3028
Views & Citations2028
Likes & Shares
Background: Advances in antiretroviral therapy in the Human Immunodeficiency Virus (HIV) had led to increased longevity but also to long-term complications, mostly represented by drug-induced liver injury and non-alcohol-related steatohepatitis. Vertically HIV-infected youths are more likely susceptible to liver injury, because of the duration of HIV disease and drugs’ exposure. Noninvasive methods to assess and to monitor long-term liver disease are a priority, especially in pediatric population. To date, studies with a complete assessment of liver function combining both noninvasive clinical tests and instrumental imaging are scarce among the pediatric population, especially among vertically-HIV-infected patients.
Aim: The aim of our study is to perform a complete assessment of liver function among vertically-HIV-infected youths on antiretroviral therapy, combining biochemical markers, noninvasive scores and instrumental imaging (liver ultrasound and transient elastography),and compare it with healthy subjects.
Methods: A group of 41 vertically-HIV-infected children, adolescents and young adults, negative for HBV and HCV infections and a group of age and sex matched healthy controls were enrolled. In all patients a complete liver serum and instrumental assessment were performed.
Results: This study showed a global good liver function in vertically HIV-infected youths, but a significant difference of some mean serum values (AST, ALT, γGT, LDL, HDL, apoliprotein A1, alpha-2-macroglobulin), clinical scores (APRI, FLI) and liver stiffness was found between the two groups.
Conclusion: These data suggest that noninvasive assessment of liver steatosis and fibrosis should be a part of the routine clinical management in vertically HIV-infected young patients to exclude the long-time drug toxicities and comorbidities of HIV infection.
Keywords: HIV liver disease, Liver fibrosis, Liver stiffness, NAFLD
Abbreviations: HIV: Human Immunodeficiency Virus; AST: Aspartate Aminotransferase; ALT: Alanine-Aminotransferase; γGT: Gamma Glutamil-Transferase; LDL: Low Density Lipoprotein; HDL: High Density Lipoprotein; FIB-4: Fibrosis score-4; APRI: AST to Platelet Ratio Index; FLI: Fatty Liver Index; HAART: Highly Active Antiretroviral Therapy; NAFLD: Non-Alcoholic-Fatty-Liver-Disease; TE: Transient Elastography; HBV: Hepatitis B Virus; HCV: Hepatitis C Virus; CDC: Centre for Disease Control and Prevention; RNA: Ribonucleic Acid; PCR: Polymerase Chain Reaction; BMI: Body Mass Index; ULN: Upper Limit of Normal; kPa: kilo Pascal; HGF: Hepatocyte Growth Factor
KEYPOINTS
Advances in HIV therapy had led to long-term complications like liver-related diseases.HIV-infected children are more susceptible to liver injury, because they face longtime infection and drugs’ exposition.
Noninvasive liver assessment should be part of routinely management of these patients.
INTRODUCTION
Advances of antiretroviral therapy (HAART) in management of Human Immunodeficiency Virus (HIV)-infected patients have resulted in increased longevity, but also in long-term complications [1]. During the last decade, liver-related diseases have become a leading cause of hospitalization and death in these patients, representing 9%-12% of deaths in HIV-infected patients [2, 3].
Due to well-controlled HIV, opportunistic infections involving the liver are now rarely seen and the most frequent causes of liver diseases in patients with HIV include drug-induced liver injury, viral hepatitis, and both alcohol and non-alcohol-related steatohepatitis (Non-alcohol-fatty liver-disease, NAFLD). Among these, NAFLD has become a new concern in the clinical management of HIV positive patients [4]. NAFLD involved a spectrum of condition from steatosis, with accumulation of triglycerides in the hepatocytes, to fibrosis and cirrhosis.
The pathophysiological mechanisms of liver steatosis and fibrosis in HIV-infected patients are multiple and complex. They include chronic inflammatory activation and immune-mediated injury due to HIV infection itself, with direct liver damage and cytotoxicity; antiretroviral therapy effects, with lipotoxicity; metabolic alterations caused by HIV and HAART, including insulin-resistance; mitochondrial toxicity and injury, with oxidative stress patterns activation; and hepatic inflammation correlated with gut microbial translocation to portal circulation [5-8].
