2840
Views & Citations1840
Likes & Shares
Purpose: Orbital tumors are relatively rare. Surgical resection of these tumors can be challenging on account of the complexity of intraorbital anatomy. Intraoperative injuries to intraorbital structures may result in severe complications; the surgical approach must therefore be carefully selected. We present a case series of 120 patients surgically treated for orbital tumors between 1990 and 2005.
Patients and methods: All cases were reviewed using imaging and histopathological analysis. Among the three surgical approaches considered, we have identified the most suitable surgical approach for each case: an anterior approach was used in 10 cases, a lateral approach in 53 cases, and a transcranial approach in 57 cases. Except for tumors diagnosed as malignant or inflammatory orbital pseudotumors on intraoperative histopathological diagnosis, tumors were completely resected.
Results: Postoperatively, three patients experienced visual impairments, three exhibited tonic pupil, and two developed permanent eye movement impairment.
Conclusion: In our case series, the indication for a lateral approach was more prevalent than in previous reports. This approach, however, is not suitable for tumors in the medial compartment of the orbit or for tumors extending beyond the orbital apex. The transcranial approach is suitable for tumors in the medial compartment of the orbit, as well as those extending intracranially. The indication for an anterior approach was relatively limited; it was only used for tumors on the anterior half of the globe. Orbital tumors can be safely removed using the appropriate surgical approach.
Keywords: Intraorbital neoplasms, Lateral approach, Transcranial approach, Anterior approach
Abbreviations: CT: Computed Tomography; MRI: Magnetic Resonance Imaging
INTRODUCTION
Orbital tumors are relatively rare [1]; a reliable estimate of the incidence of orbital lesions is difficult to obtain, as the reported incidence varies greatly [2]. Left unaddressed, these tumors can cause severe ophthalmological deficits, including loss of vision, difficulties in eyeball movement and exophthalmos [3]; thus, treating patients with symptomatic orbital tumors is critical.
The complex anatomy of the orbit and the bony walls surrounding it hinder access for tumor removal. Furthermore, intra- and extra-orbital structures are closely associated with visual functions and eyeball movement; damage to these structures may result in severe functional or cosmetic problems for the patient. Surgeons must therefore minimize complications by carefully selecting the best approach for the removal of orbital tumors.
In our case series (26 cases) published in 1996, we proposed that the relationship between the tumor and the optic nerve should inform selection criteria [4]. Subsequent large-scale surgical series have reported on surgical approaches: a recent major innovation has the potential to improve surgical intervention for orbital lesions; the endoscopic transnasal approach uses naturally present corridors and employs high-quality video to provide a clear surgical field [5]. However, this method is limited to tumors located medially to the orbit. A recent report suggests that surgeons tend to select the transcranial approach; it provides an optimal exposure of tumors and is relatively less invasive than the lateral approach [6].
The existing literature, however, does not provide adequate criteria for choosing among surgical approaches for orbital tumors. We report a review of our consecutive case series and discuss the selection of a surgical intervention based on a tumor’s anatomical location.
MATERIALS AND METHODS
Clinical materials and methods
A retrospective review of 120 cases was performed. We included patients who had undergone surgical resection of orbital tumors at our institution between 1990 and 2005. Data were collected from hospital records, outpatient clinic records and operative notes. All patients provided written informed consent. The study was approved by the institutional review board of Juntendo University and conformed to the Declaration of Helsinki.
All patients were examined using computed tomography (CT) and magnetic resonance imaging (MRI) to determine tumor location and size. Neuro-ophthalmic functions, such as ocular fundus, visual acuities, visual fields and Hess charts, were also assessed before tumor removal. These assessments informed our selection of surgical approach.
For histopathological diagnosis, tissue samples were stained with hematoxylin and eosin and examined by neuropathologists under a light microscope. If necessary, immunohistological stains were used for a definitive diagnosis.
Postoperative assessments were performed in the outpatient clinic. Patients were given general and neuro-ophthalmological examinations. Neuroradiological evaluations (MRI and/or CT) were performed within 2 weeks following surgery. Tumor resection rates were determined with postoperative MRIs.
Surgical technique
We selected one of three surgical approaches for each case: (1) lateral, (2) transcranial, and (3) anterior [7,8]. The details for each approach are described in the following sections.
