2505
Views & Citations1505
Likes & Shares
The long-term outcome of ART-naïve people living with HIV (PLHIV) with baseline CD4 count ≥ 350 is not well described. We aimed to analyze mortality in an ART-naïve West African cohort of PLHIV followed for 18 years according to the effects of CD4 cell count stratum (350-499 versus ≥ 500), HIV serotype and immune activation (IA).
The cumulative incidences of death in this cohort were calculated using competing risk estimators; treating ART initiation as a competing event for death. To evaluate the effect of baseline CD4 cell counts, HIV serotype and beta 2 microglobulin (β2m) level on mortality with the presence of competing event, we used competing risks regression models to calculate the corresponding sub-distribution hazard ratios (sHR) with adjustment for confounding bias.
During 18 years of follow up, the 908 patients realized 5367 clinical visits totaling 2138.1 person-years (PY) of exposure time. A total of 160 deaths was recorded and 67 patients initiated ART; resulting in a mortality incidence rate of 7.5/100 PY. After adjustment, baseline CD4 cell counts between 350 and 499/µL was associated with a 49% increase in mortality compared to baseline CD4 cell counts ≥ 500/µL (adjusted sHR (asHR)=1.49; 95% CI=1.07-2.07). HIV serotype was also associated with mortality (the asHR were respectively 1.66 (95% CI=1.14-2.43) and 2.15 (95% CI=1.25-3.73) for HIV-1 and HIV-1+HIV-2 dual infection (HIV-D) compared to HIV-2 infection). Mortality was also associated with β2m level (≥ 2.85 mg/L compared to
Our study provides a picture of real-life long-term differences in mortality in a context of high prevalence of HIV and immune activation. It confirms the benefits of early initiation of ART found in randomized clinical trials (RCT) and suggests a potential benefit of the reduction of IA level in African PLHIV.
Keywords: ART-naïve, Mortality, CD4 cell-stratum, HIV serotype, Immune activation
INTRODUCTION
In developed countries, cohort analyses have evaluated mortality and morbidity in antiretroviral therapy (ART) naïve people living with HIV (PLHIV) by CD4 cell counts strata [1-6]. Such data are very scarce in Sub Saharan Africa: the few existing studies are poorly powered due to short durations of follow up or limited sample sizes [7-11] particularly in patients with high CD4 cell count. Therefore, their interpretation is difficult.
The debate on the best moment to initiate ART remains of interest in resource-limited countries (RLC). This is despite the WHO recommendations for universal ART that is every PLHIV should receive ART regardless of CD4 cell count and clinical stage [12]. Such recommendations were strongly influenced by the randomized clinical trials (RCT) HPTN 052, START and TEMPRANO [13-15]. These RCTs were globally designed to compare early ART initiation (CD4 cell count ≥ 500/µL) and deferred ART initiation (CD4 cell count <500/µL or <350/µL or WHO clinical stage 3/4). The modest clinical benefits reported in these studies suggest that treatment regardless of CD4 may not translate into broader public health benefits in RLC for the following reasons. First, previous observational studies reported conflicting results [16-18] in line with a previous systematic review that recommended initiating ART at 350-500 CD4/µL, not above 500 [19]. Second, subsequent studies showed that there was no statistically significant difference in mortality between early ART and deferred ART groups in HPTN 052, START and TEMPRANO [13-15]. Third, ART is a lifelong treatment whose effectiveness over the long term can be compromised by adherence issues, drug resistance, long-term side effects, and costs: antiretroviral drugs are expensive and require sustainable funding mechanisms. Additional costs in terms of service providers, monitoring tools, additional health infrastructures and their impact on health systems with insufficient resources may be important barriers to the feasibility of universal ART in RLC. The current financial environment poses significant challenges for funding universal treatment and sustaining existing treatment programs. Considering these concerns, observational studies done in “real world” conditions may be a more reliable guide to the impact of treatment guidelines than RCTs, which were conducted in highly resourced settings.
It has also been shown that mortality varied by HIV serotype: HIV-2 infection is associated with lower mortality than HIV-1 infection [20-22] and HIV-1+HIV-2 dual infection (HIV-D) [23]. Some studies claim that co-infection with HIV-2 slows the progression of HIV-1 infection while others refute this theory [23-28].
Recent data on the pathogenesis of HIV argue that chronic immune activation is the main factor driving the progression of HIV infection [29]. Despite this fact, it was not sufficiently studied in Sub-Saharan Africa.
Our study is the first one to evaluate the clinical impact of early treatment in African patients in a “real-world” setting. We aimed to describe mortality in an ART-naïve West African cohort with high baseline CD4 (≥ 350) followed for 18 years. The effects of CD4 cell count stratum (350-499 versus ≥ 500), HIV serotype and immune activation were determined in order to provide a more accurate picture of these long-term differences.
METHODS
We conducted a prospective cohort study. Our source population was the Fajara cohort which has been previously described [30,31]. It was a clinical cohort attached to the genito-urinary medicine unit of the Medical Research Council (MRC) clinic in Banjul, Gambia which was the HIV national reference center.
