Research Article
Bundle Designed Psychosocial Interventions to Improve the Mental Health of HIV-Infected Patients during Hospitalization
2825
Views & Citations1825
Likes & Shares
Background: Depression and anxiety are the most common comorbidities among people living with HIV/AIDS (PLWHA). These mental health complications are associated with suboptimal outcomes including high mortality. Evidence has suggested that psychological interventions could improve mental health, quality of life, and HIV care outcomes. This study aimed to evaluate the effectiveness of bundle-designed psychosocial interventions in the reduction of anxiety and depression in PLWHA.
Methods: This retrospective cohort study tested the effectiveness of a bundle-designed psychosocial intervention package (HIV-STAR) mainly including adherence, psychology, social support and individual case management. Anxiety and depression evaluation was performed and compared using the Hospital Anxiety and Depression Scale (HADS) at the hospital admission and discharge. The McNemar test was used to assess changes of HADS before and after HIV-STAR intervention. Logistic regression was performed to identify risk factors.
Results: Among the 97 PLWHA enrolled after screening, 36% were positive for anxiety and 30% for depression. The overall incidence of anxiety and/or depression was significantly decreased from 46% at admission to 23% at discharge after HIV-STAR with an average intervention duration of 14 days, (p<0.001). The mean score of HADS was 11.2 (SD 7.4) at the admission and 8.1 (SD 6.2) at the discharge (p<0.001). In multivariate analysis, female sex (OR=23.64; 95% CI, 1.43-392.26), current recreational drug use (OR=3.07; 95% CI, 1.16-8.13), and risk group for HIV infection other than MSM (OR=3.32; 95% CI, 1.02-10.86) were significantly associated with high HADS (≥8.0) at the discharge.
Conclusions: The high prevalence of mental health complications among PLWHA in Taiwan underscores the importance of integrated psychosocial care. While the newly developed HIV-STAR has provided an effective intervention to reduce anxiety and depression in general, more advanced psychosocial care bundles will be warranted to address specific risk factors for intervention-resistant anxiety and depression.
Keywords: Bundle care, Psychosocial intervention, Depression and anxiety, HIV
INTRODUCTION
Although antiretroviral therapy (ART) has transformed HIV disease into a manageable, chronic medical condition, HIV infection affects all dimensions of wellness, including physical, mental and spiritual well-being. Depression and anxiety are the most common comorbidities among people living with HIV/AIDS (PLWHA) [1,2]. The prevalence of depression and anxiety ranged from 15-80% and 9-40% in PLWHA, respectively [3-6]. HIV-associated neurocognitive disorders, side effects from ART (e.g., efavirenz), and psychosocial factors (e.g., awareness of chronic disease,
functional disability, isolation, lack of support, discrimination, stigma, and substance use disorders) may all contribute to such a high prevalence of anxiety and depression [1]. These mental health complications are associated suboptimal outcomes, including increased health-care utilization, decreased quality of life, and poor adherence to ART [7,8]. Furthermore, greater depression burden increased mortality rate up to 2-fold in this population [8].
ART has been made available to all HIV-infected patients, approximately 40,000 in Taiwan, through the National Health Insurance (NHI) program since 1997. According to the previous studies among 769 participants living with HIV in Taiwan, 20% became depressed after the diagnosis and the prevalence of depression significantly increased from 2000 to 2011 [9]. The other cohort study indicated that 24% had psychiatric disorders in PLWHA, among which 77% were anxiety disorder and 8% were major depressive disorder [10].
According to the World Health Organization (WHO) recommendations, psychosocial support can assist PLWHA in making informed decisions, coping better with illness and dealing more effectively with discrimination. Evidence has suggested that psychological interventions could improve mental health, quality of life, and HIV care engagement [11]. These interventions included general psychological (e.g., cognitive-behavior therapy, cognitive behavior stress management, and peer support/ counseling group therapy), psychotropic (e.g., use of trazodone), physical (e.g., massage therapy and exercise training), psychosocial (e.g., case management community care, social support group, and community health workers), and HIV-specific health psychology interventions (e.g., HIV information support, self-care symptom management intervention, enhanced risk prevention intervention, and medication reminder device) [12-15].
Care bundles have recently emerged as a new healthcare entity containing three to five elements with a strong clinician agreement, which will be delivered collectively and consistently with the aim of improving patient outcomes, particularly those difficult to achieve through an individual care module [16]. Besides using multidisciplinary care team to improve the quality of care, the Institute for Healthcare Improvement recommended that compliance with bundles be measured using all-or-none measurement, with a goal of 95 percent or greater. Care bundles have been used across healthcare settings, mainly in the infection control to reduce the incidence of central line-associated bloodstream infections, ventilator-associated pneumonia, catheter-related urinary tract infection and surgical site infection [17].
Evidence of psychosocial interventions for PLWHA remains sparse in the Asian populations. Furthermore, to our knowledge, bundle-designed psychosocial interventions have never been tested in PLWHA. In this study, we developed such a psychosocial intervention bundle by combining several psychological, physical and social components and evaluated its effectiveness in the reduction of anxiety and depression in PLWHA in Taiwan.