Currently vertically HIV-infected children survive into adulthood and face longtime infection and treatment, so they are more likely susceptible to liver injury, because of the duration of HIV disease and the long exposition to drug toxicity.
The gold standard assessment of hepatic alterations is biopsy, but it is an invasive method, with possible side effects. For that reason, noninvasive methods to assess and to monitor long-term liver disease are a priority, especially in pediatric population. They can include biochemical markers, inidces and scores (Fibrosis score-4 [FIB-4], AST to Platelet Ratio Index [APRI], Fatty Liver Index [FLI]) and imaging diagnostics (abdominal ultrasonography, transient elastography [TE]).
APRI and FIB-4 have been reported to predict hepatic fibrosis in perinatal HIV-infected children [9-12]. FLI is another algorithm developed to detect steatosis [13]. Regarding instrumental methods, TE is a noninvasive test evaluating liver stiffness, which is correlated to the stage of liver fibrosis; its high negative predictive value allows excluding advanced fibrosis [14, 15].
To date, studies on liver damage among vertically-HIV-infected children are limited and in particular studies with a complete assessment of liver function combining both noninvasive clinical tests and instrumental imaging are scarce.
The aim of this study is to evaluate the prevalence of liver dysfunction using noninvasive methods in a population of vertically-HIV-infected children, adolescents and youths and to compare it with a control group.
METHODS
Study design
We performed a cross-sectional study in 41 HIV-vertically-infected children, adolescents and youths followed at Pediatric Infectious Disease Unit-“Luigi Sacco” Hospital, Milan, to assess their liver function with noninvasive methods.
Inclusion criteria comprehended maternal-fetal transmission of HIV infection, age 8-30 years, absence of hepatic disease, hepatitis and other medical condition during the study.
Exclusion criteria were hepatitis B and C co-infection, other viral hepatitis, genetic, oncological or autoimmune hepatic diseases, chronic pharmacological therapy (except for antiretroviral therapy) and alcohol or substance abuse.
We recorded patients’ sex, age, HIV staging according to the Centre for Disease Control and Prevention (CDC) classification [16], past and present exposure and number of HAART regimen for each patient. Peripheral blood T lymphocytes and subclass assay (CD4, CD8) were determined by flow cytometry. Plasma HIV RNA was calculated by VERSANT HIV-1 RNA 1.5 Assay (kPCR) with VERSANT kPCR Molecular System (Siemens Healthcare Diagnostics).
A control group of 16 age and sex matched healthy controls was also enrolled. For each HIV patient and each control subject we performed anthropometric assessment, including weight, height, Body Mass Index (BMI) and waist circumference. Weight was measured to the nearest 0.1 kg using a beam scale (Seca GmbH & Co. KG, Hamburg, Germany) and height was measured to the nearest 0.1 cm using a wall-mounted stadiometer (Holtain Ltd, Crosswell, UK). BMI was calculated as weight ((kg)/height (m2)). Waist circumference was taken with a soft tape in standing position, considering the narrowest circumference between the costal margin and the iliac crest.
Written informed consent was collected from all the study’s participants and/or from their legal tutors, as appropriate, according to the Declaration of Helsinki and its further amendments. The study was conducted conforming to ethical standards.
Liver Function Assessment
Biochemical markers: All blood samples were collected after an overnight fasting. For each HIV patient and each healthy subject liver function was assessed by aspartate-aminotransferase (AST), alanine-aminotransferase (ALT), gamma-glutamyl-transpeptidase (γGT), total and fractionated bilirubin, haptoglobin, total cholesterol, high density lipoprotein (HDL) cholesterol, low density lipoprotein (LDL) cholesterol, triglycerides, A1 apolipoprotein and alpha-2-macroglobulin.
Measurements were analyzed according to the manufacturer’s instruction on the same automate (Architect c16000, Abbott).
Indices and Scores: FLI, APRI and FIB-4 are algorithms used for the detection of liver fibrosis and fatty liver disease.
FLI was calculated using Bedogni’s formula [13]:
e(0.953 × ln (triglycerides) + 0.139 × BMI + 0.718 × ln (γGT) + 0.053 × waistcircumference - 15.745)/(1 + e(0.953 × ln (triglycerides) + 0.139 × BMI + 0.718 × ln (γGT) + 0.053 × waistcircumference - 15.745 ) × 100
Triglycerides are expressed as mg/dL, BMI as kg/m2, γGT as IU/L and waist circumference in cm.