Between 1988 and 2010, 4078 patients found to be HIV positive were included in this observational cohort and followed until November 2010. They were included in our analysis if: 1) they were aged >16 years; 2) they had a measure of CD4 cell count in a period of three months following their entry in the cohort; 3) the initial CD4 cell counts was ≥ 350/mm3; and 4) duration of follow up was >1 day.
Data collection
After inclusion, patients were invited to attend the clinic at least once every three months, irrespective of symptoms. Those who failed to attend the clinic were visited at home by a fieldworker and were encouraged to return to the clinic. Field workers made quarterly visits up country to enroll individuals referred from outside of the greater Banjul area.
During these clinic visits including baseline visit, clinical data (weight, height and WHO clinical stage) were collected. Socio-demographic data (age, sex, marital status, education and occupation) were also collected at recruitment (baseline). ART became available from 2004 onwards and start dates for ART were collected during clinical visits. Blood sample was collected at baseline visit and at according to the physician in the course of follow-up for CD4 count testing. HIV serotype testing was determined for all patients. Blood samples of a subset of patients were bio-banked and used for immune activation testing.
The study protocol was approved by the Institutional Ethical and Research Review Boards of the participating institutions in Senegal: (Comité national d’éthique pour la recherche en santé (CNERS) of the Ministry of Health) and in Canada (Research Ethical Board of Sainte-Justine University Hospital, Montreal).
HIV serotype testing
The following serological tests were employed to determine HIV sero-status at baseline.
Until August 1996, Wellcozyme HIV 1+2 (Murex Diagnostics Ltd) tests were used. If reactive (positive), samples were re-tested by type specific ELISA (Wellcozyme HIV-1 or HIV-2).
From August 1996, we used the ICEHIV-1.O.2 (Murex) test. If reactive, samples were re-tested using the ICE-HIV-2 test. Samples positive for both HIV sub-types were further tested by Pepti-Lav 1-2 (Sanofi) with a second test completed on a sample taken 2-8 weeks after the initial positive result.
Classic PCR testing of peripheral blood mononuclear cells (PBMC) was also used to confirm dual infections.
CD4 cell counts quantification
CD4 percentage was estimated by FACScan (Becton-Dickinson, Oxford, UK) until August 1997 and by FACS Calibur (Becton-Dickinson) from September 1997 onwards.
Beta-2-microglobulin measurement (β2m)
Plasma levels of β2m were measured using the integrated automated Abbott Architect ci4100 system (Abbott Laboratories, Wiesbaden, Germany) using Quantia β2m reagents under calibrated conditions. Thawed plasma samples that were collected in heparinized tubes were used for a single time point assessment. The results were expressed in mg/L of β2m based on the WHO International Standard [32].
Mortality
Mortality data were collected and updated regularly. The dates of death were extracted from hospital records for patients who died at the MRC hospital or through interviews with relatives and neighbors.
DATA ANALYSIS
The characteristics of the population were described using medians with interquartile ranges (IQR) for continuous variables numbers with corresponding proportions for categorical variables. Comparisons of categorical variables were done using Chi2 test. The continuous variables were compared between two groups by t-test or Mann Whitney/Wilcoxon test and between three groups by ANOVA or Krusskal Wallis test.
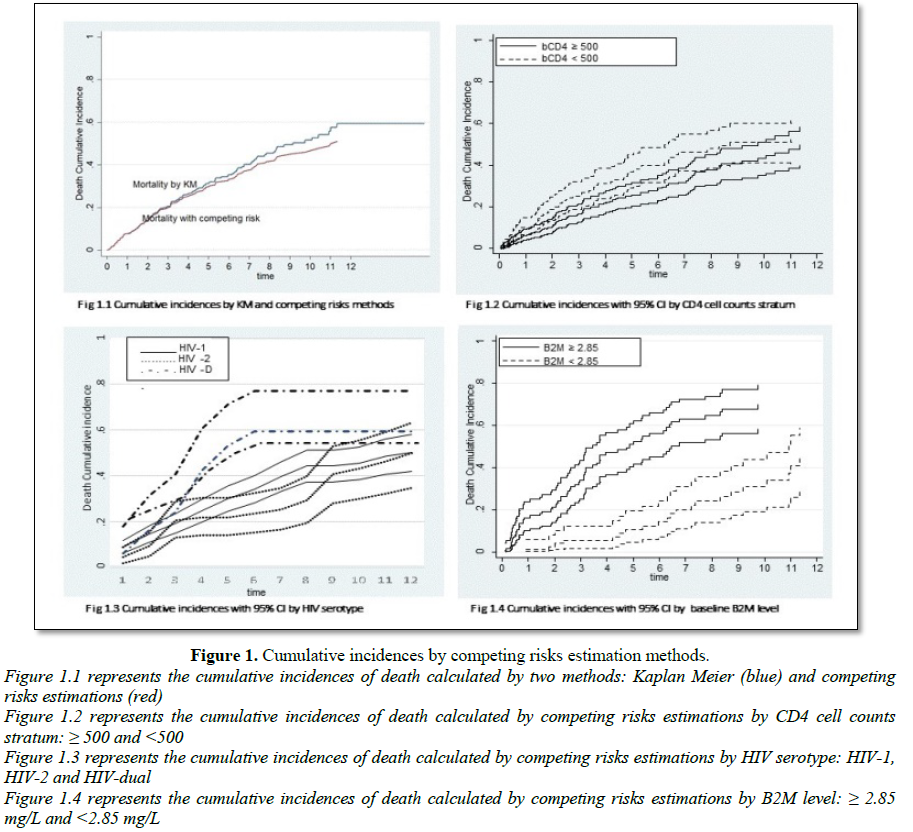
Survival analyses of death were conducted using competing risk estimators. Survival time was the period between the date of entry into the cohort and the date of death or date of ART initiation. For non-deceased individuals, data were censored on the date of last contact. The cumulative incidence of death in this cohort was calculated using competing risk estimators; treating ART initiation as a competing event for death. The comparison of the survival curves between the two strata of CD4 cell counts at baseline (350-499 and ≥ 500), the three categories of HIV serotype (HIV-1, HIV-2 and HIV-1+HIV-2) and the two categories of β2m level (≥ 2.85 mg/L and
