2952
Views & Citations1952
Likes & Shares
Background: The rate, risk factors and consequence of adjacent level disease (ALD) in cervical disc arthroplasty (CDA) remains unclear. The purpose of this study is to determine the rate, risk factors and clinical outcome of ALD in CDA.
Methods: Retrospective review of 166 patients with a minimum five-year follow-up of a CDA was performed. Multi-level surgeries, including hybrid procedures, were included. Multiple implant types were included. The two inter-vertebral discs (IVD) cranial of the CDA were monitored for radiologic degeneration. No funding was attained for this study and the authors declare no conflict of interest.
Results: The rate of ALD in CDA was 28.3%, with most affecting the immediately adjacent IVD (27.4% and 7.6% respectively p=0.000). Age (p=0.209) and sex (p=0.201) did not relate to ALD, nor did pre-operative degeneration (p=0.117) or spondylolisthesis (p=0.315) adjacent to the CDA. The number of operated levels (p=0.890), number of fused levels (p=0.354), implant alignment (0.255), ROM (p=0.569) and implant induced spondylolisthesis (p=0.402) did not affect the rate of ALD. However, fusion of the most cranial implant significantly increased the rate of ALD (p=0.032).
The visual analogue pain scale (VAS) was significantly worse in those patients with ALD (VAS neck 2.7 versus 1.5 p=0.029; VAS arm 0.9 versus 2.3 p=0.002). The five-year functional outcomes were worse in those who developed ALD (NDI 20.1 versus 12.3 p=0.011).
No patients required a reoperation during the course of this study.
Conclusion: ALD is common after CDA and worsens the patient’s functional outcome, but not their need for revision surgery within five years. Fusion of the most cranial implant is a major risk for developing ALD, whereas the initial implant alignment and function do not construe a risk.
Keywords: Adjacent level disease, Proximal junctional failure, Junctional kyphosis, Cervical disc arthroplasty, Spondyloli
INTRODUCTION
Degenerative disorders of the cervical spine are an increasing health burden and are predicted to affect two-thirds of the population [1]. Most patients are successfully treated non-operatively, but a select few require operative intervention [2,3]. Traditionally, there have been two surgical approaches (anterior or posterior) and two techniques (fusion or non-fusion) utilized, depending on the pathology being treated.
For anterior approaches, the most common procedures performed for inter-vertebral disc (IVD) degeneration, are anterior cervical discectomy and fusion (ACDF) and cervical disc arthroplasty (CDA). The presumed benefit of CDA is that it preserves motion and therefore reduces the relative forces on the adjacent levels, which is theoretically believed to reduce the risk of adjacent level degeneration (ALD) [4,5]. However, to date there is no conclusive evidence to support the notion that CDA reduces the rate of ALD when compared to ACDF, with the relative rates of ALD, within the first five years being similar between the procedures [6-8].
Unfortunately, the limited number of publications assessing the outcome of CDA beyond five years limits our understanding on whether CDA reduces the risk of longer term ALD. However, it is likely that ALD is multi- factorial and not solely related to adjacent stiffness. This is evidenced by procedures with intuitively no, or little, risk of causing clinically significant stiffness, such as a posterior foraminotomy, which still carry a 5% 10 year risk of symptomatic ALD [9].
When compared to CDA, patients undergoing ACDF are more likely to be older, with more advanced degeneration, including facet involvement, mal-alignment and disc space collapse. Therefore, a selection bias may limit our understanding of the cause of ALD if we simply compare ACDF to CDA. We therefore aimed to account for these biases and solely analyze patients undergoing CDA to determine the rate, risk factors and clinical outcome of adjacent level disease (ALD) within this population.
MATERIALS AND METHODS
We hypothesized that ALD in CDA patients is rare and most likely caused by subsequent auto-fusion of the CDA. We performed a retrospective review of 166 patients with prospectively collected standardized radiographs (AP, standing lateral and flexion/extension) of CDAs performed within our institution with a minimum five-year follow-up. Patients were included if they had pre-operative, early post-operative (5 years from procedure) radiographs. Patients were excluded if they had rigid ankylosing conditions or severe mal-alignment requiring deformity correction (chin-on-chest deformity and loss of horizontal gaze). Multi-level CDAs were included, as well as hybrid procedures (multi-level surgery with CDA and anterior cervical discectomy and fusion (ACDF)). Only sub axial articulations were included and therefore the C1/2 articulation was excluded.