As recommended by Bedogni et al., FLI
APRI was calculated using Wai’s formula [17]:
ULN stands for upper limit of normal, expressed as IU/L, and platelet count as platelets x109.
As recommended by Wai et al., values ≤ 0.5 and ≥ 1.5 are predictive of absence and presence of significant hepatic fibrosis, respectively. APRI≤ 1 and ≥ 2 are predictive of absence and presence of cirrhosis, respectively.
FIB-4 was calculated using Sterling’s formula [18]:
Age is expressed in years, AST and ALT in IU/L and platelet count as platelets x109.
As recommended by Sterling at al., FIB-4 ≤ 1.45 and ≥ 3.25 are predictive of absence and presence of significant hepatic fibrosis, respectively.
Instrumental evaluation
Each participant of the study underwent abdominal ultrasound to detect the presence of liver steatosis and fibrosis. It was conducted by the same operator (L.M.), expert in liver evaluation.
TE was performed with Fibro Scan (Echosens, Paris, France), according to the manufacturer’s instructions, by the same trained operator (G.G.) in same condition for each study’s participant. A standard ultrasound evaluation was preventively conducted between two ribs, with patient in supine position with right arm in abduction. The aim was to identify a portion of the liver’s right lobe without main vascular structures or gallbladder’s interposition. The TE probe transducer was placed perpendicularly to cutaneous surface of the chosen area between two ribs. At least ten measurements for each patient were performed. The median value, expressed in kilopascal (kPa), was considered to be representative of liver stiffness. We accepted as reliable results those with the success rate at least 60% and the interquartile range less than 30%.
We considered stiffness cut-off values used for HIV infected patients: 12.5 kPa for cirrhosis (F4) [19]
Statistical analysis
Descriptive data are expressed as median, standard deviation and range values. Prevalence is expressed as percentage. Box and Whiskers Plots were performed to show data distribution in HIV-infected and healthy groups. T-test was used to compare the analysis in the two groups. P values below 0.05 are considered to support statistical significance. Statistical analysis was performed using STATA 14 software (StataCorp, College Station, Texas, USA).
RESULTS
Population
Forty-one vertically-HIV-infected children, adolescents and youths were enrolled. Their mean age was 18.7 years old (8-27). Twenty-seven of them were female (27/41, 65.9%).
Sixteen comparable healthy subjects were also enrolled, with mean age of 23.7 years old (16-29). Thirteen of them (13/16, 81.3%) were female.
Among HIV patients, 39/41 (95%) were on HAART, with a mean duration of 16 years (3 months–24 years).
Features regarding the severity of HIV infection and the characteristics of HAART regimens are summarized in Table 1.
Considering anthropometric assessment, among HIV patients mean weight was 55.4 kg (24.5-87) and mean height was 159.6 cm (124.7-180). In control group mean weight was 59.6 kg (46-74.6) and mean height was 171.9 cm (157-182). Regarding BMI assessment, 11/41 HIV-infected patients (27%) were overweight, with mean BMI of 21.3 kg/m2 (15.8-28.9). All healthy controls showed normal weight, with mean BMI of 20 kg/m2 (16.9-24.9). Comparing the two populations, T-test did not show a statistical significant difference in the anthropometric assessment (p=0.11).