To evaluate the association between baseline CD4 cell counts (bCD4) (350-499 versus ≥ 500) and mortality with the presence of competing event, we used competing risks regression models to calculate the corresponding sub-distribution hazard ratios (sHR). We estimated crude and adjusted sHR with 95% confidence intervals (95% CI). Confounding was controlled using a 10% change in estimate method (variables that change the estimate by ≥|10%| were included in the model) among the following potential confounders: age (16-24/25-30/31-38/39-76), sex (male vs. female), education level (absence/elementary/high school/university), occupation (absence/informal/employee/executive), inclusion period (1992-2004/2005-2010), WHO clinical stage (stages 1-2/stages 3-4), HIV serotype (HIV-1/HIV-2/HIV-1+HIV-2) and CD4 cell counts during follow up (fCD4) which was treated as time-dependent variable. Age, sex and inclusion period were tested as effect modifiers. The associations between mortality and HIV serotype (HIV-1/HIV-2/HIV-1+HIV-2) and β2m level (≥ 2.85/<2.85) were assessed using the same methodology and considering the same potential confounders. HIV serotype was also tested as effect modifier for the association between mortality and β2m level.
In order to compare our results, Kaplan Meier method and Cox proportional models (censuring PLHIV at ART initiation) were also used to estimate survival time and compute hazard ratios (HR).
RESULTS
The cumulative incidence was 49.6% (95% CI=40.0%-58.4%) in the stratum “CD4 ≥ 500”) and 53.5% (95% CI=42.8%-62.9%) in the stratum “350-499”) (Figure 1.2). The crude and adjusted sHR of the effect of bCD4 stratum are summarized in Table 2. After adjustment on age, sex and fCD4, bCD4 of 350-499/µL was associated with a 49% increase risk of death compared with baseline CD4 ≥ 500/µL (asHR=1.49; 95% CI=1.07-2.07; p=0.017). This effect was not modified by the following variables: age (p=0.596), sex (p=0.808), inclusion period (p=0.747) (data not shown). Classical Cox regression model showed similar results.
Death cumulative incidence at one year was 8.8% (95% CI=6.3-11.8) among HIV-1 patients and 29.9% (95% CI=24.4-35.7) after five years of follow up. In HIV-2 patients, cumulative incidence at one year and five years were 4.4% (95% CI=1.8-8.9) and 21.6% (95% CI=14.1-30.3), respectively. Cumulative incidences were 6.2% (95% CI=1.1-17.9) and 53.2% (95% CI=30.9-71.2) for HIV-D patients (Figure 1.3). Table 3 presents the univariable and multivariable analysis of the association between HIV serotype and mortality. The sHR were adjusted on age, sex, education level and inclusion period. Comparing HIV-2 infection, HIV-1 and HIV-D infection were associated with a higher risk of death: the respective asHR were 1.66 (95% CI=1.14-2.43) and 2.15 (95% CI=1.25-3.73). The risk of death was not statistically different between HIV-D infected patients and HIV-1 mono infected patients: asHR=1.36 (95% CI=0.84-2.21). Similar results were obtained using classic Cox regression.
The effect of beta 2 microglobulin (β2m) on mortality was evaluated in a subpopulation of 263 individuals. The characteristics of patients included for the analysis of β2m are summarized in Table 1. The β2m level was dichotomized with the median value as the cut-off point: ≥ 2.85 mg/l or Cumulative incidence of death varied with the level of β2m: after ten years, the cumulative incidence of death was 51.0% (95% CI=40.2-60.7) in case of baseline β2m level ≥ 2.85 and 10.6% (95% CI=4.6-19.5) in case of baseline β2m level (Figure 1.4). Crude and adjusted sHR and 95% CI for the association between β2m ≥ 2.85 mg/l and mortality are presented in Table 4. After adjustment on age and sex, Plasma β2m was associated with mortality. PLHIV who presented with baseline β2m ≥ 2.85 mg/l had more than three times higher mortality than people who presented with baseline β2m: asHR=3.26 (95% CI=2.03-5.25). The association was similar for HIV-1 infection (asHR=3.31; 95% CI=1.77-6.20) and HIV-2 infection (asHR=3.22; 95% CI=1.37-7.55) as well as for HR obtained with classic proportional hazard model.