METHODS
Study design and sample size
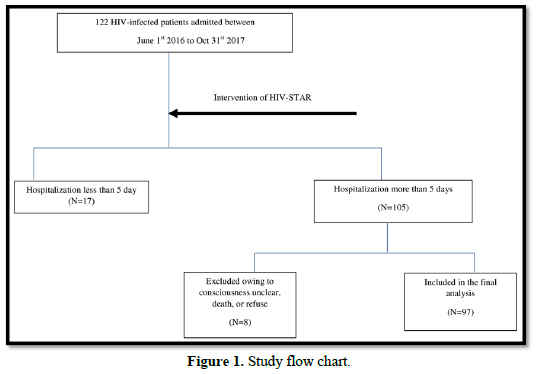
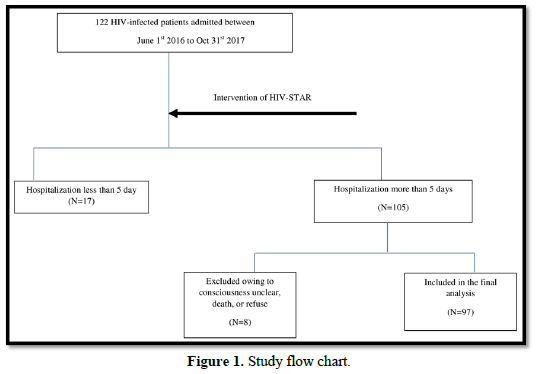
This was a retrospective cohort study. We used a pre- and post-comparison design with the same individuals being followed up over time. Participants included all HIV-infected patients admitted to Taichung Veterans General Hospital, which was a 1500-bed medical center in central Taiwan. A total of 122 HIV-infected patients were admitted during July 1, 2016 to October 31, 2017. Depression and Anxiety score was performed within three days after admission and re-evaluated before discharge. Psychosocial interventions were given during hospitalization. HIV-infected patients with hospitalization less than 5 days were not included due to the difficulty of completing psychosocial intervention in such short time. 105 patients were admitted more than 5 days. After excluding those patients with unclear consciousness, death or refusing consent, a total 97 HIV-infected patients were included in the final analysis (Figure 1). The Ethics Committee of Taichung Veterans General Hospital approved this study.
Description of the intervention
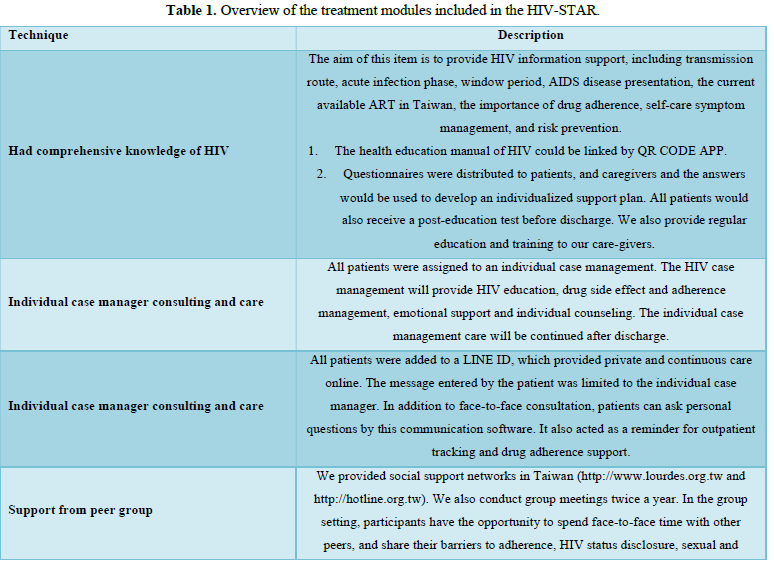
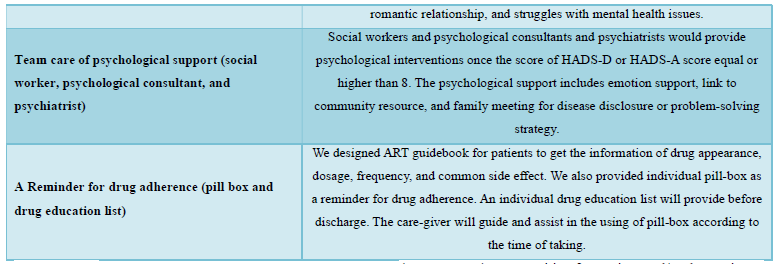
We designed a psychosocial intervention package that was delivered by multidisciplinary care team for all HIV-infected patients during hospitalization. All the staffs in HIV-STAR team included HIV-specific physicians, psychiatrists, HIV care managements, care nurses, social workers, and pharmacists. This psychosocial intervention package (HIV-STAR) included six elements: Had comprehensive knowledge of HIV, Individual case manager consulting and care, Via-Line@ (privacy and continuous care), Support from peer group, Team care of psychological support (social worker, psychological consultant, and psychiatrist), A-Reminder for drug adherence (pill box and drug education list). The detail description of HIV-STAR is showed in Table 1. HIV-STAR was a bundle-designed intervention. Each patient had a checklist to ensure adherence to each element >95%.
Mental health measurements
Anxiety and depression evaluation was performed using the Hospital Anxiety and Depression Scale (HADS) [18]. This score consisted of seven self-report instrument items each relating to anxiety (HADS-A) and depression (HADS-D), respectively. All items were answered on an ordinal response scale with four response categories (0-3). A score of equal or higher than 8 (≥8) on the HADS-A or HADS-D subscales was considered a positive screen for symptoms of anxiety or depression.