Biochemical markers
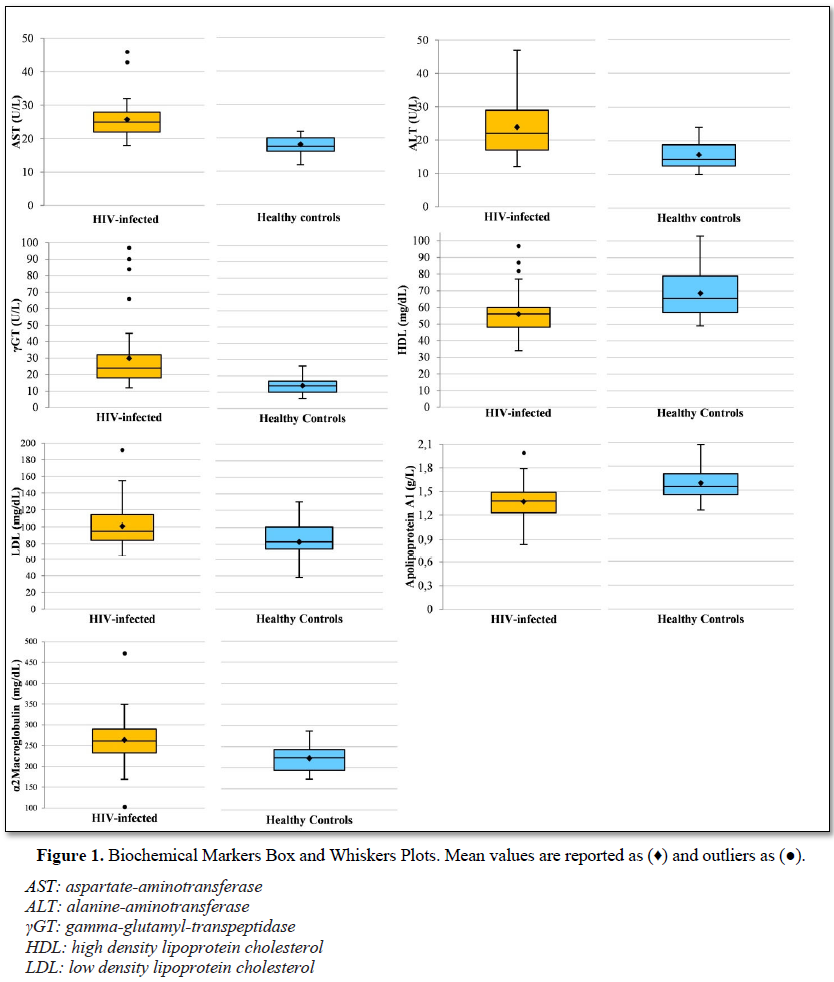
AST was elevated in 2 HIV-infected patients (2/41, 5%) and normal in all healthy subjects. ALT was normal in all subjects. γGT was increased in 4 HIV-infected patients (4/41, 10%) and normal in control group. Two (2/41, 5%) HIV-infected subjects showed slight increase in total serum bilirubin concentrations (>1.5 mg/dL), mainly indirect bilirubin. Three healthy controls (3/16, 18.8%) had elevated total bilirubin concentrations, with increase of both direct and indirect bilirubin. Haptoglobin showed values above reference limits calculated by age in 4 HIV-infected subjects (4/41, 10%), whereas it was under reference limits in all healthy subjects. Total cholesterol was elevated in 9 HIV-infected patients (9/41, 22%), and in 5 healthy controls (5/16, 31%). HDL was under reference limits in 8 HIV patients (8/41, 19.5%) and in one healthy control (1/16, 6%). LDL was above reference limits in 18 HIV patients (18/41, 44%), and in 4 healthy controls (4/16, 25%). Triglycerides were elevated in 4 HIV patients (4/41, 10%) and in 2 healthy controls (2/16, 12.5%). Apolipoprotein A1 was lower in one HIV patient (1/41, 2%), and was normal in all healthy controls. Alpha-2-macroglobulin was higher in 22 HIV-infected patients (22/41, 54%) and in 2 healthy subjects (2/16, 12.5%).
Mean value, standard deviation and range of AST, ALT, γGT, bilirubin, total cholesterol, HDL, LDL, triglycerides, apolipoprotein A1 and alpha-2-macroglobulin are shown in Table 2.
T-test showed significant difference between HIV-infected patients and healthy subjects in mean values of AST (25.81 vs 18.06 IU/L, p<0.01), ALT (23.88 vs 15.81 IU/L, p<0.01), γGT (29.93 vs 14 IU/L, p<0.01), HDL (56 vs 68.63 mg/dL, p<0.01), LDL (101.07 vs 82.88 mg/dL, p<0.01), apolipoprotein A1 (1.37 vs 1.59 g/L, p<0.01) and alpha 2 macroglobulin (263.71 vs 222.75 mg/dL, p<0.01). No significant differences in haptoglobin, total cholesterol and triglycerides mean values were found in the two groups (Table 2 and Figure 1).