DISCUSSION
We conducted a prospective cohort study on a population of 908 ART-naïve Gambian PLHIV followed between 1992 and 2010 with an average exposure time of 2.5 years. We described pre-ART mortality and estimated the association of bCD4, HIV serotype and β2m level using competing risks estimation methods and classical methods.
After adjustment, mortality was influenced by CD4 stratum, HIV serotype and β2m level.
In this study, we recruited and followed a significant ART-naïve population over two decades. Our observational cohort reflects the real-life conditions in Sub-Saharan Africa over secular time, offering a powerful basis for generalizability across different contexts.
Our mortality rate was similar to those found in Sub-Saharan Africa although some authors reported lower values related to the differences in methods of survival analysis [7-11,20]. However, the exposure times in these studies were relatively short. In developed countries like North American and European countries, mortality among ART naïve PLHIV is lower than what has been found in our cohort: it was lower than 0.7/100 PY in the stratum “350-499” and lower than 0.5/100 PY in the stratum “≥ 500” [1,2]. In EuroSIDA, the mortality rate was 1.4/100 PY in a population with ≥ 350 CD4/µL while in CASCADE, cumulative incidences were 10% and 44% at five years and ten years after seroconversion, respectively [3,6].
After adjustment, our study found a 49% increase in mortality in the stratum of baseline CD4 350-499 compared with the stratum CD4 ≥ 500. A difference in mortality by CD4 cell count stratum was previously reported in one study [16] but not in others [13,18,34-36]. Importantly, these studies used a different methodology, considering the time spent in a specific CD4 cell count stratum instead of baseline CD4 cell count only. These studies have contributed to the debate on the best moment to initiate ART without bringing about a clear consensus. A systematic review including 24 studies with three RCT was in favor of initiating ART at 350-500 CD4/µL [19] and supported the WHO guidelines for ART initiation issued in 2013 [37]. In 2015, the results of two major RCTs confirmed those of HPTN 052 and recommended initiation of ART regardless CD4 cell count and clinical stage [12]. In these RCTs, a composite measure of the primary endpoint was death, AIDS event or serious non-AIDS event. The authors argued there was a clinical benefit from early initiation of ART, although the differences in mortality were not statistically significant [13-15].
One major limitation of RCTs is external validity: this is particularly the case for HPTN 052 which was conducted in a population of serodiscordant couples (SDC). The PLHIV in SDC are known to have an important social support and a high risk perception that might contribute to good adherence and good results [38-40]. More generally, international RCTs are conducted under optimal conditions that do not reflect those found in real-life: additional technical, financial and social supports are given in order to maximize adherence. This may explain in part the low number of events and deaths in these trials, and their inability to show statistically significant differences in mortality. In Sub Saharan Africa, it is usually through RCTs or other clinical research settings that it is possible to obtain frequent measures of CD4 cell count and even viral load among ART-naïve PLHIV. Significant loss to follow up has already been reported in these PLHIV and it is important to take into account this parameter [41-44]. For these reasons, real-life data are necessary to support the results of RCTs: this is crucial to convince decision-makers that results of RCTs can be reproduced in resource-poor settings and even scaled up.
In this analysis, based on real-life data, we showed that ART initiation at CD4 cell count ≥ 500 would reduce mortality compared to initiating at a CD4 cell count of 350-499. We have chosen to consider baseline CD4 cell count as the main exposure variable and to adjust for CD4 cell count measurements during follow up. The first visit and baseline CD4 cell count determine future clinical management of PLHIV in Sub Saharan Africa. Before ART initiation, consecutive visits usually depend on the wellbeing of the patient and are often irregular. Our study offers insight into the natural history of HIV in ART-naïve patients in Sub Saharan Africa under real-world conditions.
Our observation that mortality was higher in HIV-1 and HIV-D infected than in HIV-2 infected patients has previously been reported [20-23]. HIV-2 is less virulent with a slower progression than HIV-1. The underlying mechanisms are not fully understood yet, but some authors linked it to the virus-cell interactions during surface envelope Glycoprotein engagement of cell receptors, the difference in the humoral response or genomic differences [45-48]. We found that mortality of HIV-D patients was higher than that of HIV-1 patients but the difference was not statistically significant as it was the case of the majority of previous studies [23,25,27]. This is in contradiction with the results of Esbjörnsson et al. who reported an inhibition of HIV-1 disease progression by contemporaneous HIV-2 infection and a lower mortality in HIV-D patients [26,28]. It was important to produce valid estimations of mortality by HIV serotype in West Africa where both viruses remain endemic.