Data analysis
Differences between individuals with or without anxiety and/or depression at baseline were evaluated using chi-square tests for categorical variables and Wilcoxon rank-sum tests for continuous variables. In cases of small sample sizes, Fisher’s exact test was performed. We used the McNemar test at the bivariate level to assess changes of HADS score over time. Univariable and multivariable logistic regression analyses were used to identify risk factors for anxiety and depression at admission and at discharge. Variables included in these analyses were age, gender, HIV risk group, current employed, comorbidity, AIDS status at admission, and recreational drug use history. Results are presented as odds ratios (ORs) with 95% confidence intervals (CIs). Statistical analyses were performed using SAS software (Version 9.3).
RESULTS
Patient characteristics
Among 122 patients screened, a total of 97 HIV-infected patients were included in the final analysis and nearly half (n=45, 46.4%) were positive for anxiety and/or depression at the time of admission. Mental health evaluation revealed 36.1% (n=35) positive for anxiety and 29.9% (n=29) positive for depression. Demographic and clinical characteristics of the two groups (with anxiety and/or depression, n=45; without anxiety and/or depression, n=52) were summarized in Table 2. The mean age of the two groups was 35.8 years versus 34.0 years, with the majority being male (100.0% vs. 95.6%, p=0.213) in both groups. No significant differences were noted in terms of education level, married status, financial dependent, family disclosure, or recreational drug and smoking history. Men who have sex with men (MSM) was the major risk factor, and predominant in patients without anxiety or depression (94.2% vs. 68.9%, p=0.002). Patients with anxiety and/or depression were more likely to be without current employment (13.5% vs. 37.8%, p=0.011), alcohol consumption (1.9% vs. 17.8%, p=0.011) and underline comorbidities (13.5% vs. 40.0%, p=0.006) (Table 1). The mean hospitalization days was apparently longer for patients with mental health problems (14.4 vs. 19.0, p=0.201).
