00 AM at days 1, 10, and 21 after admission in IS patients and once in Control group patients.
Flow cytometry
The phenotype of monocytes was analyzed immediately after blood removal. Blood samples (100 μl) were transferred into polystyrene FACS tubes and incubated with mouse anti-human CD14 V500 (BD Bioscience, San Jose, CA), mouse anti-human TLR2 PE-Cy7 (eBioscience, San Diego, CA), mouse anti-human TLR4 FITC (IMGENEX, San Diego, CA), and mouse anti-human HLA-DR PerCP-Cy5.5 conjugated (BD Bioscience, San Jose, CA) antibodies. After 30 min at room temperature, 3 ml of FACS lysing solution (FACS Lysing Solution, BD Pharmingen) was added for additional 10 min. Then, the samples were centrifuged at 400 g for 5 min and washed twice with 4 ml phosphate-buffered saline (PBS) supplemented with 0.5% bovine serum albumin (FACS buffer). Immediately after the supernatants were removed, the cells were diluted in 300 μl FACS buffer and were then subjected to flow cytometry with the Cytomics FC-500 flow cytometer (Beckman Coulter Inc. Miami, USA). CXP Cytometer software (Beckman Coulter Inc. Miami, USA) was used for cytometry analysis. Corresponding mean fluorescence intensity (MFI) values were obtained for each expression. The monocyte population was defined by gating CD14+ cells in the corresponding forward and side scatter scan. From each sample, a minimum of 20.000 monocytes was measured.
Cytokines
Serum IL-1β, IL-6, IL-10 cytokine levels were measured using an enzyme immunoassay kits (Vector-Best, Novosibirsk, Russia) for the quantitative determination of the cytokines in serum according to the manufacturer’s protocol.
Statistical Analysis
The distribution of categorical variables was expressed as numbers and percentages. As the variables were non-normally distributed, they are presented as the median and interquartile range (IQR). For statistical comparison of more than two groups, one-way ANOVA was conducted using Bonferroni’s post-hoc test. The unpaired t-tests were used to compare only two groups. Correlations were calculated with the Spearman Rank correlation coefficient. All tests were performed using the Statistica v. 6 software packages (StatSoft, Tulsa, OK, USA). p
RESULTS
Baseline characteristics
The main characteristics of the patients and the control groups are presented in (Table 1) Among 68 patients who presented with acute ischemic stroke, forty-six patients (67.6 %) had mild IS (defined as IHSS ≤8), and the remaining 22 patients had moderate or severe (NIHSS ≥9) stroke. Demographic characteristics, as well as a rate of arterial hypertension, diabetes, hyperlipidemia, atrial fibrillation, smoking status, blood pressure, and laboratory characteristics, were not significantly different between the two IS groups. Nevertheless, the IS groups were characterized by higher levels of systolic and diastolic blood pressure, blood glucose and CRP than the Control group.
Monocytes and serum IL–1β, IL-6 and IL–10 levels cytokine levels
There were no differences in baseline monocytes counts (Table 1) and serum IL-1β and IL-10 levels (Table 2) between IS patients and controls at baseline. IL-6 concentrations were higher in patients with moderate and severe IS (9.26, IQR 4.39-20.11) than in mild IS patients (4.78, IRQ 4.03-6.45) and control subjects (1.74, IQR 1.05-3.31). Serum concentrations of IL-1β and IL-6 were significantly increased in IS patients after 10 days (Table 3).
Correlation between monocyte numbers in peripheral blood, TLR2/TLR4 expression on the monocytes and IL-1β, IL-6 and IL-10 levels cytokine levels
Expression of TLR2, TLR4 on monocytes of peripheral blood in IS patients and Control group is presented in (Table 2) and and serum concentrations of IL–1β, IL–6 and IL–10 in IP patients and Control group are presented in (Table 3).