Lateral approach
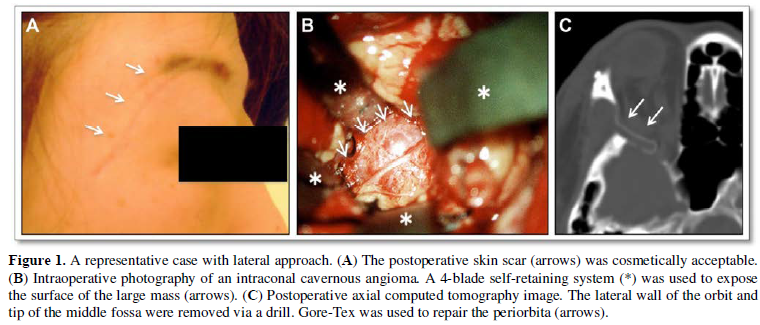
Surgical procedures for lateral orbitotomy have been described previously [4]. An S-shaped skin incision was made superior and lateral to the eyebrow, posteriorly along the upper margin of the zygomatic arch (Figure 1A). We performed osteotomies above and one below the frontozygomatic suture; the lower osteotomy was at the level of the upper margin of the zygomatic arch. We further removed segments of the sphenoidal bone to reveal lesions within the muscle cone.
The orbital fat was separated until most of the tumor surface was visible. Several self-retaining retractors were used to hold the dissected orbital fat (Figure 1B). If the lesions were circumscribed or encapsulated - as is the case in cavernous angiomas - en bloc resections were possible with the dissection of orbital fat and intraorbital structures. If the tumor was large and/or attached to the optic nerve, internal debulking between the tumor and optic nerve was performed before dissection.
After tumor resection, the orbital contents were covered with a Gore-Tex membrane, as the periorbita is very fragile: in most cases, it is disrupted and resutured (Figure 1C). Finally, the lateral orbital rim was fixed with titanium plates and screws. The lateral orbital wall was not replaced with prosthetic material.
Transcranial approach
For the transcranial approach, a typical bicoronal skin incision was made, and the skin flap was elevated. The pericranial flap was prepared for placement over a defect in the frontal fossa at the end of surgery, because the frontal or ethmoid sinus was opened during craniotomy in most cases. The supraorbital nerve was dislocated in a subperiosteal manner and wedged from the supraorbital notch to preserve sensation in the forehead. After the typical frontotemporal craniotomy, the superior orbital rim and orbital roof were removed. When the tumor was situated in the orbital apex or in the optic canal, the entire orbital roof was excised. Unroofing of the optic canal was then performed in an extradural manner. The periorbita was subsequently incised, and the orbital fat was dissected down to the tumor with two or three blades of a self-retaining system. The route into the muscle cone traversed between the complex of the levator and superior rectus muscles and superior oblique muscles. The tumor was removed with microsurgical dissection, as described for the lateral approach. When tumors, such as optic sheath meningiomas, extended into the optic canal, the cranial dura and optic sheath were opened to remove the tumor. The annual tendon was incised medial to the levator and superior rectus muscles to avoid injury to the oculomotor nerve, which passes through the oculomotor foramen lateral to these muscles.
After tumor resection, the orbital contents were covered with an artificial membrane as described for the lateral approach. In cases with annual tendon opening, it was reapproximated with two or three sutures. Finally, the superior orbital rim and cranial bone flap were fixed with titanium plates and screws. Except in patients in whom both the orbital roof and lateral wall were removed, the orbital roof was not replaced with a prosthetic material.