Scores
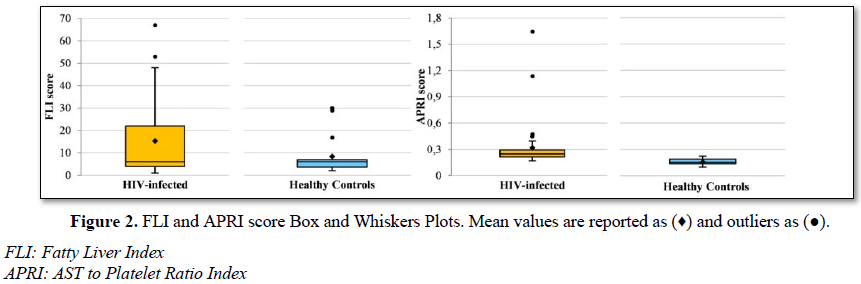
FLI was 60 in one of them (1/41, 2%). Among healthy control group, FLI reached the value of 30 only in one patient.
APRI was 2, had APRI 1.64 and also high FIB-4 value (2.3) and FLI (53), with not performed TE due to abdominal fat and a normal liver ultrasound. All control subjects had APRI (Figure 2).
FIB-4 values were normal in 40/41 HIV patients and in all healthy subjects (not exceeding the value of 0.58 in the healthy group). No significant differences in FIB-4 mean values were found between the two populations (0.48 vs. 0.38, p>0.05).
Instrumental analysis
Abdominal ultrasound showed mild steatosis in 6 HIV-infected patients (6/41, 15%) and mild hepatomegaly in 4/41 (10%). No splenomegaly was found. Ultrasonography was normal in all healthy subjects.
TE was performed in all subjects, but in 2 HIV patients the assessment was invalidated because of abdominal fat. According to Metavir Score, F0 stage was found in 21/39 HIV patients (54%); F1 in 13/39 (33%); F2 in 4/39 (10%), and 1 patient showed F3 stage. Fourteen healthy controls (14/16, 87.5%) showed F0 stage and 2/16 (12.5%) F0-F1 stage. Considering stiffness, 34 HIV-infected patients (34/41, 87%) had values (Figure 3).
DISCUSSION
In this study we described the state of biomarkers of liver disease in a cohort of vertically-HIV-infected children, adolescents and young adults. Up to date, there remain concerns about the exact contribution of HAART and HIV itself to hepatic damage [7, 20-22]. Moreover, NAFLD is one of the emergent diseases in HIV-infected people, as the result of a dysmetabolic process involving dyslipidemia and insulin resistance.
Liver biomarkers and other noninvasive methods are widely used in the clinical practice to assess liver dysfunction instead of invasive methods (e.g. liver biopsy).
As markers of hepatic damage, we used AST, ALT and γGT and the prevalence of their elevations in our study was lower than other studies [23,24]. AST were above reference limits only in 5% of our patients and none of them had ALT elevation. Rubio et al. [23] considered a population of vertically HIV-infected children and found increased AST and ALT values in 46% and 31% of them, respectively.
Stirnadel et al. [24] analyzed liver biochemical markers in 312 HIV-infected children, among whom 25% had ALT elevation. γGT values are associated with HGF (Hepatocyte Growth Factor), implicated in liver fibrosis development process. In our study, only 10% of HIV-infected patients had elevated γGT values, whilst prevalence in literature is 19% [23]. Despite that, we found a significant difference of AST, ALT and γGT mean values between HIV-infected patients and healthy subjects, with greater liver dysfunction in HIV group.
As serum markers of liver disease, also apolipoprotein A1 and alpha 2 macroglobulin were collected: they have proved to be a predictive marker of liver fibrosis in literature. In vitro studies about the deposition of liver fibrotic septa showed the reduction in apolipoprotein A1 levels, which remained trapped into extracellular matrix proteins bonds [25]. Alpha 2 macroglobulin, instead, is elevated during a fibrotic process because it could activate hepatic stellate cells and it could inhibit degradation of extracellular matrix protein due to its protease inhibiting activity [26]. In our study, alpha-2-macroglobulin values were higher in 54% of HIV patients, while apoliprotein A1 was found lower in only 1 HIV patient, with significant difference of both mean values between HIV and healthy population.