Serum concentrations of IL-1β were positively correlated with CD14+CD284+(%) (r=0.64; p<0.05) (Figure 1), CD14+CD284+(х103/µL) (r=0.51; p<0.05) (Figure 2), CD14+HLADR+CD284+ (%) (r=0.43; p<0.05) (Figure 3) and CD14+HLADR+CD 284+ (х103/ µL) (r=0.43; p<0.05) (Figure 4) at day 10. There was no significant correlation between serum concentrations of IL-1β and TLR2 expression on the monocytes in both IS and control groups. There were no significant correlations between NIHSS with IL-6 and IL-10 levels and TLR2 expression on the monocytes.
Correlation between NIHSS with TLR2/TLR4 expression on the peripheral blood monocytes, IL–1β, IL-6, and serum IL–10 levels cytokine levels at day 10
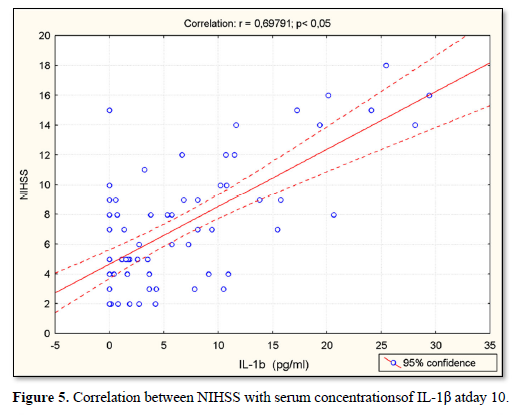
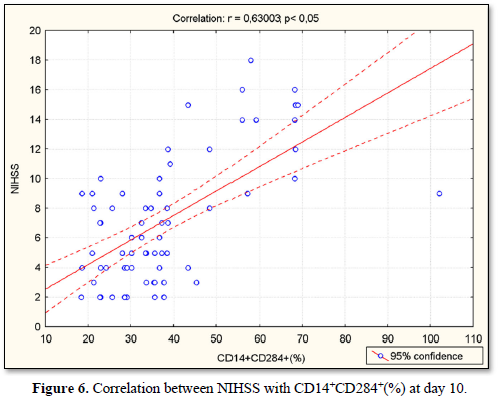
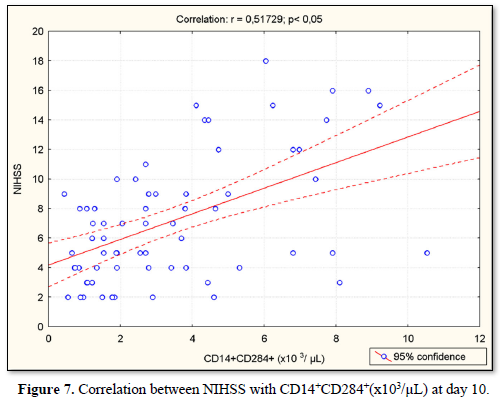
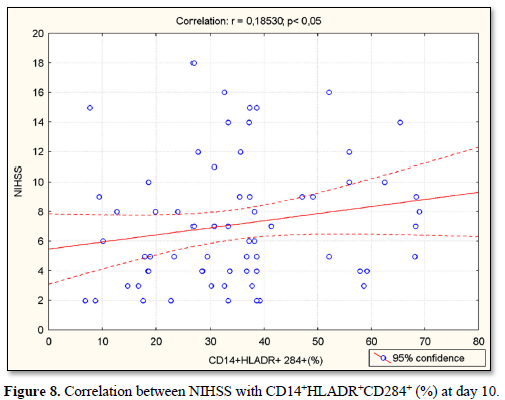
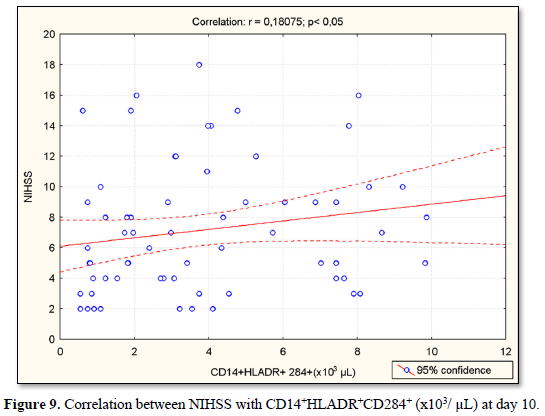
At day 10, NIHSS was positively correlated with serum concentrations of IL-1β (r=0.69; p<0.05) (Figure 5), with CD14+CD284+(%) (r=0.63; p<0.05) (Figure 6), with CD14+CD284+(х103/µL) (r=0.51; p<0.05) (Figure 7), with CD14+HLADR+CD284+ (%) (r=0.19; p<0.05) (Figure 8) and with CD14+HLADR+CD284+ (х103/ µL) (r=0.18; p<0.05) (Figure 9). No significant correlations were found between NIHSS and TLR2 expression on the monocytes. Likewise, no significant correlations were observed between NIHSS and IL-6/IL-10 levels.