Anterior approach
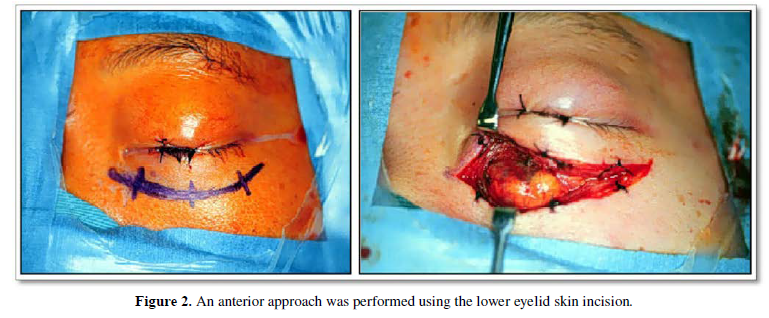
The anterior approach typically includes transconjunctival and skin approaches [7,9]. In this report, we excluded the former. Skin approaches were divided into superior and inferior. An upper or lower eyelid skin incision was made depending on the tumor location (Figure 2). If possible, the eyelid crease incision was used to improve cosmetic appearance after surgery. After skin incision, the subcutaneous layer was undermined to expose the periosteum. The lesion was then approached extraperiosteally. The periosteum was reflected and the periorbita was incised to gain access to the lesion. If possible, the periorbita was closed once tumor resection was accomplished. The orbicularis muscle, subcutaneous tissues, and skin were then approximated. If periorbital repair was not possible, the periorbita was left as it was.
DATA ANALYSIS
We reviewed the criteria for the surgical approaches for tumors from our hospital records. As our review was based on the relationships between the tumor and optic nerve, we classified tumors into three types based on anatomical locations: (1) medial to the optic nerve, (2) lateral to the optic nerve and (3) anterior to the globe.
RESULTS
Patients and lesion characteristics
The mean patient age was 54.2 ± 14.2 years (range, 1–72 years). We recruited a total of 120 patients (65 women, 55 men). Presenting symptoms were as follows: proptosis, 113 patients (94.1%); diplopia, 47 patients, (39.2%); visual impairment, 30 patients (25%); and pain, 20 patients (16.7%) (Table 1A). Pathological diagnoses for the patients are summarized in Table 1B.
Surgery and outcome
The lateral approach was performed in 53 cases, the transcranial approach in 57 cases, and the anterior approach in 10 cases. Except in cases of diffuse malignant tumors, such as inflammatory orbital pseudotumors and malignant lymphomas, tumors were completely resected. Table 2 summarizes postoperative symptoms and the approaches that may have caused them. Three patients experienced postoperative visual impairment, three patients experienced tonic pupil, and 12 patients experienced impaired eye movement (10 transient and 2 permanent). Tonic pupil was only seen in patients with intraconal tumors located in the lateral orbital apex that were removed using a lateral approach. Most symptoms, however, resolved after tumor removal, particularly those associated with visual function. None of the patients died of the surgical procedure.
DISCUSSION
Historically, both ophthalmologists and neurosurgeons have managed intraorbital tumors [8]. However, the two fields have diverged in their approach for treating such tumors: representing an ophthalmological viewpoint, Bernardini et al reported that lacrimal gland tumors were mainly treated with an anterior approach [10]; arguing from a neurosurgical perspective, Maroon et al reported that meningiomas were removed using a transcranial approach [8,10]. In both these reports, presurgical assessment of the tumor size, demarcation, and type informed the approach. Our analysis used tumor location to determine the best surgical approach.
Indications for surgical procedure
Surgical treatment of orbital tumors is indicated for patients with symptoms such as proptosis, visual disturbance, orbital pain, and eye movement disturbance [8]. Some benign lesions, such as orbital varices with minimum symptoms, benign circumscribed tumor without symptoms, optic sheath meningiomas, and optic gliomas with useful vision, should be managed conservatively [11]; patients with these tumors should be monitored while visual function is intact, because there is a high possibility that surgical resection will result in loss of visual acuity [12]. This is particularly important for patients with optic sheath meningiomas and optic gliomas.
In this series, patients with benign tumors, such as cavernous angiomas, neurinomas, optic gliomas, and optic sheath meningiomas, showed painless proptosis (37/38, 97%). On the other hand, patients with malignant tumors, such as lymphoid lesions and inflammatory processes, tended to complain about pain (19/20, 95%). Pain may therefore be used as a symptom in preoperative diagnosis.