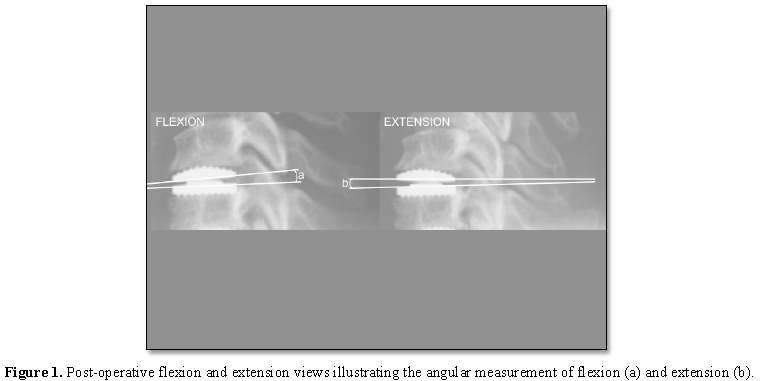
Sagittal alignment was based on the posterior vertebral line, on the early post-operative standing lateral x-ray, as described by Harrison et al. [10]. The flexion and extension views were used to determine the range of movement (ROM) and mid-flexion point (defined as the mid-point between maximal flexion and maximal extension) based on the angle between the upper and lower components of each implant, with extension defined as posterior convergence and flexion defined as anterior convergence (Figure 1).
Spondylolisthesis was defined as a break in the posterior vertebral line on the flexion or extension x-rays and graded according to the Meyerding classification [11]. IVD degeneration was graded according to the Kettler criteria using the AP and lateral standing profile views [12]. Post-operatively the adjacent levels were monitored for progressive degeneration (ALD) defined as an increase in Kettler grade [12]. Fusion of an implant was defined as 0° ROM on follow-up flexion/extension x-rays.
Neck and arm visual analogue pain scores (VAS) were recorded pre-operatively, early post-operatively and at five years post-operative. Functional outcomes (Neck Disability Index (NDI)), at five years post-operative. The clinical notes were reviewed for revision procedures, including adjacent level procedures.
Statistical methods
Descriptive statistics were conducted to describe the participants of this study. Following this, chi-square analyses were conducted in order to determine the association between ALD and age, sex, spondylolisthesis, implant type and level. Mann-Whitney tests assessed the relationship between ALD and the various post-operative non-parametric parameters (alignment, ROM, flexion, extension, mid-flexion point, VAS score and NDI). Fisher exact tests were conducted to determine the association between fusion of the upper instrumented level and ALD.
Statistical analysis was performed using SPSS software. Statistical significance was defined as a two tailed p value of less than 0.05.
RESULTS
One hundred and sixty-six patients were reviewed. The average age of the cohort was 45 years (range 23-65 years), 51% were female (85 female, 81 male) and the average follow-up was 6.5 years (range 5-10 years). The CDAs used were the Bryan® disc from Medtronic (43 patients), the Discocerv® from Alphatech Spine (38 patients), Mobi-C® from LDR (31 patients) and the Baguera-C® from Spineart (54 patients). One hundred and sixty-six IVDs were assessed immediately adjacent and a further 159 IVDs were assessed two levels cranial to the most cranial implant.
Ninety-six patients had a single-level CDA, 58 had a double-level (43 double CDA and 15 hybrid procedures), 11 had a triple-level (five single-level CDA with double-level ACDF; one double-level CDA with single-level ACDF) and one had a four-level (triple-level CDA and single-level ACDF) procedure. Only two patients had an ACDF as their most cranial implant and both cases successfully fused.