DISCUSSION
The results of this study suggest that activated TLR4 signaling on peripheral blood monocytes may possibly have some role in the pathophysiology of IS. A number of observations support this conclusion: (1) Baseline TLR4 expression levels on blood monocytes were similar in IS and in control subjects without IS; (2) Rises in TLR4 expression on blood monocytes in IS patients at day 10 were positively correlated with serum IL-1β levels; (3) Higher expression of TLR4 on blood monocytes was associated with poor functional outcome in IS patients.
Growing data suggest that IS alters the phenotype of the peripheral immune system; therefore, a better understanding of this response could provide new insights into IS pathophysiology. Our observation goes in agreement with the previous study reporting the independent association between increased expression of TLR4 in monocytes and poor outcome after IS in humans [16]. Moreover, previous experimental studies also showed that TLR4-deficient mice had smaller infarctions and less inflammatory response after IS [17] and that the brain damage caused by IS prime mechanisms that signal through TLR4 [18], even so poor outcome in IS was not associated with TLR2 in adjusted models [19].
In this study, we showed that peripheral blood monocytes with TLR4 expression were significantly increased at day 10 after IS and that these laboratory changes were positively correlated with the levels of inflammatory cytokine IL-1β and NIHSS. In IS pathophysiology, both central and peripheral inflammatory alterations occur immediately after cerebral infarction, including mobilization of immature pro-inflammatory Ly6C(hi)CD43(lo) monocytes that acutely infiltrate the ischemic tissue reaching the core of the lesion [19]. In human studies, IS triggered an increased number of total monocytes in the blood circulation; while the number of classical monocyte subsets (CD14++CD16−), significantly increased in blood circulation, whereas the number of the non-classical monocyte subsets (CD14+CD16++) decreased [20]. While most studies implicating the bone marrow as the source of monocytes examined the accumulation of monocyte-derived cells at target sites several days after the tissue inflammation, the recent important study using models of myocardial ischemia has revealed that the spleen represents the dominant reservoir for monocytes during early stages of inflammation [21]. Furthermore, the reduced number of monocyte subsets in the spleen was found to be strongly correlated with their increased number in the ischemic brain [22]. Recently, Hsi-Min Hsiao et al. [23] demonstrated that splenic monocyte-derived IL-1β orchestrates critical steps of acute ischemia-reperfusion injury. It has also been shown that TLR4 activation in monocytes may lead to the blood–brain barrier (BBB) leakage through upregulation and release of proinflammatory cytokines [24] and blood monocytes that are stimulated with the TLR4 agonist can cross the BBB in response to CCL19 [25].