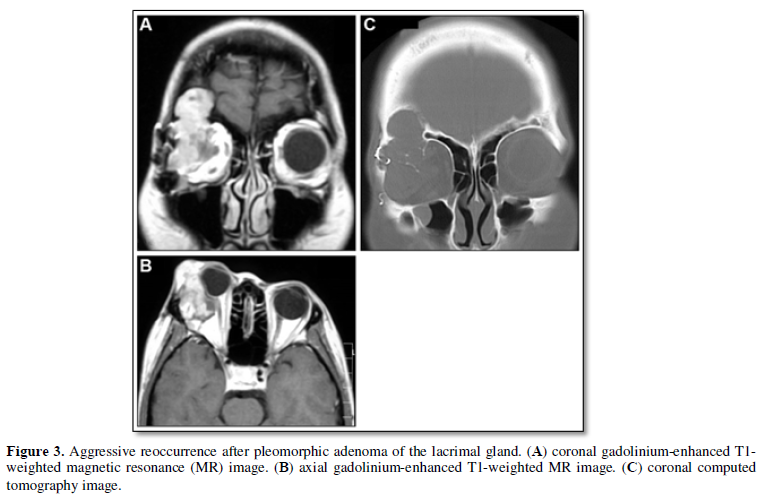
Surgical procedures for treating orbital tumors include needle biopsy, incisional biopsy, debulking surgery, excisional biopsy, and orbital exenteration. Needle biopsies or incisional biopsies are indicated for patients with invasive malignant tumors, such as rhabdomyosarcoma, diffuse metastatic tumors, lymphoid tumors, and extensive poorly circumscribed malignant epithelial tumors of the lacrimal gland, as surgical reduction of tumor size dose not play a main role in the treatment of these tumors [13]. However, in patients in whom pleomorphic adenoma of the lacrimal gland is suspected, incisional biopsy should be avoided, as surgical rupture of the capsule may lead to recurrence; this is particularly pertinent for patients with slow progression, absence of pain, and bony erosion (Figure 3) [14,15]. In these cases, a complete en bloc excision decreases the chance of recurrence and malignant transformation.
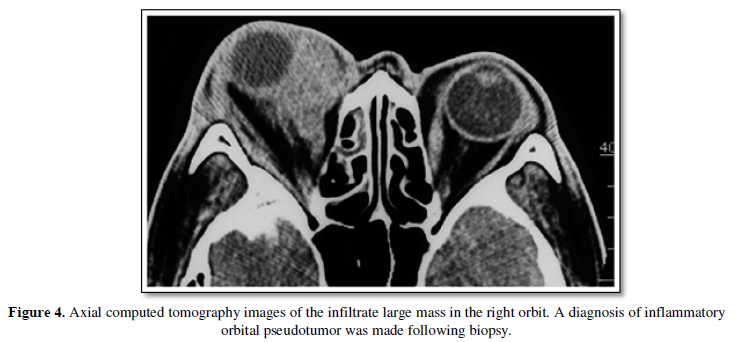
On the basis of histopathological analysis, a second surgery may be performed after biopsies. In this series, orbital exenterations were performed after biopsy in eight patients. Diagnostic biopsies were also performed for patients with inflammatory orbital pseudotumors, which are difficult to differentiate from infiltrative tumors (Figure 4).
Choice of surgical approaches
The choice of the appropriate surgical approach for orbital tumors is critical for minimizing complications and achieving satisfactory therapeutic results [8].