Through this study, we also found that the plasmatic level of β2m was strongly associated with death, similarly in HIV-1 and HIV-2 infected patients. The role of immune activation (IA) in HIV disease progression and death was previously reported by studies from different contexts [31,49-55]. However, these studies were cross-sectional, of short duration or mainly concerned PLHIV in advanced disease stages. Two studies conducted in Rwanda and Zambia did not find a role of β2m in mortality of HIV patients [56,57]. Our study confirmed the role of IA in HIV-1 and HIV-2 disease progression and mortality even in PLHIV with high CD4 cell count and followed for 18 years. There is little data examining these relationships in the context of high prevalence of both IA and HIV infection. In addition to HIV disease progression, IA has been recognized as the main risk factor for non-AIDS related morbidity and mortality in PLHIV even under effective ART [29,58-60]. As a result, IA has become a therapeutic target and specific therapies are being developed [29,61]. Our study can contribute to inform the best usage of such therapies in Sub Saharan Africa. Moreover, we used β2m as soluble marker of IA. β2m has already been shown to be suitable marker for a general IA in HIV infection and is stable for long-term storage of plasma samples and freeze-thawing [62]. This low cost and easily quantifiable marker has a public health interest in contrast with cellular markers which are usually preferred in research contexts.
However, our study presents some limitations: potential confounders such as body mass index (BMI), hemoglobin level and viral load were not collected although their roles in mortality were not unanimously recognized [31,33]. The effect of β2m level was evaluated in a subpopulation of the cohort with a possibility of selection bias. However, the selection of this subpopulation preceded the event and was not related to any of our exposure variables, minimizing a possibility of such bias. Moreover, a significant proportion were lost to follow up. In sensitivity analyses, if we considered all those lost to follow up as deceased, the effects of our exposure variables were underestimated (toward the null), while considering them all as alive did not significantly change the associations (supplementary results). It is more likely that the lost to follow-up remained alive for several reasons. Firstly, they were younger, were more frequently working, had a higher level of education and were more frequently infected by HIV-2. Secondly, the procedures of the study described in the methods were very effective to report deaths even if they occurred out of hospital. Thirdly, the existence of alternative therapeutic options (traditional medicine and then ART programs in surrounding countries) was considered the main reason of lost to follow up.
CONCLUSION
In this observational cohort of ART-naïve Gambian PLHIV followed for two decades, we demonstrated that mortality was higher in those with baseline CD4 cell count of 350-499 compared to those with a baseline CD4 ≥ 500. We confirmed that mortality is lower in HIV-2 infection than in HIV-1 and HIV-D infections with no argument of the inhibition of HIV-1 progression by HIV-2 co-infection. Using a soluble marker of IA with potential public health interest, we found a strong and durable association between β2m and mortality. These data are scare in Sub Saharan Africa and provide a picture of real-life long-term differences in mortality. They clearly indicate that early ART initiation and lower levels of IA would reduce mortality in a context of high prevalence of HIV and IA.
ACKNOWLEDGEMENT
The authors acknowledge Pr. Papa Salif Sow, all the members of the WAPHIR, the biologists and the lab technicians of the MRCG, the research assistant of Sainte Justine Research Center (Louise Laporte), the laboratory of Pasteur Institute in Dakar (Dr. Raymond Bercion and Mr. Khadim Dramé).
FUNDING
This work is part of a PhD program (epidemiology) that was funded by the West African Platform for HIV Intervention Research (WAPHIR) and the GHRI/IDRC (Global Health Research Institute/International Development Research Centre, Canada and Medical Research Council, The Gambia (MRCG).
AD benefitted scholarships from the WAPHIR, the Graduate Institute of International and Development Studies and the “Faculté des Etudes Supérieures et Post Doctorales (FESP)” of Université de Montréal.
HT holds a salary award (chercheur-boursier) from the Fonds de la recherche du Québec en santé (FRQ-S) and from CIHR (New investigator salary award).
AUTHORS’ CONTRIBUTIONS
AD, HT, VKN and AJ conceived the work.
AD, GT, MT and AS coordinated data collection and quality control.
AS realized beta-2-microglobulin measurements.
MT realized viral load measurements.
AD and HT realized the analysis and the interpretation of the data.
AD realized the first draft of the manuscript.
HT, VKN, GT, MT, AS, AJ and SM revised the consecutive drafts.
1. Lodwick RK, Sabin CA, Porter K, Ledergerber B, et al. (2010) Death rates in HIV-positive antiretroviral-naive patients with CD4 count greater than 350 cells/µL in Europe and North America: A pooled cohort observational study. Lancet 376: 340-345.
2. Phillips AN, Gazzard B, Gilson R, Easterbrook P, Johnson M, et al. (2007) Rate of AIDS diseases or death in HIV-infected antiretroviral therapy-naïve individuals with high CD4 cell count. AIDS 21: 1717-1721.
3. Mocroft A, Ledergerber B, Katlama C, Kirk O, Reiss P, et al. (2003) Decline in the AIDS and death rates in the EuroSIDA study: An observational study. Lancet 362: 22-29.
4. Mocroft A, Katlama C, Johnson AM, Pradier C, Antunes F, et al. (2000) AIDS across Europe, 1994-1998: The EuroSIDA study. Lancet 356: 291-296.