Therefore, our observation could possibly be in agreement with the hypothesis that peripheral blood monocytes with upregulated TLR4 expression may lead to secondary injury when they penetrate the BBB, consequently aggravating brain damage and neural functional deficits [26]. Recently, the first evidence that peripheral immune cells accumulate over 14 days post-stroke at sites of secondary neurodegeneration, triggering microglia activation and progressive neuronal loss, has been provided in one experimental study [27]. The migration of monocytes through the BBB is regulated by CC chemokine receptor 2 (CCR2), which is upregulated on endothelial cells in the ischemic area [28]. Approximately two weeks after IS, the factors released by ischemic neurons, including ATP and Fas ligand, induce morphological changes in local microglia transmigrated peripheral monocytes to promote their more pro-inflammatory M1 phenotype [29]. The M1 phenotype is characterized by the release of inflammatory mediators such as TNFα and IL-1β and reactive oxygen species (ROS) [30]. It was also experimentally demonstrated that activated TLR4 monocytes are able to increase ROS production by upregulating expression of inducible nitric oxide synthase and IL-1β [31]. Moreover, these monocytes are the primary source of IL-1β [32] and it has been clinically confirmed that blocking IL-1β receptor may improve long-term outcomes after IS [33]. In our study, there was a strong positive correlation between serum IL-1β, TLR4 expression on peripheral blood monocytes and NIHSS at day 10 after IS. A well-recognized detrimental action of IL-1β in cerebral ischemia is the induction of adhesion molecules, thus stimulating the infiltration of circulating immune cells and the release or synthesis of other proinflammatory mediators [34].
Interestingly, although serum IL–6 concentrations were higher in our IS patients than in the control subjects after 10 days, we did not observe any correlation between IL-6 levels and TLR4 expression on blood monocytes. Previously, one small clinical study showed that serum IL-6 correlated significantly with infarct size measured by MRI at multiple time points [35]. On the contrary, another study found an inverse relationship between the levels of serum IL-6 and neurological impairment and infarct size three months after IS [36], suggesting that elevated IL-6 may be suggestive of neuroprotection rather than damage. Interestingly, IL-1β, but not IL-6 has been specifically associated with neurotoxic effects after ischemia-reperfusion-induced brain injury [37,38].
In summary, coincident with other researches [5], we show that upregulation of TLR4 expression on peripheral blood monocytes could be one of the peripheral mechanisms of inflammatory injury after IS. Inhibiting harmful immune responses of peripheral monocytes may possibly result in the development of “immunomodulatory strategies” for the better adjunct therapies of ischemic stroke.
STATEMENT OF ETHICS
The study was approved by the local ethics committee at Chita Medical Academy, Russia, and all participants or their legal representatives signed a written informed consent.
FUNDING SOURCES
The manuscript represents the authors’ own work, without external finding.
AUTHOR CONTRIBUTIONS
Krohaleva YuA, Strambovskaya NN, Kuznik BI, contributed to the implementation of the research and to the analysis of the results, Magen E contributed to the design of the research and to the writing of the manuscript.
1. Eltzschig HK, Eckle T (2011) Ischemia and reperfusion--from mechanism to translation. Nat Med Nov 17: 1391-1401.
2. Winters L, Winters T, Gorup D, Mitrečić D, Curlin M, et al. (2013) Expression analysis of genes involved in TLR2-related signaling pathway: Inflammation and apoptosis after ischemic brain injury. Neurosci 238: 87-96.
3. Kaczorowski DJ, Mollen KP, Edmonds R, Billiar TR (2008) Early events in the recognition of danger signals after tissue injury. J Leukoc Biol 83: 546-552.
4. Winters L, Winters T, Gorup D, Mitrečić D, Curlin M, et al. (2013) Expression analysis of genes involved in TLR2-related signaling pathway: Inflammation and apoptosis after ischemic brain injury. Neurosci 238: 87-96.
5. Yang QW, Li JC, Lu FL, Wen AQ, Xiang J (2008) Upregulated expression of toll-like receptor 4 in monocytes correlates with severity of acute cerebral infarction. J Cereb Blood Flow Metab 28: 1588-96.
6. Yang QW, Lu FL, Zhou Y, Wang L, Zhong Q, et al. (2011) HMBG1 mediates ischemia-reperfusion injury by TRIF-adaptor independent Toll-like receptor 4 signaling. J Cereb Blood Flow Metab 31: 593-605.