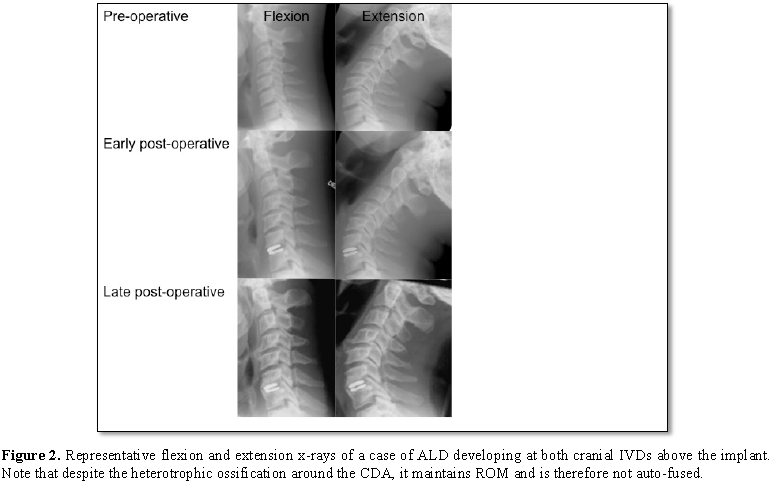
Adjacent level degeneration occurred in 47 patients (28.3%) and 57 (17.8%) of IVDs assessed. There was no significant difference of age (p=0.209) or sex (p=0.201) between those patients who did or did not develop ALD. The most common level of the most cranial implant was C5/6, (range C3/4-C6/7). There was no relationship identified between the cervical level of the most cranial implant and ALD (p=0.33). However, the rate of ALD was significantly higher for those IVDs immediately adjacent to the most cranial implant than the two IVD spaces cranial of the implant (27.4% and 7.6% respectively p=0.000) (Figure 2).
Radiological evidence of pre-operative degeneration was seen in 13.0% of IVDs. The rate of ALD was higher in those IVDs with pre-operative degeneration (27.0%) than in those without pre-operative degeneration (16.5%), but this did not reach statistical significance (p=0.117). A maximum of grade 1 (<25%) pre-operative spondylolisthesis of the adjacent levels was identified and this did not significantly increase the rate of post-operative degeneration (13.8% ALD in those with pre-operative spondylolisthesis compared to 19.1% for those without pre-operative spondylolisthesis p=0.315).
There was no relationship between the number of levels operated and the risk of ALD (p=0.890) (Table 1).
In addition, in multi-level surgery, there was no discernable relationship between ALD and the number of levels fused (p=0.354) (Table 2) or the total number of fused or replaced levels (including congenital fusions) (p=0.883). However, there was a statistically significant, although clinically insignificant, relationship between the average ROM per level and the rate of ALD (7.2° for those without ALD and 5.3° for those with ALD p=0.04).
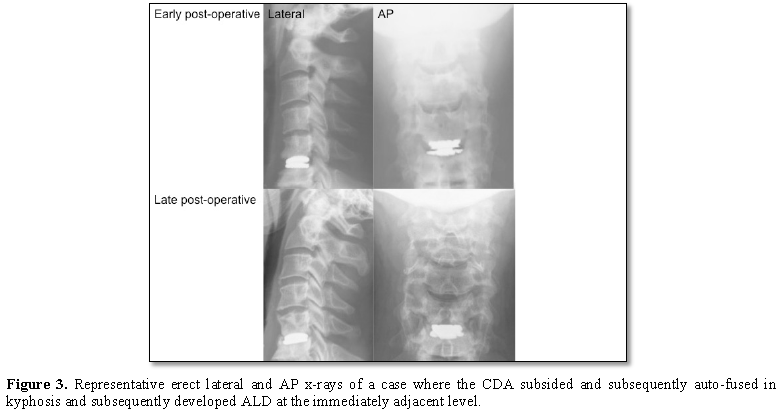
Auto-fusion of the most cranial implant occurred in 32 patients (19.3%) and was associated with a significantly increased risk of ALD (28.3% with fusion in contrast to 16.3% without fusion p=0.032) (Figure 3).
There was no significant relationship between the most cranial CDA early alignment (p=0.255), ROM (p=0.569), maximal flexion (p=0.433), maximal extension (p=0.511) and mid-flexion point (p=0.510) with ALD (Table 3). Furthermore, in multi-level surgery, there was no significant difference between the early average alignment of each operated level (p=0.255) and ALD.
There was a significantly higher rate of ALD seen in the Bryan® CDA compared to all other prostheses (p=0.005) (Table 4). This trend was also apparent when only the immediately adjacent IVD was assessed, although this failed to reach statistical significance (14.3% for the Bryan® compared to an average 7.6% for all implant types p=0.205).
There was no significant difference in pre-operative adjacent level degeneration and fusion of the most cranial implant identified between prosthetic types (p=0.130 and p=0.297, respectively) (Table 5).
Only a maximum of grade 1 spondylolisthesis of the most cranial implant was identified. Spondylolisthesis of the most cranial implant itself was not related to an increased rate of ALD (17.4% ALD in those with spondylolisthesis of the most cranial CDA in contrast to 18.6% ALD for those without spondylolisthesis p=0.402).