There is a high prevalence of dyslipidemia in vertically-HIV-infected patients, as consequence of chronic viral infection and HAART exposure, in particular to PI (protease inhibitors) [27]. Lainka et al. [27] analyzed 37 vertically-HIV-infected children, with LDL values elevation in 47% and HDL values reduction in 12% of them [27]. Our study showed similar prevalence: 44% of HIV-infected children, adolescents and young adults had high LDL values and 19.5% had low HDL values. These data suggest that dyslipidemia is an important concern in HIV-infected young patients and it can be involved in hepatic damage progression, so an appropriated HAART regimen should be chosen.
Our study also considered combined scores to identify liver steatosis and fibrosis. FLI was used to find patients with greater risk of fatty liver. Although only one patient (2%) showed pathologic value of FLI>60 indicating fatty liver, a significant difference between HIV and healthy population emerged. However, up to date, this index lacks of validation in HIV patients and further studies are necessary.
As marker of liver fibrosis, APRI is reported in some previous studies involving HIV-infected children [9, 10, 12, 28]. Two studies from Siberry et al. [9, 10] used 1.5 cut-off to define the presence of significant liver fibrosis. One of these study involved a population of Latin-American vertically-HIV-infected children, where APRI was >1.5 in 3.2% [9]; the other one involved, instead, a population of US infected children, where APRI was >1.5 in 0.8% [10]. Aurpibul et al. [28] considered a cohort of Asian HIV-infected children with APRI>1.5 in 8.5% of them. The only study conducted in Europe is one from Pokorska-Spiewak et al. [12]. In a population of Poland HIV-infected children APRI was above normal values (>0.5) in 5% of patients and 1% of them showed APRI values >1.5, indicative for significant fibrosis. Our study showed similar prevalence of APRI alteration, with increased values (>0.5) in two HIV patients (5%). Even if only one patient had APRI >1.5, we proved a significant difference between HIV and control group, indicating that long-term liver follow-up in vertically HIV-infected children and youths is needed.
The FIB-4 index has been validated to predict hepatic fibrosis in HIV-HCV co-infected patients [18]. It enables identification of patients with severe fibrosis and cirrhosis (F3–F4 in METAVIR scale for liver pathology), according to published ranges to define mild, moderate and advanced disease (<1.45, 1.45-3.25, >3.25) [18]. In HIV patients, FIB-4 basal values and their variation over the time are important predictive factors of major hepatic events and death [29]. Few previous studies used FIB-4 in HIV-infected children. Aurpibul et al. [28] identified FIB-4 alteration (>1.3) in 2.7% of Asian HIV children prior to HAART and in another 0.7% during follow-up. Kapogiannis et al. [11] found increased FIB-4 values in 2% of a population of US vertically-HIV-infected children, without a significant statistical difference compared to healthy children (2% vs 1%, p=0.42). In the Poland HIV-infected population from Pokorska-Spiewak, FIB-4 was >1.45 in only one patient (1%), with HCV co-infection [12]. In our study, the prevalence of FIB-4 alteration is comparable to literature, because it was increased in only one HIV-infected young patient (2%) and a significant difference between HIV and healthy subjects was not found.
To complete the noninvasive liver assessment we performed TE. There are no studies reporting the prevalence of liver stiffness alterations in vertically-HIV-infected children. Previous studies proved that TE is a valid noninvasive method to discriminate significant liver fibrosis and cirrhosis from non-fibrotic stages also in pediatric population, especially with chronic liver diseases and NAFLD [14,30]. Behairy et al. [30] showed a statistical correlation between liver stiffness and histological fibrotic stage, but it pointed out the need of fixing different cut-off values according to different etiologies of hepatic damage. Data on TE utilization in HIV-infected children are lacking and further studies are needed. In our study, 13% of HIV-infected young patients had stiffness >7.1 kPa, standing for significant fibrosis (F ≥2), however the prevalence is 42% and 21.1% in two studies involving HIV-infected adults [15,31]. The difference between these percentages can be explained by the progression of hepatic damage over the time. Our study also demonstrated a statistical significant difference of liver stiffness mean values of between HIV-infected patients and healthy group. So a complete liver assessment including TE is important also in HIV-infected children and not only in adults.
Our study has some limitations: the small size of the two groups; the lack of a structured measurement of alcohol intake; the lack of standardization in pediatric age of APRI, FIB-4 and FLI, as well as TE in HIV-infected children. In addition, liver biopsies were not performed to validate the results of the noninvasive assessment.