Lateral approach
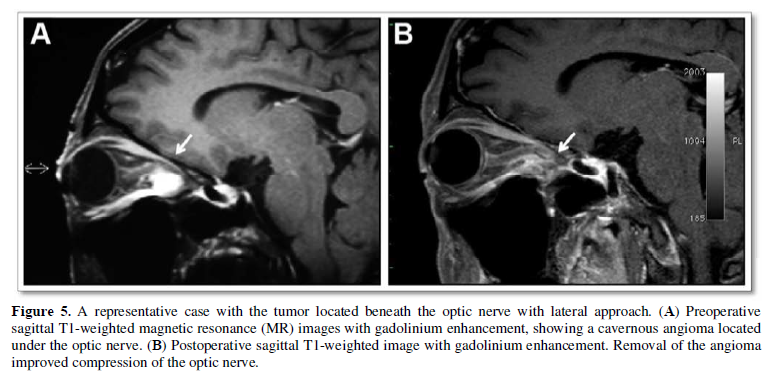
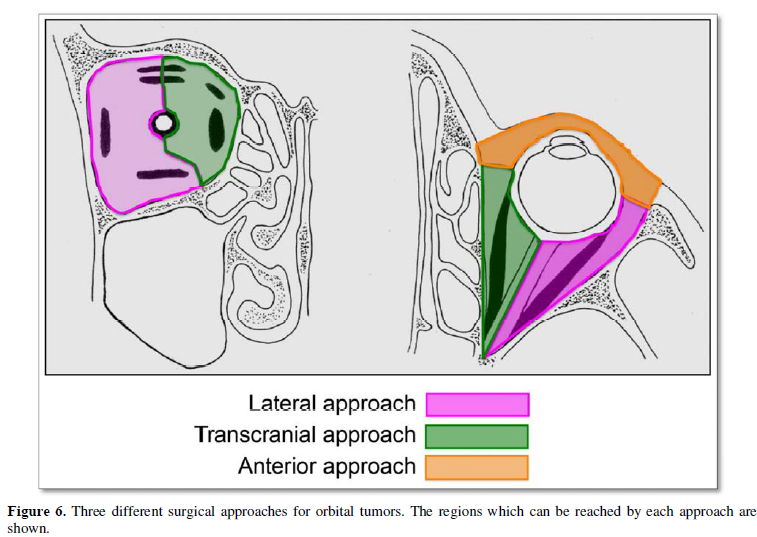
The lateral orbital approach has primarily been considered suitable for lesions confined to the lateral aspect of the orbit, such as lacrimal gland tumors [4,8,16]. However, we found that a modifying this approach by extending lateral orbitotomy provides surgeons a better view and manipulating space for the deep lateral portion of the muscle cone. With this additional step, this approach could be applied to tumors located in the lateral apex, intraconal tumors, and tumors located beneath the optic nerve (Figure 5) [17]. Unless the tumor extends to the superior orbital fissure or optic canal, these tumors can be removed safely through lateral orbitotomy. For tumors located beneath the optic nerve, the lateral approach may be the only approach (Figure 5, Figure 6) [17,18].
The lateral approach may, however, produce cosmetic complications: an incision has to be performed on the patient’s face, and the removal of the superior or lateral wall of the orbit could result in enophthalmos [4]. In our series, even among patients in whom the orbital roof or lateral wall was not replaced with a prosthetic material, enophthalmos was not observed. Further, wounds healed with minimal scarring within three months.
Transcranial approach
The transcranial approach has been widely employed for all orbital tumors [8]. Neurosurgeons favor this method for tumors with intracranial extension, [19,20] tumors located in the orbital apex or optic canal, [14,19,21-23] and intracranial tumors with extension into the orbit [20,24]. The transcranial approach is also suitable for tumors located in the medial compartment of the orbit (Figure 6) and may be used to resect tumors located in the lateral compartment [25]. The lateral approach may, however, provide an effective alternative for lateral tumors; craniotomy is much more invasive than lateral orbitotomy [17].
In the present investigation, the transcranial approach was used for intraconal tumors medial to the optic nerve, including tumors existing in the medial compartment of the orbit and/or extending intracranially through the optic canal or superior orbital fissure. For all eight patients with optic sheath meningioma, the optic nerve was extirpated with the tumor; indication for removal of these tumors is thus limited to patients without useful visual function.
Anterior approach
The anterior approach is indicated for anterior orbital tumors [7]. This approach has the advantage of exposing the tumor without orbitotomy. However, once the tumor extends behind the globe, the operative field is limited with this method. The anterior approach should therefore be limited to tumors that are located in the anterior half of the globe (Figure 6) [14].
Surgical complications
Postoperative visual impairments were observed in three patients; though such cases may have been due to the compromise of the central retinal artery or undue retraction of the optic nerve, the cause remains unclear. Central retinal arteries penetrated the optic sheath at or near the junction of the middle and anterior third of the length of the intraorbital optic nerve an average of 18.6 mm distal to the optic canal [26]. Preservation of the retinal artery is critical if the tumor extends below the optic nerve.
We observed only one symptom associated with a specific approach in our series: three patients who developed tonic pupil postoperatively had intraconal cavernous angiomas removed through the space between the lateral and inferior rectus muscles using the lateral approach. Tonic pupil was believed to be caused by injuries to ciliary nerves and ganglion. The ciliary ganglion is located on the lateral side of the optic nerve and gives rise to the short ciliary nerves that run above and below the optic nerve to reach the posterior surface of the globe [4]. The lateral approach thus presents the potential risk of injury.