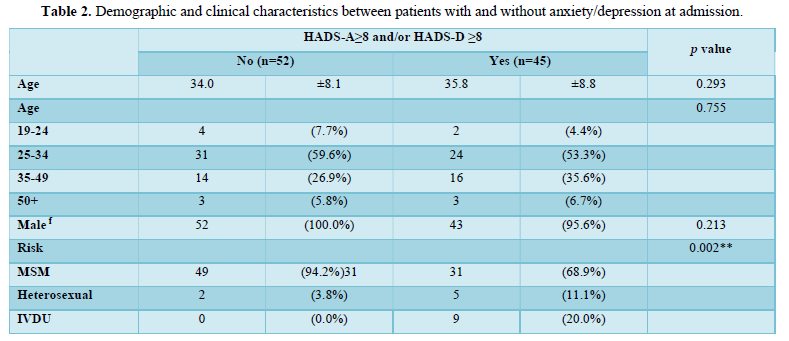
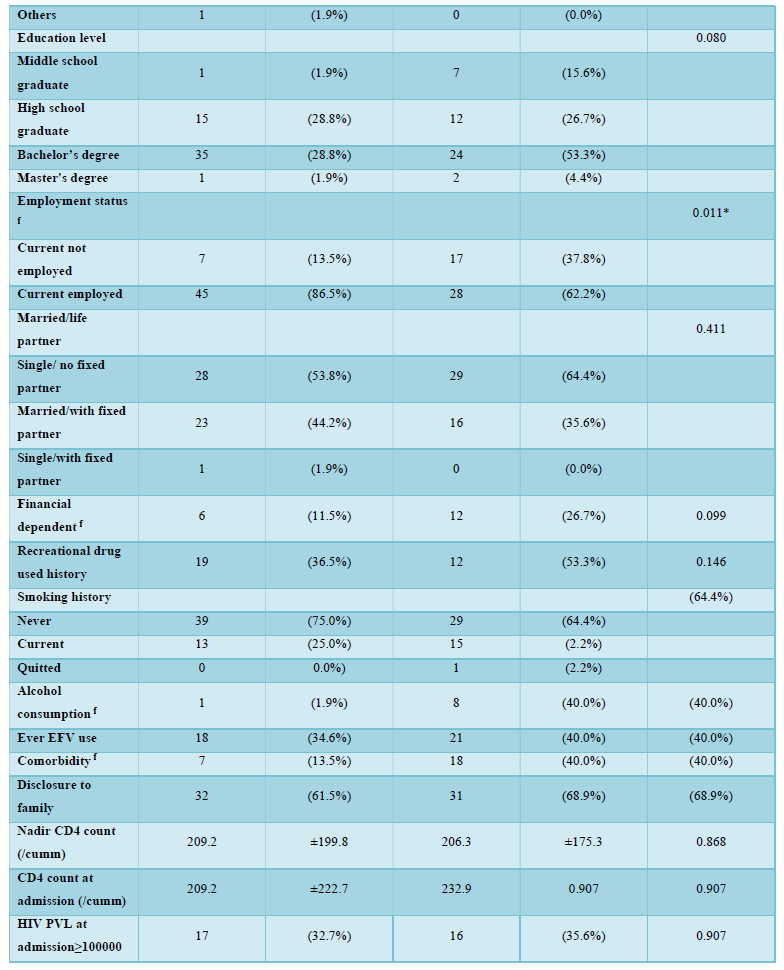
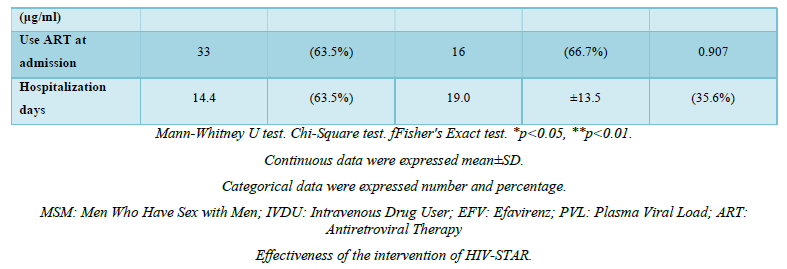



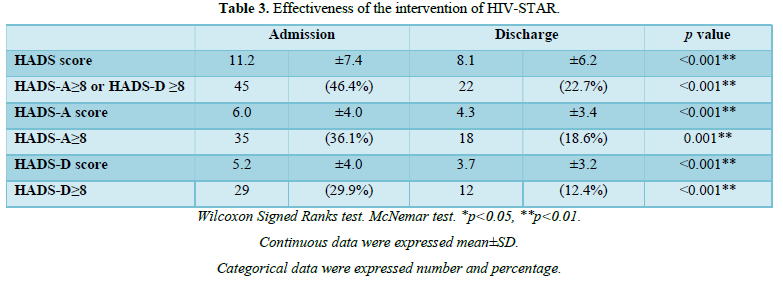
The effectiveness of HIV-STAR was evaluated and summarized in Table 3 and Figure 2. HIV-STAR intervention resulted a significant reduction in mean HADS scores from 11.2 (SD 7.4) at the admission to 8.1 (6.2) at the discharge (p<0.001). Specifically, the mean score of anxiety was reduced from 6.0 (4.0) at the admission to 4.3 (3.4) at the discharge (p<0.001). The mean score of depression decreased from 5.2 (4.0) at the admission to 3.7 (3.2) at the discharge (p<0.001). The high prevalence of anxiety and/or depression at the admission (46.4%) was significantly reduced to 22.7% at the discharge after HIV-STAR intervention (p<0.001), with an improvement rate of 51.1%. The bundle care intervention of HIV-STAR significantly improved the mental health of HIV-infected patients during hospitalization.
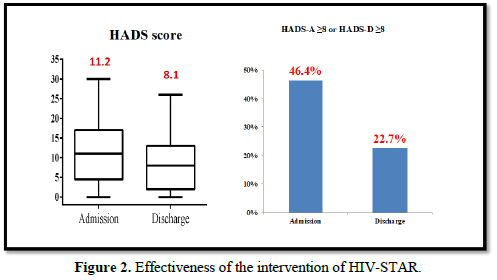


Multivariable analysis of risk factors for either anxiety or depression
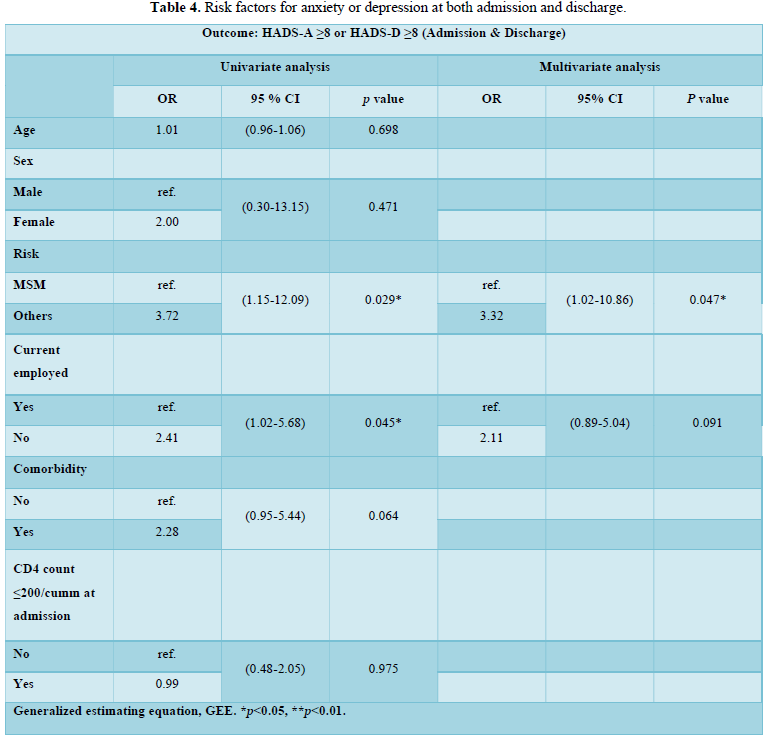
The risk factors for either anxiety or depression at the admission and the discharge were summarized in Table 4. In the univariate analysis, risk group for HIV infection other than MSM (OR=3.72; 95% CI, 1.15-12.09) and current unemployed (OR=2.41; 95% CI, 1.02-5.68) were significantly associated with anxiety or depression as indicated by HADS score ≥ 8. In multivariate analysis, only risk group for HIV infection other than MSM (OR=3.32; 95% CI, 1.02-10.86) was significantly associated with anxiety or depression.