CONCLUSION
In conclusion, vertically HIV-infected patients on HAART do not exhibit relevant biochemical and structural liver abnormalities. Although within a normal range, the statistically significant difference between the two groups in mean liver biomarkers (AST, ALT, γGT, LDL, HDL, apoliprotein A1, alpha-2-macroglobulin), indices/scores (APRI, FLI) and stiffness, suggests that a complete noninvasive assessment of liver steatosis and fibrosis should be routinely performed during the long-term follow up of vertically HIV-infected patients.
1. Patel K, Hernán MA, Williams PL, Seeger JD, McIntosh K, et al. (2008) Long-term effectiveness of highly antiretroviral therapy on the survival of children and adolescents with HIV infection: A 10 year follow-up study. Clin Infect Dis 46: 507-515.
2. Weber R, Sabin CA, Møller NF, Reiss P, El-Sadr WM, et al. (2006) Liver-related deaths in persons infected with the Human Immunodeficiency Virus: The D:A:D study. Arch Intern Med 166: 1632-1641.
3. Bica I, McGovern B, Dhar R, Stone D, McGowan K, et al. (2001) Increasing mortality due to end stage liver disease in patients with Human immune deficiency virus infection. Clin Infect Dis 32: 492-497.
4. Maurice JB, Patel A, Scott AJ, Patel K, Thursz M, et al. (2017) Prevalence and risk factors of nonalcoholic fatty liver disease in HIV-mono infection. AIDS 31: 1621-1632.
5. Jones M, Núñez M (2012) Liver toxicity of antiretroviral drugs. Semin Liver Dis 32: 167-176.
6. Puri P, Kumar S (2016) Liver involvement in human immunodeficiency virus infection. Ind J Gastroenterol 35: 260-273.
7. Debes JD, Bohjanen PR, Boonstra A (2016) Mechanism of accelerated liver fibrosis progression during HIV infection. J Clin Transl Hepatol 4: 328-335.
8. Kaspar MB, Sterling RK (2017) Mechanism of liver disease in patients infected with HIV. BMJ Open Gastro 4: e000166.
9. Siberry GK, Cohen RA, Harris DR, Cruz MLS, Oliveira R, et al. (2014) NISDI PLACES Protocol, prevalence and predictors of elevated aspartate aminotransferase-to-platelet ratio index in Latin American perinatally HIV-infected children. Pediatr Infect Dis J 33: 177-182.
10. Siberry GK, Patel K, Pinto JA, Puga A, Mirza A, et al. (2014) Elevated Aspartate Aminotransferase-to-Platelet Ratio Index in Perinatally HIV-Infected Children in the United States. Pediatr Infect Dis J 33: 855-857.
11. Kapogiannis BG, Leister E, Siberry GK, Dyke RBV, Rudy B, et al. (2016) Prevalence of and progression to abnormal non-invasive markers of liver disease (aspartate aminotransferase-to-platelet ratio index and Fibrosis-4) among US HIV-infected youth. AIDS 30: 889-898.
12. Pokorska-Spiewak MP, Perka AS, Popielska J, Oldakowska A, Coupland U, et al. (2017) Prevalence and predictors of liver disease in HIV-infected children and adolescents. Sci Rep 7: 12309.
13. Bedogni G, Bellentani S, Miglioli L, Masutti F, Passalacqua M, et al. (2006) The Fatty Liver Index: A simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol 6:33.
14. Nobili V, Vizzutti F, Arena U, Abraldes JG, Marra F, et al. (2008) Accuracy and reproducibility of transient elastography for the diagnosis of fibrosis in pediatric non-alcoholic steatohepatitis. Hepatology 48: 442-448.
15. Morse CG, McLaughlin M, Proschan M, Koh C, Kleiner DE, et al. (2015) Transient elastography for the detection of hepatic fibrosis in HIV-monoinfected adults with elevated aminotransferases on antiretroviral therapy. AIDS 29: 2297-2302.
16. Centers for Disease Control and Prevention (CDC) (2014) Revised surveillance case definition for HIV infection. United States MMWR Recomm Rep 63: 1.