Postoperative eye movement impairment occurred in response to traumatic retraction or stress to the extraocular muscles and/or branches of the oculomotor, abducens, and trochlear nerves. When the intraconal compartment is exposed, retraction of the extraocular rectus muscle is necessary to provide an adequate view of the target area. Severe retraction of these muscles results in impairment of eye movement; however, impairment is transient in most cases [27]. On the other hand, damage to nerves innervating extraocular muscles may result in permanent eye movement impairment.
Special care needs to be taken to preserve nerves; for instance, when the medial region of the orbital apex is exposed, the anatomical path of the trochlear nerve has to be considered. This nerve passes through the superior orbital fissure and then traverses medially above the levator muscle to reach the superior oblique muscle [4]. As trochlear nerve palsy does not result in cosmetic complications following the loss of visual function, the nerve was removed along with the optic nerves in all eight patients with optic sheath meningioma.
When opening the posterior third of the muscle cone, the relationship between the abducens and oculomotor nerves should be considered. The abducens nerve traverses through the annulus inferolateral to the oculomotor nerve, passes close to the lateral rectus muscle, and then enters its medial surface [4]. The risk of injury to the abducens nerve is much lower than that to the oculomotor and trochlear nerves because of their anatomical length. The branch to the inferior oblique muscle can occur when the anterior two-thirds of the muscle cone is exposed through the inferior route [4].
In this study, we did not discuss the transconjuctive and transnasal approaches; when treating large tumors, the former requires transecting more ocular muscle, which severely limits eye movements [27,28]. Though the transnasal approach may have been effective in our patients with tumors located immediately medial to the orbit without extension into the intracranial space, the overexpansion of its indication could cause severe complications; we therefore used the transcranial approach in such cases [5].
CONCLUSIONS
On the basis of this review of 120 patients, we suggest that orbital tumors can be safely removed with the proper surgical approach. The lateral approach is appropriate for tumors in the lateral compartment of the orbit, as well as for those extending over half of the orbit, optic nerve, and orbital apex. The transcranial approach is optimal for tumors in the medial compartment of the orbit and those extending intracranially through the optic canal or superior orbital fissure. The anterior approach is useful only for tumors situated on the anterior half of the globe.
ETHICS APPROVAL AND INFORMED CONSENT
This study was approved by the institutional review board of Juntendo University. All patients provided written informed consent.
DATA AVAILABILITY
All data related to this study is present in the manuscript.
FUNDING
This study was supported by Juntendo University Research Institute for Diseases of Old Age (Tokyo, Japan). The funding source had no role in any of the stages of the study.
DISCLOSURE
The author reports no conflicts of interest in this work.
ACKNOWLEDGMENTS
We acknowledge all the members of the Department of Neurosurgery, Jutendo University Faculty of Medicine, Tokyo, Japan.
1. Ohtsuka K, Hashimoto M, Suzuki Y (2005) A review of 244 orbital tumors in Japanese patients during a 21-year period: origins and locations. Jpn J Ophthalmol 49: 49-55.
2. Shields JA, Shields CL, Scartozzi R (2004) Survey of 1264 patients with orbital tumors and simulating lesions: The 2002 Montgomery Lecture, part 1. Ophthalmol 111: 997-1008.
3. Demirci H, Shields CL, Shields JA, Honavar SG, Mercado GJ, et al. (2002) Orbital tumors in the older adult population. Ophthalmol 109: 243-248.
4. Arai H, Sato K, Katsuta T, Rhoton AL Jr. (1996) Lateral approach to intraorbital lesions: anatomic and surgical considerations. Neurosurg 9: 1157-1162.
5. Bleier BS, Castelnuovo P, Battaglia P, et al. (2016) Endoscopic endonasal orbital cavernous hemangioma resection: global experience in techniques and outcomes. Int Forum Allergy Rhinol 6: 156-161.
6. Scheuerle AF, Steiner HH, Kolling G, Kunze S, Aschoff A (2004) Treatment and long-term outcome of patients with orbital cavernomas. Am J Ophthalmol 138: 237-244.
7. Cockerham KP, Bejjani GK, Kennerdell JS, Maroon JC (2001) Surgery for orbital tumors. Part II: transorbital approaches. Neurosurg Focus 10: E3.