7. Lin YC, Chang YM, Yu JM, Yen JH, Chang JG, et al. (2005) Toll-like receptor 4 gene C119A but not Asp299Gly polymorphism is associated with ischemic stroke among ethnic Chinese in Taiwan. Atherosclerosis 180: 305-309.
8. Urra X, Cervera A, Obach V, Climent N, Planas AM, et al. (2009) Monocytes are major players in the prognosis and risk of infection after acute stroke. Stroke 40: 1262-1268.
9. Garcia JH, Liu KF, Yoshida Y, Lian J, Chen S, et al. (1994) Influx of leukocytes and platelets in an evolving brain infarct (Wistar rat). Am J Pathol 144: 188-199.
10. Medzhitov R (2001) Toll-like receptors and innate immunity. Nat Rev Immunol 1: 135-145.
11. Offner H, Subramanian S, Parker SM, Afentoulis ME, Vandenbark AA (2006) Experimental stroke induces massive, rapid activation of the peripheral immune system. J Cereb Blood Flow Metab 26: 654-65.
12. Abulafia DP, Vaccari JPDR, Lozano JD, Lotocki G, Keane RW, et al. (2009) Inhibition of the inflammasome complex reduces the inflammatory response after thromboembolic stroke in mice. J Cereb Blood Flow Metab 29: 534-544.
13. Cao CX, Yang QW, Lv FL, Cui J, Fu HB (2007) Reduced cerebral ischemia-reperfusion injury in Toll-like receptor 4 deficient mice. Biochem Biophys Res Commun 53: 509-514.
14. Jauch EC, Saver JL, Adams HP Jr, Bruno A, Connors JJ, et al. (2013) American heart association stroke council; council on cardiovascular nursing; council on peripheral vascular disease; council on clinical cardiology. Guidelines for the early management of patients with acute ischemic stroke: A guideline for healthcare professionals from the american heart association/american stroke association. Stroke 44: 870-947.
15. Urra X, Cervera Á, Obach V, Climent N, Planas AM, et al. (2009) Monocytes are major players in the prognosis and risk of infection after acute stroke. Stroke 40: 1262.
16. Caso JR, Pradillo JM, Hurtado O, Lorenzo P, Moro MA, et al. (2007) Toll-like receptor 4 is involved in brain damage and inflammation after experimental stroke. Circulation 115: 1599-1608.
17. Caso JR, Pradillo JM, Hurtado O, Leza JC, Moro MA, et al. (2008) Toll-like receptor 4 is involved in subacute stress-induced neuro inflammation and in the worsening of experimental stroke. Stroke 39: 1314-1320.
18. Li M, Carpio DF, Zheng Y, Bruzzo P, Singh V, et al. (2001) An essential role of the NF-kappa B/Toll-like receptor pathway in induction of inflammatory and tissue-repair gene expression by necrotic cells. J Immunol 166: 7128-7135.
19. Miró-Mur F, Pérez-de-Puig I, Ferrer-Ferrer M, Urra X, Justicia C, et al. (2016) Immature monocytes recruited to the ischemic mouse brain differentiate into macrophages with features of alternative activation. Brain Behav Immun 53: 18-33.
20. Kaito M, Araya S, Gondo Y, Fujita M, Minato N, et al. (2013) Relevance of distinct monocyte subsets to clinical course of ischemic stroke patients. PLoS One 8: e69409.
21. Swirski FK, Nahrendorf M, Etzrodt M, Wildgruber M, Cortez-Retamozo V, et al. (2009) Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science 325: 612-616.
22. Kim E, Yang J, Beltran CD, Cho S (2014) Role of spleen-derived monocytes/macrophages in acute ischemic brain injury. J Cereb Blood Flow Metab 34: 1411-1419.
23. Hsiao HM, Fernandez R, Tanaka S, Li W, Spahn JH (2018) Spleen-derived classical monocytes mediate lung ischemia-reperfusion injury through IL-1β. J Clin Invest 128: 2833-2847.