5. Phillips A, Pezzotti P; CASCADE Collaboration (2004) Short-term risk of AIDS according to current CD4 cell count and viral load in antiretroviral drug-naïve individuals and those treated in the monotherapy era. AIDS 18: 51-58.
6. Smit C, Geskus R, Walker S, Sabin C, Coutinho R, et al. (2006) Effective therapy has altered the spectrum of cause-specific mortality following HIV seroconversion. AIDS 20: 741-749.
7. Badri M, Lawn SD, Wood R (2006) Short-term risk of AIDS or death in people infected with HIV-1 before antiretroviral therapy in South Africa: A longitudinal study. Lancet 368: 1254-1259.
8. Hargrove JW, Humphrey JH; ZVITAMBO Study Group (2010) Mortality among HIV-positive postpartum women with high CD4 cell counts in Zimbabwe. AIDS 24: F11-F14.
9. Masiira B, Baisley K, Mayanja BN, Kazooba P, Maher D, et al. (2014) Mortality and its predictors among antiretroviral therapy naïve HIV-infected individuals with CD4 cell count ≥ 350 cells/mm3 compared to the general population: Data from a population-based prospective HIV cohort in Uganda. Global Health Action 7: 21843.
10. Anglaret X, Minga A, Gabillard D, Ouassa T, Messou E, et al. (2012) AIDS and non-AIDS morbidity and mortality across the spectrum of CD4 cell counts in HIV-infected adults before starting antiretroviral therapy in Cote d'Ivoire. Clin Infect Dis 54: 714-723.
11. Lewden C, Gabillard D, Minga A, Ekouévi DK, Avit D, et al. (2012) CD4-specific mortality rates among HIV-infected adults with high CD4 counts and no antiretroviral treatment in West Africa. J Acquir Immune Defic Syndr 59: 213-219.
12. World Health Organization WHO (2016) Consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection. Recommendations for a public health approach - 2nd Edn.
13. Grinsztejn B, Hosseinipour MC, Ribaudo HJ, Swindells S, Eron J, et al. (2014) Effects of early versus delayed initiation of antiretroviral treatment on clinical outcomes of HIV-1 infection: Results from the phase 3 HPTN 052 randomised controlled trial. Lancet Infect Dis 14: 281-290.
14. INSIGHT START Study Group, Lundgren JD, Babiker AG, Gordin F, Emery S, et al. (2015) Initiation of antiretroviral therapy in early asymptomatic HIV infection. N Engl J Med 373: 795-807.
15. TEMPRANO ANRS 12136 Study Group; Danel C, Moh R, Gabillard D, Badje A, et al. (2015) A trial of early antiretrovirals and isoniazid preventive therapy in Africa. N Engl J Med 373: 808-822.
16. Kitahata MM, Gange SJ, Abraham AG, Merriman B, Saag MS, et al. (2009) Effect of early versus deferred antiretroviral therapy for HIV on survival. N Engl J Med 360: 1815-1826.
17. Guiguet M, Porter K, Phillips A, Costagliola D, Babiker A (2008) Clinical progression rates by CD4 cell category before and after the initiation of combination antiretroviral therapy (cART). Open AIDS J 2: 3-9.
18. HIV-CAUSAL Collaboration, Cain LE, Logan R, Robins JM, Sterne JA, et al (2011) When to initiate combined antiretroviral therapy to reduce mortality and AIDS-defining illness in HIV-infected persons in developed countries: An observational study. Ann Intern Med 154: 509-515.
19. Anglemyer A, Rutherford GW, Easterbrook PJ, Horvath T, Vitória M, et al. (2014) Early initiation of antiretroviral therapy in HIV-infected adults and adolescents: A systematic review. AIDS 2: S105-S118.
20. Hansmann A, Schim van der Loeff MF, Kaye S, Awasana AA, Sarge-Njie R, et al (2005) Baseline plasma viral load and CD4 cell percentage predict survival in HIV-1 and HIV-2-infected women in a community-based cohort in The Gambia. J Acquir Immune Defic Syndr 38: 335-341.
21. Poulsen AG, Aaby P, Larsen O, Jensen H, Nauclér A, et al. (1997) 9 year HIV-2-associated mortality in an urban community in Bissau, west Africa. Lancet 349: 911-914.
22. Berry N, Jaffar S, Schim van der Loeff M, Ariyoshi K, Harding E, et al. (2002) Low level viremia and high CD4% predict normal survival in a cohort of HIV type-2-infected villagers. AIDS Res Hum Retroviruses. 18: 1167-1173.
23. Alabi AS, Jaffar S, Ariyoshi K, Blanchard T, Schim van der Loeff M, et al. (2003) Plasma viral load, CD4 cell percentage, HLA and survival of HIV-1, HIV-2 and dually infected Gambian patients. AIDS 17: 1513-1520.
24. Månsson F, Biague A, da Silva ZJ, Dias F, Nilsson LA, et al. (2009) Prevalence and incidence of HIV-1 and HIV-2 before, during and after a civil war in an occupational cohort in Guinea-Bissau, West Africa. AIDS 23: 1575-1582.