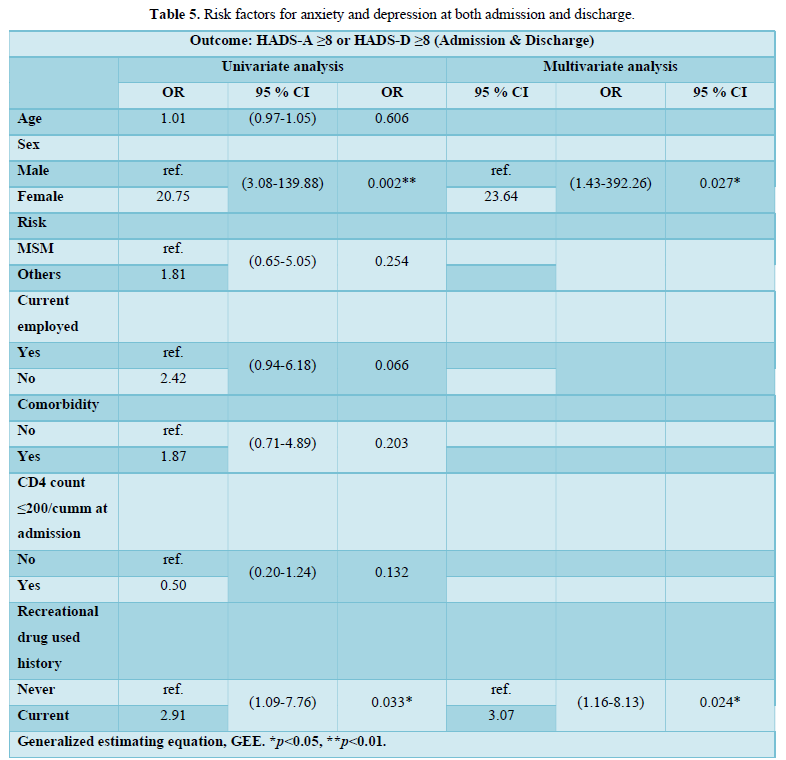
The risk factors for a small portion of patients with both anxiety and depression at the admission (n=19) and the discharge (n=8) were summarized in Table 5. In univariate analysis, female sex (OR=20.75; 95% CI, 3.08–139.88) and current recreational drug use (OR=2.91; 95% CI, 1.09-7.76) were significantly associated with concurrent anxiety and depression (HADS score ≥ 8). In multivariate analysis, female sex (odds ratio, OR=23.64; 95% CI, 1.43–392.26) and current recreational drug use (OR=3.07; 95% CI, 1.16-8.13) remained significantly associated with concurrent anxiety and depression.

DISCUSSION
In this retrospective cohort study, we tested a newly developed bundle-care intervention (HIV-STAR) to improve psychosocial well-being for HIV-infection patients during hospitalization, which was implemented by a multidisciplinary care team. We noticed that after the intervention, the prevalence of anxiety and/or depression symptoms markedly decreased from 46.4% to 22.7%, suggesting a significant short-term improvement in the mental health as a result of HIV-STAR. The risk factors for persistent mental health issues after psychosocial intervention were identified including risk group for HIV infection other than MSM, female sex, and current recreational drug use.
Our study confirmed the high prevalence of mental health complications in PLWHA. Nearly half of the study cohort (46.4%) exhibited symptoms of anxiety and/or depression at the admission, among which 36.1% were positive for anxiety and 29.9% were positive for depression. Our findings were essentially consistent with others based on HADS, such as 34.4/32.0% in Ethiopia [19], 42/36% in China [20], 33.3/15.5% in Western Europe and Canada [4], 30.6/25.4% in South Africa [21], and 60.3/38.7% in London, England [22]. The prevalence of anxiety/depression might vary depending upon sex, age, AIDS status, and current ART use. In our study, ~40% patients were admitted due to opportunistic infections, which might contribute to the high prevalence of anxiety and/or depression. In a long-term follow-up study in Thailand, the prevalence of severe anxiety/depression (HADS >11) decreased to 4.8/3.1% after a mean 7.7-year ART, suggesting the importance of early psychosocial intervention for mental health complications in addition to ART [23]. Thus, an early evaluation of mental health should be recommended for PLWHA especially among those newly diagnosed and presented with AIDS, i.e., opportunistic infections.
HIV-STAR was a bundle-care psychosocial intervention developed and tested in our study, to our knowledge, the first one for Asian PLWHA. A bundle is a structured way of improving the processes of care and patient outcomes. Care bundles were used widely to decrease the incidence of nosocomial infection and improve the quality of patient care [17]. We collected the questionnaires from patients and caregivers first to identify the most frequently faced problems about patient care and formulated problem-solving strategies. We adapted the psychosocial interventions, including CBT [24-27], adherence support and HIV/AIDS symptoms management [28,29], peer support intervention [30], and telephone technology [31]. We also modified the interventions according to our own available resources. In order to ensure a continuous and consistent care, we assembled a multidisciplinary team including nurses, infectious disease specialists, psychiatrists, case managers, and social workers to implement HIV-STAR. Every caregiver received training and a regular education session was provided to each patient. HIV-STAR has become a routine care plan in our HIV Clinic to improve mental health in HIV-infected patients during hospitalization and part of the key elements in continuous care during outpatient follow-up.