No patients required further surgery on their cervical spine during the timeframe of this study. This included reoperations or revisions of their implants and extension for ALD. There was no significant difference in the pre- and early post-operative VAS scores between those who developed and those who did not develop ALD (Table 6). However, after five years there were significantly worse neck and arm pain scores identified in those with ALD.
Functional outcomes were significantly worse in those patients who developed ALD (NDI 20.1 versus 12.3 p=0.011).
DISCUSSION
This study has found that radiographic ALD is common and occurs in 28.3% of patients undergoing CDA. We found no evidence that the patient’s age or sex predisposed to ALD. However, our results illustrate that ALD most commonly occurs at the IVD immediately adjacent to the most cranial implant, rather than the IVD two spaces from the implant (27.4% and 7.6%, respectively, p=0.000). In addition, we found that there was a significantly higher rate of ALD in patients with auto-fusion of the most cranial CDA (28.3% with fusion in contrast to 16.3% without fusion p=0.032).
This confirms that a stiffened motion segment is a risk factor for ALD and therefore supports the notion of motion preservation. However, this does not, in itself, support the use of a CDA in the most cranial operated level [13,14]. In this series an overall CDA fusion rate of 18.1% was identified. Thus, CDAs do not eliminate the concern of motion segment stiffness. Furthermore, the long-term outcome of CDA, particularly when coupled with adjacent fusion procedures, remains unknown. Therefore, to extrapolate our results to support the use of “topping off” multi-level surgery with a CDA is unfounded.
Intuitively, it seems likely that pre-operative adjacent level degeneration increases the risk of ALD. Our results support this presumption (27.0% rate of ALD in those with pre-operative degeneration in contrast to 16.5% without pre-operative degeneration), although this result did not reach statistical significance (p=0.117). Further research with higher numbers of patients is required to confirm this finding.
We found no evidence that pre-operative adjacent spondylolisthesis predisposes to ALD. However, we only had patients with mild (<25%) spondylolisthesis identified in this series, thus the effect of more severe slippage remains unknown.
We hypothesized that the most cranial implants alignment and mobility contributed to the rate of ALD. However, we found no relationship between ALD and the most cranial implants early post-operative alignment, ROM, maximal flexion, maximal extension or mid-flexion point.
Because this study excluded severe deformity we did not assess the global cervical alignment. However, we did assess the alignment of each operated level as a pragmatic surrogate to what surgeons can achieve intra-operatively if corrective osteotomies are not performed. We found no relationship between individual level alignment and ALD. Therefore our results suggest that surgeons do not need to modify implant sizing to correct cervical alignment in order to prevent ALD in such a population group.
We also found no relationship between ALD and the total number of levels operated or the number of levels fused. Although, we did find a statistically significant increase in ALD with less ROM per operated level. However, despite being statistically significant (p=0.04), we feel the less than 2° difference per level between those who developed ALD and those who did not (5.3° versus 7.2°, respectively), is clinically insignificant and within the inaccuracies of radiographic measurements. We therefore feel that this finding supports surgeons treating each IVD on its own merits in multi-level surgery, including fusing a segment when appropriate, rather than solely performing CDAs at every level.
We are uncertain as to why the Bryan® CDA had a significantly higher rate of ALD than the other prostheses assessed (p=0.005). Our findings show that auto-fusion of the most cranial implant significantly increases the rate of ALD (p=0.032) and therefore a higher rate of implant auto-fusion would account for this finding. However, the Bryan® CDA was not found to have a significantly higher rate of auto-fusion of the most cranial implant when compared to the other CDAs (p=0.297). In addition, although the increased rate of ALD in adjacent level pre-operative degeneration failed to reach statistical significance (p=0.117), it may affect the prosthetic result. Again, however, patients with the Bryan® CDA did not have a significantly increased rate of pre-operative adjacent level degeneration (p=0.130). We are therefore uncertain as to why there is a higher rate of ALD in our cohort of patients who underwent a Bryan® CDA and therefore further research into the implant specific rates of ALD are advocated.
We feel that ALD can be defined radiologically, clinically (with severe symptomatic and functional impact) and surgically (with a surgical indication). Our study specifically utilized a radiological approach to the diagnosis of ALD, with subsequent review of the clinical and surgical implications.
To date, there is no radiographic score that can accurately define the prognosis and therefore treatment of disc degeneration [15]. We used the Kettler criteria because it is reportedly the most reliable and specific x-ray measure for disc degeneration in the cervical spine [12,15]. Although cross-sectional MRI imaging would offer earlier identification of ALD, our cohort did not undergo such follow-up scans. Because none of our cases required a reoperation we feel that the x-ray follow-up was sufficient to identify significant ALD in our cohort.