8. Maroon JC, Kennerdell JS (1984) Surgical approaches to the orbit: Indications and techniques. J Neurosurg 60: 1226-1235.
9. Westfall CT, Shore JW, Nunery WR, Hawes MJ, Yaremchuk MJ (1991) Operative complications of the transconjunctival inferior fornix approach. Ophthalmol 98: 1525-1528.
10. Bernardini FP, Devoto MH, Croxatto JO (2008) Epithelial tumors of the lacrimal gland: an update. Curr Opin Ophthalmol 19: 409-413.
11. Wulc AE, Bergin DJ, Barnes D, Scaravilli F, Wright JE, et al. (1989) Orbital optic nerve glioma in adult life. Arch Ophthalmol 107: 1013-1016.
12. Smee RI, Schneider M, Williams JR (2009) Optic nerve sheath meningiomas—non-surgical treatment. Clin Oncol (R Coll Radiol) 21: 8-13.
13. Tijl JW, Koornneef L (1991) Fine needle aspiration biopsy in orbital tumours. Br J Ophthalmol 75: 491-492.
14. Bejjani GK, Cockerham KP, Kennerdel JS, Maroon JC (2001) A reappraisal of surgery for orbital tumors. Part I: extraorbital approaches. Neurosurg Focus 10: E2.
15. Finger PT (2009) Radiation therapy for orbital tumors: concepts, current use, and ophthalmic radiation side effects. Surv Ophthalmol 54: 545-568.
16. Maroon JC, Kennerdel JS (1976) Lateral microsurgical approach to intraorbital tumors. J Neurosurg 44: 556-561.
17. Hassler W, Schick U (2009) The supraorbital approach—a minimally invasive approach to the superior orbit. Acta Neurochir (Wien) 151: 605-611.
18. Carta F, Siccardi D, Cossu M, Viola C, Maiello M (1998) Removal of tumours of the orbital apex via a postero-lateral orbitotomy. J Neurosurg Sci 42: 185-188.
19. Love JG, Dodge HW Jr (1953) Transcranial removal of intraorbital tumors. AMA Arch Surg 67: 370-380.
20. Van Buren JM, Poppen JL, Horrax G (1957) Unilateral exophthalmos; a consideration of symptom pathogenesis. Brain 80: 139-175.
21. Craig WM, Gogela LJ (1950) Meningioma of the optic foramen as a cause of slowly progressive blindness; report of three cases. J Neurosurg 7: 44-48.
22. Housepian EM (1969) Surgical treatment of unilateral optic nerve gliomas. J Neurosurg 31: 604-607.
23. Wilson WB, Gordon M, Lehman RA (1979) Meningiomas confined to the optic canal and foramina. Surg Neurol 12: 21-28.
24. Dandy WE (1941) Results following the transcranial operative attack on orbital tumors. Arch Ophthalmol 25: 191-216.
25. Natori Y, Rhoton AL Jr (1994) Transcranial approach to the orbit: microsurgical anatomy. J Neurosurg 81: 78-86.
26. Tsutsumi S, Rhoton AL Jr (2006) Microsurgical anatomy of the central retinal artery. Neurosurg 59: 870-878.
27. Cha DS, Lee H, Park MS, Lee JM, Baek SH (2010) Clinical outcomes of initial and repeated nasolacrimal duct office-based probing for congenital nasolacrimal duct obstruction. Korean J Ophthalmol 24: 261-266.
28. García JC, Pérez IM, Perdomo FA, Fernández NG, Álvarez AMG (2017) Endonasal endoscopic approach for primary and secondary orbital lesions. J Clin Exp Ophthalmol 8: 668.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Carcinogenesis and Mutagenesis Research (ISSN: 2643-0541)
- Journal of Psychiatry and Psychology Research (ISSN:2640-6136)
- Journal of Allergy Research (ISSN:2642-326X)
- Journal of Nursing and Occupational Health (ISSN: 2640-0845)
- Journal of Ageing and Restorative Medicine (ISSN:2637-7403)
- Archive of Obstetrics Gynecology and Reproductive Medicine (ISSN:2640-2297)
- BioMed Research Journal (ISSN:2578-8892)