HIV-STAR demonstrated its high effectiveness in this study that the severity based on the mean score of HADS significantly decreased from 11.2 at the admission to 8.1 at the discharge after HIV-STAR intervention and more than 50% patients reported an improvement in the symptoms of anxiety and/or depression. Previous studies also reported the benefits of psychosocial interventions in PLWHA and various types of intervention were tested including symptoms-orientated intervention (e.g., cognitive and/or behavior therapy, stress management, and interpersonal therapy), supportive intervention (e.g., peer support, and psycho-education), and meditation intervention (e.g., mindfulness, meditation, and relaxation) [32]. Symptoms-orientated intervention was tested mostly using cognitive behavior therapy as the major vehicle, which significantly decreased symptoms of depression and/or anxiety in PLWHA [24-27]. In the study by Simona et al., cognitive–behavioral stress management significantly reduced both HADS-A and HADS-D from the baseline [24]. Other techniques of symptoms-orientated intervention, such as stress management [31,33] and interpersonal therapy [34] also improved the depression symptoms. Supportive intervention, including adherence support [28], peer support [30,35], and HIV/AIDS symptom management [29], was effective in depression management. Thus, while designing the bundle-care HIV-STAR, we included both symptoms-orientated and supportive intervention and our findings not only supported the benefits of psychosocial intervention, but also suggested high effectiveness of the combination care for improving mental health in PLWHA.
Factors associated with persistent anxiety and/or depression even after psychosocial intervention in our study was HIV infection other than MSM, female sex, and current recreational drug use. Previous studies revealed that female sex, older age, teenagers, pre-infection history of major depression, and HIV disease stage contributed to higher risk of major depression [36]. Among PLWHA, a higher rate of anxiety or depression was reported in women compared to men [4,23]. Women also suffered from higher psychology and global symptom distress than that in men [37]. HIV-related stigma and the experience of adulthood violence contributed to depression in women [38]. In Taiwan, the major risk factor for HIV infection in women was intravenous drug use. Compared to those without substance use disorders, women with intravenous drug use experienced more psychosocial distress and poor quality of life [39]. Although only a small portion of women included in our study, women in Taiwan living with HIV did suffer more from life distress, stigma, and stress from child rising, therefore, continuous psychosocial support and intervention should be recommended to this vulnerable population.
The relationship between HIV transmission and depression remains inconclusive. A meta-analysis study showed the risk of major depression disorder was similar in both homosexual and bisexual men in PLWHA [40]. A recent cohort study in Canada has indicated that compared to heterosexual group, gay, bisexual and MSM had higher incidence of mood disorder [41], which appeared to be contradicted to the higher prevalence of anxiety and/or depression (3.3 times higher) in non-homosexual group observed in our study. One possible explanation was the differences in social support. Unlike homosexual patients who had a supportive system in Taiwan and typically could discuss their disease to their peers, most heterosexual ones chose not to disclose their HIV status to their family. Furthermore, most non-homosexual ones with intravenous drug use faced substantial financial burden and were abandoned by their family. These life distress and lack of support, therefore, might explain the higher prevalence of mental health complications among patients with HIV infection other than MSM.
Our study also indicated that current recreational drug users (n=43, 43.9%) had 3 times higher risk of persistent anxiety and/or depression even after the intervention of HIV-STAR compared to the non-users. Recreational drug use was prevalent among PLWHA [42], and illicit drug use and drug dependence were associated with psychiatric disorders [5]. In Taiwan, substance use disorders were associated with a 2.7-fold increase in the prevalence of depression in PLWHA and the use of club drugs has become a major problem nowadays [9]. Since both substance use disorders and mental health complications such as depression result in poor adherence and worsen the outcomes of HIV management [43], our findings not only confirmed the long-standing challenges in HIV care including mental health and substance use disorders, but also reinforced importance and necessity of a bundle-care module to address the challenges that should be incorporated into current HIV care.
Certain limitations were identified in our study. First, the sample size was relatively small and we only included HIV-infected patients from the inpatients setting, 40% of whom were admitted due to opportunity infections. However, the sample size was similar to that of the Boston Depression Care study, and larger than that of the CBP study in Hong Kong, both were well accepted for their pioneer work on psychosocial interventions in PLWHA. Despite the potential selection bias, the opportunistic comorbidities appeared to aggravate anxiety and depression that rendered mental health care rather critical for this population. It was possible that the effectiveness of HIV-STAR for mental health improvement might reflect a better management of opportunistic infection and reinforce the importance of bundle-care. Another limitation was that the follow-up assessment was only performed before the discharge and only short-term effectiveness of HIV-STAR could be assessed. To address the long-term benefits of this bundle-care, we have also introduced some elements of HIV-STAR into a continuous care module that will be used for follow-up at the outpatient setting.
CONCLUSION
In conclusion, the high prevalence of mental health complications among PLWHA in Taiwan underscores the importance of integrated psychosocial care. While the HIV-STAR has provided an effective intervention to reduce anxiety and depression in general, more advanced psychosocial care bundles will be warranted to address specific risk factors for intervention-resistant anxiety and depression. Future clinical prospective studies are warranted to further address the benefit of integrated psychosocial care, especially in female sex, current recreational drug use, and risk group for HIV infection other than MSM.
ACKNOWLEDGMENTS
The authors wound like to thank for all patients participating in this study and the staffs in HIV care. This study would not have been possible without HIV-STAR care team, including HIV physicians, psychiatrists, HIV care managements, nurses, social workers, and pharmacists in Taichung Veterans General hospital. Qing Ma is supported in part by National Institute on Aging R01AG063659.