25. Holmgren B, da Silva Z, Vastrup P, Larsen O, Andersson S, et al. (2007) Mortality associated with HIV-1, HIV-2 and HTLV-I single and dual infections in a middle-aged and older population in Guinea-Bissau. Retrovirology 4: 85.
26. Esbjörnsson J, Månsson F, Kvist A, Isberg PE, Nowroozalizadeh S, et al. (2012) Inhibition of HIV-1 disease progression by contemporaneous HIV-2 infection. N Engl J Med 367: 224-232.
27. Prince PD, Matser A, van Tienen C, Whittle HC, Schim van der Loeff MF (2014) Mortality rates in people dually infected with HIV-1/2 and those infected with either HIV-1 or HIV-2: A systematic review and meta-analysis. AIDS 28: 549-558.
28. Esbjörnsson J, Månsson F, Kvist A, Isberg PE, Nowroozalizadeh S, et al. (2014) Effect of HIV-2 infection on HIV-1 disease progression and mortality. AIDS 28: 614-615.
29. Miedema F, Hazenberg MD, Tesselaar K, van Baarle D, de Boer RJ, et al (2013) Immune activation and collateral damage in AIDS pathogenesis. Front Immunol 4: 298.
30. Schim van der Loef MF, Jaffar S, Aveika AA, Sabally S, Corrah T, et al (2002) Mortality of HIV-1, HIV-2 and HIV-1/HIV02 dually infected patients in a clinic-based cohort in The Gambia. AIDS 16: 1775-1783.
31. Nyamweya S, Townend J, Zaman A, Steele SJ, Jeffries D, et al. (2012) Are plasma biomarkers of immune activation predictive of HIV progression: A longitudinal comparison and analyses in HIV-1 and HIV-2 infections? PLoS One. 7: e44411.
32. WHO International Laboratory for Biological Standards (1996) The International Standard for beta 2 microglobulin. WHO, Medicine and Healthcare Products Regalatory Agency.
33. Lawn SD, Harries AD, Anglaret X, Myer L, Wood R (2008) Early mortality among adults accessing antiretroviral treatment programmes in sub-Saharan Africa. AIDS 22: 1897-1908.
34. Writing Committee for the CASCADE Collaboration (2011) Timing of HAART initiation and clinical outcomes in human immunodeficiency virus type 1 seroconverters. Arch Intern Med 171: 1560-1569.
35. When To Start Consortium, Sterne JA, May M, Costagliola D, de Wolf F, et al. (2009) Timing of initiation of antiretroviral therapy in AIDS-free HIV-1-infected patients: A collaborative analysis of 18 HIV cohort studies. Lancet 373: 1352-1363.
36. Palella FJ Jr, Deloria-Knoll M, Chmiel JS, Moorman AC, Wood KC, et al (2003) Survival benefit of initiating antiretroviral therapy in HIV-infected persons in different CD4+ cell strata. Ann Intern Med 138: 620-626.
37. World Health Organization (2013) Consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection. Recommendations for a Public Health Approach.
38. Goldenberg T, Stephenson R (2015) The more support you have the better: Partner support and dyadic HIV care across the continuum for gay and bisexual men. J Acquir Immune Defic Syndr 69: S73-S79.
39. Ware NC, Pisarski EE, Haberer JE, Wyatt MA, Tumwesigye E, et al (2015) Lay social resources for support of adherence to antiretroviral prophylaxis for HIV prevention among serodiscordant couples in sub-Saharan Africa: A qualitative study. AIDS Behav 19: 811-820.
40. Sidibe S, Pack AP, Tolley EE, Ryan E, Mackenzie C, et al. (2014) Communicating about microbicides with women in mind: Tailoring messages for specific audiences. J Int AIDS Soc 17: 19151.
41. Togun T, Peterson I, Jaffar S, Oko F, Okomo U, et al. (2011) Pre-treatment mortality and loss-to-follow-up in HIV-1, HIV-2 and HIV-1/HIV-2 dually infected patients eligible for antiretroviral therapy in The Gambia, West Africa. AIDS Res Ther 8: 24.
42. Bassett IV, Wang B, Chetty S, Mazibuko M, Bearnot B, et al. (2009) Loss to care and death before antiretroviral therapy in Durban, South Africa. J Acquir Immune Defic Syndr 51: 135-139.
43. McGrath N, Glynn JR, Saul J, Kranzer K, Jahn A, et al. (2010) What happens to ART-eligible patients who do not start ART? Dropout between screening and ART initiation: A cohort study in Karonga, Malawi. BMC Public Health 10: 601.
44. Amuron B, Namara G, Birungi J, Nabiryo C, Levin J, et al. (2009) Mortality and loss-to-follow-up during the pre-treatment period in an antiretroviral therapy programme under normal health service conditions in Uganda. BMC Public Health 9: 290.
45. Azevedo-Pereira JM, Santos-Costa Q (2016) HIV interaction with human host: HIV-2 as a model of a less virulent infection. AIDS Rev 18: 44-53.