Despite none of our patients requiring a reoperation on their cervical spine within the timeframe of this study, we found that patients meeting the radiographic criteria for ALD had significantly worse five-year neck and arm pain scores as well as worse functional outcomes (p=0.011). While it should be recognized that the cause of the poorer pain scores and functional outcomes may relate to factors other than ALD, the statistical significance suggests that the radiographic ALD identified in this study represents a clinically important condition and warrants longer-term follow-up to determine if delayed operative intervention is necessary.
In conclusion, the rate of ALD in this cohort of patients undergoing CDA was 28.3%. Most ALD affects the immediately adjacent IVD and auto-fusion of the most cranial implant is an important risk factor. However, pre-operative adjacent level spondylolisthesis, the number of levels operated or fused and the early post-operative alignment and function of each CDA do not affect the rate of ALD. ALD is associated with increased pain and functional disability but not an increased rate of reoperation within five-years.
1. Todd AG (2011) Cervical spine: Degenerative conditions. Curr Rev Musculoskelet Med 4: 168-174.
2. DePalma AF, Subin DK (19650) Study of the cervical syndrome. Clin Orthop Relat Res 38: 135-142.
3. Rothman RH, Rashbaum RF (1978) Pathogenesis of signs and symptoms of cervical disc degeneration. Instr Course Lect 27: 203-215.
4. Mummaneni PV, Amin BY, Wu JC (2012) Cervical artificial disc replacement versus fusion in the cervical spine: A systematic review comparing long-term follow-up results from two FDA trials. J Evid Based Spine Care 3: S59-66.
5. Wu JC, Hsieh PC, Mummaneni PV (2015) Spinal motion preservation surgery. BioMed Res Int 372502.
6. Botelho RV, Moraes OJ, Fernandes GA (2010) A systematic review of randomized trials on the effect of cervical disc arthroplasty on reducing adjacent-level degeneration. Neurosurg Focus 28: E5.
7. Nunley P, Jawahar A, Kerr EJ (2012) Factors affecting the incidence of symptomatic adjacent-level disease in cervical spine after total disc arthroplasty: 2 to 4 year follow-up of 3 prospective randomized trials. Spine 37: 445-451.
8. Fallah A, Akl EA, Ibrahim GM (2012) Anterior cervical discectomy with arthroplasty versus arthrodesis for single-level cervical spondylosis: A systematic review and meta-analysis. PLoS One 7: e43407.
9. Clarke MJ, Ecker RD, Krauss WE (2007) Same-segment and adjacent-segment disease following posterior cervical foraminotomy. J Neurosurg Spine 6: 5-9.
10. Harrison DE, Harrison DD, Cailliet R (2000) Cobb method or Harrison posterior tangent method: Which to choose for lateral cervical radiographic analysis. Spine 25: 2072-2078.
11. Meyerding HW (1932) Spondylolisthesis. Surg Gynecol Obstet 54: 371-377.
12. Kettler A, Rohlmann F, Neidlinger-Wilke C (2006) Validity and interobserver agreement of a new radiographic grading system for intervertebral disc degeneration: Part II. Cervical spine. Eur Spine J 15: 732-741.
13. Etebar S, Cahill DW (1999) Risk factors for adjacent-segment failure following lumbar fixation with rigid instrumentation for degenerative instability. JNS Spine 90: 163-169.
14. Eck J, Humphreys SC, Lim TH (2002) Biomechanical study on the effect of cervical spine fusion on adjacent-level intradiscal pressure and segmental motion. Spine 27: 2431-2434.
15. Vialle EN, Vialle LRG, Simões CE (2016) Clinical-radiographic correlation of degenerative changes of the spine - Systematic review. Columna 15: 325-329.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- International Journal of Surgery and Invasive Procedures (ISSN:2640-0820)
- Stem Cell Research and Therapeutics (ISSN:2474-4646)
- Journal of Immunology Research and Therapy (ISSN:2472-727X)
- Journal of Renal Transplantation Science (ISSN:2640-0847)
- Journal of Forensic Research and Criminal Investigation (ISSN: 2640-0846)
- Journal of Cardiology and Diagnostics Research (ISSN:2639-4634)
- Ophthalmology Clinics and Research (ISSN:2638-115X)