AUTHOR DISCLOSURE STATEMENT
No competing financial interests exist.
- Nanni MG, Caruso R, Mitchell AJ, Meggiolaro E, Grassi L (2015) Depression in HIV infected patients: A review. Curr Psychiatry Rep 17(1): 530.
- Brandt C, Zvolensky MJ, Woods SP, Gonzalez A, Safren SA, et al. (2017) Anxiety symptoms and disorders among adults living with HIV and AIDS: A critical review and integrative synthesis of the empirical literature. Clin Psychol Rev 51: 164-184.
- Rane MS, Hong T, Govere S, Thulare H, Moosa MY, et al. (2018) Depression and anxiety as risk factors for delayed care-seeking behavior in HIV positive individuals in South Africa. Clin Infect Dis 67(9): 1411-1418.
- Robertson K, Bayon C, Molina JM, McNamara P, Resch C, et al. (2014) Screening for neurocognitive impairment, depression, and anxiety in HIV-infected patients in Western Europe and Canada. AIDS Care 26(12): 1555-1561.
- Bing EG, Burnam MA, Longshore D, Fleishman JA, Sherbourne CD, et al. (2001) Psychiatric disorders and drug use among human immunodeficiency virus-infected adults in the United States. Arch Gen Psychiatry 58(8): 721-728.
- Catalan J, Harding R, Sibley E, Clucas C, Croome R, et al. (2011) HIV and depression--a systematic review of interventions. Psychol Health Med 16(5): 588-611.
- Deshmukh NN, Borkar AM, Deshmukh JS (2017) Depression and its associated factors among people living with HIV/AIDS: Can it affect their quality of life? J Family Med Prim Care 6(3): 549-553.
- Pence BW, Mills JC, Bengtson AM, Gaynes BN, Breger TL, et al. (2018) Association of Increased Chronicity of Depression with HIV Appointment Attendance, Treatment Failure and Mortality Among HIV-Infected Adults in the United States. JAMA Psychiatry 75(4): 379-385.
- Lu HF, Hsiao FH, Sheng WH, Liao SC, Wu PY, et al. (2018) Prevalence and Predictors of Depression Among People Living With HIV/AIDS: A National Study. Nurs Res 67(5): 379-386.
- Chen MH, Su TP, Chen TJ, Cheng JY, Wei HT, et al. (2012) Identification of psychiatric disorders among human immunodeficiency virus-infected individuals in Taiwan, a nine-year nationwide population-based study. AIDS Care 24(12): 1543-1549.
- Sikkema KJ, Dennis AC, Watt MH, Choi KW, Yemeke TT, et al. (2015) Improving mental health among people living with HIV: A review of intervention trials in low- and middle-income countries. Glob Ment Health (Camb) 2: e19.
- Clucas C, Sibley E, Harding R, Liu L, Catalan J, et al. (2011) A systematic review of interventions for anxiety in people with HIV. Psychol Health Med 16(5): 528-547.
- Harding R, Liu L, Catalan J, Sherr L (2011) What is the evidence for effectiveness of interventions to enhance coping among people living with HIV disease? A systematic review. Psychol Health Med 16(5): 564-587.
- Han HR, Kim K, Murphy J, Cudjoe J, Wilson P, et al. (2018) Community health worker interventions to promote psychosocial outcomes among people living with HIV-A systematic review. PLoS One 13(4): e0194928.
- Wu L, Li X (2013) Community-based HIV/AIDS interventions to promote psychosocial well-being among people living with HIV/AIDS: a literature review. Health Psychol Behav Med 1(1): 31-46.
- Resar R, Griffin FA, Haraden C, Nolan TW (2012) Using care bundles to improve health care quality. IHI innovation series white paper. Institute for Healthcare Improvement.
- Lavallee JF, Gray TA, Dumville J, Russell W, Cullum N (2017) The effects of care bundles on patient outcomes: a systematic review and meta-analysis. Implement Sci 12(1): 142.
- Zigmond AS, Snaith RP (1983) The hospital anxiety and depression scale. Acta Psychiatry Scand 67(6): 361-370.
- Duko B, Toma A, Asnake S, Abraham Y (2019) Depression, Anxiety and Their Correlates Among Patients with HIV in South Ethiopia: An Institution-Based Cross-Sectional Study. Front Psychiatry 10: 290.
- Tao J, Vermund SH, Lu H, Ruan Y, Shepherd BE, et al. (2017) Impact of Depression and Anxiety on Initiation of Antiretroviral Therapy Among Men Who Have Sex with Men with Newly Diagnosed HIV Infections in China. AIDS Patient Care STDS 31(2): 96-104.
- Pappin M, Wouters E, Booysen FL (2012) Anxiety and depression amongst patients enrolled in a public sector antiretroviral treatment programme in South Africa: a cross-sectional study. BMC Public Health 12: 244.
- Lambert S, Keegan A, Petrak J (2005) Sex and relationships for HIV positive women since HAART: A quantitative study. Sex Transm Infect 81(4): 333-337.
- Prasithsirikul W, Chongthawonsatid S, Ohata PJ, Keadpudsa S, Klinbuayaem V, et al. (2017) Depression and anxiety were low amongst virally suppressed, long-term treated HIV-infected individuals enrolled in a public sector antiretroviral program in Thailand. AIDS Care 29(3): 299-305.