17. Wai CT, Greenson JK, Fontana RJ, Kalbfleisch JD, Marrero JA, et al. (2003) A simple noninvasive index can predict both significant fibrosis and cirrhosis in patients with chronic hepatitis C. Hepatology 38: 518-526.
18. Sterling RK, Lissen E, Clumeck N, Sola R, Correa MC, et al. (2006) Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 43: 1317-1325.
19. Sagir A, Glaubach B, Sahin K, Graf D, Erhardt A, et al. (2015) Transient elastography for the detection of liver damage in patients with HIV. Infect Dis Ther May 4: 355-364.
20. Rockstroth JK, Mohr R, Behrens G, Spengler U (2014) Liver fibrosis in HIV: Which role does HIV itself, long-term drug toxicities and metabolic changes play? Curr Opin HIV AIDS 9: 365-370.
21. Moodie EE, Pai NP, Klein MB (2009) Is antiretroviral therapy causing long-term liver damage? A comparative analysis of HIV-mono-infected and HIV/Hepatitis C Co-Infected Cohorts. PLoS ONE 4: e4517.
22. Kovari H, Sabin CA, Ledergerber B, Ryom L, Reiss P, et al. (2009) Antiretroviral Drugs and Risk of Chronic Alanine Aminotransferase Elevation in Human Immunodeficiency Virus (HIV)-Monoinfected Persons: The data collection on adverse events of anti-hiv drugs study. Open Forum Infect Dis 3: ofw009.
23. Rubio A, Monpoux F, Huguon E, Truchi R, Triolo V, et al. (2009) Noninvasive procedures to evaluate liver involvement in HIV-1 vertically infected children. J Pediatr Gastroenterol Nutr 49: 599-606.
24. Stirnadel HA, Bains C, Lakshmi M, Pandey R, Webb DJ, et al. (2012) Background incidence of liver chemistry abnormalities in pediatric clinical trials for conditions with and without underlying liver disease. Regul Toxicol Pharmacol 62: 329-335.
25. Paradis V, Mathurin P, Ratziu V, Poynard T, Bedossa P (1996) Binding of apolipoprotein A-I and acetaldehyde-modified apolipoprotein A-I to liver extracellular matrix. Hepatology 23: 1232-1238.
26. Kawser CA, Iredale JP, Winwood PJ, Arthur MJ (1998) Rat hepatic stellate cell expression of alpha2-macroglobulin is a feature of cellular activation: Implications for matrix remodelling in hepatic fibrosis. Clin Sci (Colch) 95: 179-186.
27. Lainka E, Oezbek S, Falck M, Ndagijimana J, Niehues T, et al. (2002) Marked dyslipidemia in human immunodeficiency virus-Infected children on protease inhibitor-Containing antiretroviral therapy. Pediatrics 110: e56.
28. Aurpibul L, Bunupuradah T, Sophan S, Boettiger D, Wati DK, et al. (2015) Prevalence and incidence of liver dysfunction and assessment of biomarkers of liver disease in HIV-Infected asian children. Pediatr Infect Dis J 34: e153-e158.
29. Mussini C, Lorenzini P, De Luca A, Lichtner M, Lapadula G, et al. (2015) Prognostic value of the fibrosis-4 index in human immunodeficiency virus type-1 infected patients initiating antiretroviral therapy with or without hepatitis C virus. PLoS ONE 10: e0140877.
30. Behairy Bel-S B, Sira MM, Zalata KR, Salama el-Se, Allah MAA, et al. (2016) Transient elastography compared to liver biopsy and morphometry for predicting fibrosis in pediatric chronic liver disease: Does etiology matter? World J Gastroenterol 22: 4238-4249.
31. Lombardi R, Lever R, Smith C, Marshall N, Rodger A, et al. (2017) Liver test abnormalities in patients with HIV mono-infection: assessment with simple noninvasive fibrosis markers. Ann Gastroenterol 30: 349-356.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Clinical Trials and Research (ISSN:2637-7373)
- Journal of Alcoholism Clinical Research
- Ophthalmology Clinics and Research (ISSN:2638-115X)
- Journal of Forensic Research and Criminal Investigation (ISSN: 2640-0846)
- Journal of Cell Signaling & Damage-Associated Molecular Patterns
- Oncology Clinics and Research (ISSN: 2643-055X)
- Journal of Immunology Research and Therapy (ISSN:2472-727X)