46. Makvandi-Nejad S, Rowland-Jones S (2015) How does the humoral response to HIV-2 infection differ from HIV-1 and can this explain the distinct natural history of infection with these two human retroviruses? Immunol Lett 163: 69-75.
47. Devadas K, Biswas S, Haleyurgirisetty M, Ragupathy V, Wang X, et al. (2016) Identification of host micro RNAs that differentiate HIV-1 and HIV-2 infection using genome expression profiling techniques. Viruses 8: E121.
48. Yamaguchi J, Brennan CA, Alessandri-Gradt E, Plantier JC, Cloherty GA, et al. (2017) HIV-2 surveillance with next-generation sequencing reveals mutations in a cytotoxic lymphocyte-restricted epitope involved in long-term non-progression. AIDS Res Hum Retroviruses 33: 347-352.
49. Sousa AE, Carneiro J, Meier-Schellersheim M, Grossman Z, Victorino RM (2002) CD4 T cell depletion is linked directly to immune activation in the pathogenesis of HIV-1 and HIV-2 but only indirectly to the viral load. J Immunol 169: 3400-3406.
50. Fahey JL, Taylor JM, Detels R, Hofmann B, Melmed R, et al (1990) The prognostic value of cellular and serologic markers in infection with human immunodeficiency virus type 1. N Engl J Med 322: 166-172.
51. Ullum H, Lepri AC, Katzenstein TL, Phillips AN, Skinhøj P, et al. (2000) Prognostic value of single measurements of beta-2-microglobulin, immunoglobulin A in HIV disease after controlling for CD4 lymphocyte counts and plasma HIV RNA levels. Scand J Infect Dis 32: 371-376.
52. Liu Z, Cumberland WG, Hultin LE, Prince HE, Detels R, et al. (1997) Elevated CD38 antigen expression on CD8+ T cells is a stronger marker for the risk of chronic HIV disease progression to AIDS and death in the multicenter AIDS Cohort Study than CD4+ cell count, soluble immune activation markers, or combinations of HLA-DR and CD38 expression. J Acquir Immune Defic Syndr Hum Retrovirol 16: 83-92.
53. Ostrowski SR, Gerstoft J, Pedersen BK, Ullum H (2003) A low level of CD4+ CD28+ T cells is an independent predictor of high mortality in human immunodeficiency virus type 1-infected patients. J Infect Dis 187: 1726-1734.
54. Michel P, Balde AT, Roussilhon C, Aribot G, Sarthou JL, et al. (2000) Reduced immune activation and T cell apoptosis in human immunodeficiency virus type 2 compared with type 1: Correlation of T cell apoptosis with beta2 microglobulin concentration and disease evolution. J Infect Dis 181: 64-75.
55. Jaffar S, Van der Loeff MS, Eugen-Olsen J, Vincent T, Sarje-Njie R, et al. (2005) Immunological predictors of survival in HIV type 2-infected rural villagers in Guinea-Bissau. AIDS Res Hum Retroviruses. 21: 560-564.
56. Kerlikowske KM, Katz MH, Allen S, Wolf W, Hudes ES, et al. (1994) Beta 2-microglobulin as a predictor of death in HIV-infected women from Kigali, Rwanda. AIDS 8: 963-969.
57. Hosp M, Lisse IM, Quigley M, Mwinga AM, Godfrey-Faussett P, et al. (2000) An evaluation of low-cost progression markers in HIV-1 seropositive Zambians. HIV Med 1: 125-127.
58. McComsey GA, Kitch D, Sax PE, Tierney C, Jahed NC, et al. (2014) Associations of inflammatory markers with AIDS and non-AIDS clinical events after initiation of antiretroviral therapy: AIDS clinical trials group A5224s, a sub-study of ACTG A5202. J Acquir Immune Defic Syndr 65: 167-174.
59. Brenchley JM, Price DA, Schacker TW, Asher TE, Silvestri G, et al. (2006) Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat Med 12: 1365-1371.
60. Kuller LH, Tracy R, Belloso W, De Wit S, Drummond F, et al. (2008) Inflammatory and coagulation biomarkers and mortality in patients with HIV infection. PLoS Med 5: e203.
61. Hsu DC, Sereti I (2016) Serious non-AIDS events: Therapeutic targets of immune activation and chronic inflammation in HIV infection. Drugs 76: 533-549.
62. Aziz N, Nishanian P, Taylor JM, Mitsuyasu RT, Jacobson JM, et al. (1999) Stability of plasma levels of cytokines and soluble activation markers in patients with human immunodeficiency virus infection. J Infect Dis 179: 843-848.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Clinical Trials and Research (ISSN:2637-7373)
- Journal of Forensic Research and Criminal Investigation (ISSN: 2640-0846)
- International Journal of Anaesthesia and Research (ISSN:2641-399X)
- Stem Cell Research and Therapeutics (ISSN:2474-4646)
- Journal of Spine Diseases
- Oncology Clinics and Research (ISSN: 2643-055X)
- Journal of Immunology Research and Therapy (ISSN:2472-727X)