- Berger S, Schad T, von Wyl V, Ehlert U, Zellweger C, et al. (2008) Effects of cognitive behavioral stress management on HIV-1 RNA, CD4 cell counts and psychosocial parameters of HIV-infected persons. AIDS 22(6): 767-775.
- Carrico AW, Antoni MH, Duran RE, Ironson G, Penedo F, et al. (2006) Reductions in depressed mood and denial coping during cognitive behavioral stress management with HIV-Positive gay men treated with HAART. Ann Behav Med 31(2): 155-164.
- Jones DL, Ishii Owens M, Lydston D, Tobin JN, Brondolo E, et al. (2010) Self-efficacy and distress in women with AIDS: the SMART/EST women's project. AIDS Care 22(12): 1499-1508.
- Garnefski N, Kraaij V, Schroevers M (2010) Effects of a cognitive behavioral self-help program and a computerized structured writing intervention on depressed mood for HIV-infected people: a pilot randomized controlled trial. Patient Educ Couns 80(2): 304-307.
- Balfour L, Kowal J, Silverman A, Tasca GA, Angel JB, et al. (2006) A randomized controlled psycho-education intervention trial: Improving psychological readiness for successful HIV medication adherence and reducing depression before initiating HAART. AIDS Care 18(7): 830-838.
- Eller LS, Kirksey KM, Nicholas PK, Corless IB, Holzemer WL, et al. (2013) A randomized controlled trial of an HIV/AIDS Symptom Management Manual for depressive symptoms. AIDS Care 25(4): 391-399.
- Nakimuli-Mpungu E, Wamala K, Okello J, Alderman S, Odokonyeroet R, et al. (2015) Group support psychotherapy for depression treatment in people with HIV/AIDS in northern Uganda: A single-center randomized controlled trial. Lancet HIV 2(5): 190-199.
- Heckman TG, Heckman BD, Anderson T, Lovejoy TI, Mohr D, et al. (2013) Supportive-expressive and coping group teletherapies for HIV-infected older adults: A randomized clinical trial. AIDS Behav 17(9): 3034-3044.
- Van Luenen S, Garnefski N, Spinhoven P, Spaan P, Dusseldorp E, et al. (2018) The Benefits of Psychosocial Interventions for Mental Health in People Living with HIV: A Systematic Review and Meta-analysis. AIDS Behav 22(1): 9-42.
- Heckman TG, Sikkema KJ, Hansen N, Kochman A, Heh V, et al. (2011) A randomized clinical trial of a coping improvement group intervention for HIV-infected older adults. J Behav Med 34(2): 102-111.
- Petersen I, Hanass Hancock J, Bhana A, Govender K (2014) A group-based counselling intervention for depression comorbid with HIV/AIDS using a task shifting approach in South Africa: A randomized controlled pilot study. J Affect Disord 158: 78-84.
- Simoni JM, Pantalone DW, Plummer MD, Huang B (2007) A randomized controlled trial of a peer support intervention targeting antiretroviral medication adherence and depressive symptomatology in HIV-positive men and women. Health Psychol 26(4): 488-495.
- Arseniou S, Arvaniti A, Samakouri M (2014) HIV infection and depression. Psychiatry Clin Neurosci 68(2): 96-109.
- Sherr L, Clucas C, Lampe F, Harding R, Johnson M, et al. (2012) Gender and mental health aspects of living with HIV disease and its longer-term outcomes for UK heterosexual patients. Women Health 52(3): 214-233.
- Logie CH, Marcus N, Wang Y, Kaida A, Patricia O'Campo, et al. (2019) A longitudinal study of associations between HIV-related stigma, recent violence and depression among women living with HIV in a Canadian cohort study. J Int AIDS Soc 22(7): 25341.
- te Vaarwerk MJ, Gaal EA (2001) Psychological distress and quality of life in drug-using and non-drug-using HIV-infected women. Eur J Public Health 11(1): 109-115.
- Ciesla JA, Roberts JE (2001) Meta-analysis of the relationship between HIV infection and risk for depressive disorders. Am J Psychiatry 158(5): 725-730.
- Closson K, Osborne C, Smith DM, Kesselring S, Eyawo O, et al. (2018) Factors Associated with Mood Disorder Diagnosis Among a Population Based Cohort of Men and Women Living with and without HIV in British Columbia Between 1998 and 2012. AIDS Behav 22(5): 1530-1540.
- Adams C, Zacharia S, Masters L, Coffey C, Catalan P (2016) Mental health problems in people living with HIV: changes in the last two decades: the London experience 1990-2014. AIDS Care 28 Suppl 1: 56-59.
- Sauceda JA, Lisha NE, Neilands TB, Christopoulos K A, Mathews W C, et al. (2019) Cognitive-affective depressive symptoms and substance use among Latino and non-Latino White patients in HIV care: An analysis of the CFAR network of integrated clinical systems cohort. J Behav Med 42(3): 561-566.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- International Journal of Anaesthesia and Research (ISSN:2641-399X)
- Journal of Alcoholism Clinical Research
- Journal of Spine Diseases
- Journal of Clinical Trials and Research (ISSN:2637-7373)
- International Journal of Surgery and Invasive Procedures (ISSN:2640-0820)
- Stem Cell Research and Therapeutics (ISSN:2474-4646)
- Journal of Cell Signaling & Damage-Associated Molecular Patterns