24. Zhou H, Lapointe BM, Clark SR, Zbytnuik L, Kubes P (2006) A requirement for microglial TLR4 in leukocyte recruitment into brain in response to lipopolysaccharide. J Immunol 177: 8103-8110.
25. Paradis A, Bernier S, Dumais N (2016) TLR4 induces CCR7-dependent monocytes transmigration through the blood-brain barrier. J Neuro Immunol 296: 12-17.
26. O'Connell GC, Chang JHC (2018) Analysis of early stroke-induced changes in circulating leukocyte counts using transcriptomic deconvolution. Trans Neurosci 9:161-166
27. Jones KA, Maltby S, Plank MW, Kluge M, Nilsson M, et al. (2018) Peripheral immune cells infiltrate into sites of secondary neurodegeneration after ischemic stroke. Brain Behav Immunol 67: 299-307.
28. Kuziel WA, Morgan SJ, Dawson TC, Griffin S, Smithies O, et al. (1997) Severe reduction in leukocyte adhesion and monocyte extravasation in mice deficient in CC chemokine receptor 2. Proc Natl Acad Sci USA 94: 12053-12058.
29. Meng HL, Li XX, Chen YT, Yu LJ, Zhang H, et al. (2016) Neuronal Soluble Fas Ligand Drives M1-Microglia Polarization after Cerebral Ischemia. CNS Neurosci Ther 22: 771-781.
30. del Zoppo GJ, Milner R, Mabuchi T, Hung S, Wang X, et al. (2007) Microglial activation and matrix protease generation during focal cerebral ischemia. Stroke 38: 646-651.
31. Deng S, Yu K, Zhang B, Yao Y, Wang Z, et al. (2015) Toll-Like Receptor 4 Promotes NO Synthesis by Upregulating GCHI Expression under Oxidative Stress Conditions in Sheep Monocytes/Macrophages. Oxid Med Cell Longev 2015: 359315.
32. Ritzel RM, Patel AR, Grenier JM, Crapser J, Verma R, et al. (2015) Functional differences between microglia and monocytes after ischemic stroke. J Neuro Inflammation 12: 106.
33. Touzani O, Boutin H, Chuquet J, Rothwell N (1999) Potential mechanisms of interleukin-1 involvement in cerebral ischaemia. J Neuro Immunol 100: 203-215.
34. Emsley HC, Smith CJ, Georgiou RF, Vail A, Hopkins SJ, et al. (2005) Acute stroke investigators: A randomised phase II study of interleukin-1 receptor antagonist in acute stroke patients. J Neurol Neurosurg Psychiatr 76: 1366-1372.
35. Shaafi S, Sharifipour E, Rahmanifar R, Hejazi S, Andalib S, et al. (2014) Interleukin-6, a reliable prognostic factor for ischemic stroke. Iran J Neurol 13: 70-76.
36. Sotgiu S, Zanda B, Marchetti B, Fois ML, Arru G, et al. (2006) Inflammatory biomarkers in blood of patients with acute brain ischemia. Eur J Neurol 13: 505-513.
37. Lambertsen KL, Biber K, Finsen B (2012) Inflammatory cytokines in experimental and human stroke. J Cereb Blood Flow Metab 32: 1677-1698.
38. Liberale L, Diaz-Cañestro C, Bonetti NR, Paneni F, Akhmedov A, et al. (2018) Post-ischaemic administration of the murine Canakinumab-surrogate antibody improves outcome in experimental stroke. Eur Heart J 39: 3511-3517.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Cancer Science and Treatment (ISSN:2641-7472)
- Journal of Nursing and Occupational Health (ISSN: 2640-0845)
- Journal of Oral Health and Dentistry (ISSN: 2638-499X)
- Journal of Infectious Diseases and Research (ISSN: 2688-6537)
- Chemotherapy Research Journal (ISSN:2642-0236)
- Advance Research on Alzheimers and Parkinsons Disease
- Journal of Allergy Research (ISSN:2642-326X)