Research Article
Clinical Predictors for the Diagnosis of Delusional Infestation
3041
Views & Citations2041
Likes & Shares
Background: Patients with delusional infestation (DI) can be challenging to diagnose.
Objective: To identify clinical predictors for a final diagnosis of DI.
Methods: This was a retrospective, single-center cohort study of 132 patients. Data was collected from surveys given to all patients at the beginning of their first visit. At the last visit, all patients received a final diagnosis of ‘delusional’ or ‘not-delusional’.
Results: Three factors on initial presentation were found to be independently associated with a final diagnosis of DI: high impact of disease on quality of life (QOL), somatic symptom severity score ≥5, and the presence of a ‘specimen sign’.
Limitations: This was a retrospective study using a non-validated survey.
Conclusion: For providers concerned their patient may have DI, it may be helpful to ask questions about their QOL, somatic symptoms, and noting the presence of the ‘specimen sign’.
Keywords: Delusional infestation, Delusions of parasitosis, Psychodermatology, Matchbox sign, Specimen sign
INTRODUCTION
Delusional infestation (DI), also known as ‘delusional parasitosis’ or ‘perceptions of infestation’, [1] is a condition in which a patient incorrectly believes that they have an infestation of the skin, whether it be parasites or a nonliving substance. Disease severity often presents as a spectrum of phenotypes, including an overvalued idea of which a patient can be reassured, a somatic preoccupation that focuses on symptoms in multiple body systems, or a delusional state of which a patient cannot be talked out of [2]. While DI is generally considered to be a rare disease, survey data in Britain and France has reported that every dermatologist has seen at least one case during their career [3,4]. DI can occur in patients of all demographics, however the most classic patient affected by DI is a middle-aged or elderly woman with few social contacts, normal cognitive and social function, and who may be disabled or retired [5,6]. This disease causes significant distress to patients and negatively impacts their quality of life. Of those patients with DI who seek help for their condition, up to 90% choose to see a dermatologist to address their concerns [7,8]. It is important to rule out any organic cause of a delusional disorder, including nutritional deficiencies, renal disease, substance use,8 prescribed or over-the-counter medications, and neurologic disease [9,10]. It is also often difficult to distinguish these patients from those with primary skin disorders as they can have similar clinical presentations. As a result, it can take several visits for a practitioner to conclude that their patient has a delusional disorder.
Recently it has been suggested that patients with longer durations of untreated delusional infestation have poorer outcomes and are less likely to respond to treatment [11]. The goal of this study was to identify predictors associated with delusional infestation so that these patients can be diagnosed more efficiently and receive timely and appropriate care.
MATERIALS AND METHODS
Participants
This was a retrospective cohort study of 132 patients seen at the Seton/University of Texas Dermatology Complex Medical Clinic between March 2014 and January 2017. This is a referral-based clinic that sees patients with complicated medical dermatology diagnoses, such as autoimmune and immunobullous disease, alongside patients with skin picking and delusional infestation. Every patient was seen and diagnosed clinically by the same dermatologist throughout the study period. When determining a patient’s final diagnosis, the diagnosis rendered at the final visit during the study period was used. Patient diagnoses were grouped into either not delusional (ND) or delusional (D). ND group included any primary skin disorder as well as skin picking disorders, while the D group included patients thought to have either a somatoform preoccupation or a delusional state per the delusional spectrum proposed by Brown [2]. The study was approved by the University of Texas at Austin Dell Medical School Institutional Review Board.
Data Collection
Data was gathered via a survey administered at the beginning of each patient visit. The survey included a Patient Health Questionnaire 15-Item Somatic Symptom Severity Scale (PHQ15), which is a standardized, validated test used to assess for signs of depression and somatic symptoms [12]. The survey also included questions about symptom quality, effect of symptoms on quality of life (QOL), duration of symptoms, number of doctors seen for condition, and goals of care (Figure 1). Once completed by the patient, the clinical staff noted on this survey whether the patient brought any skin or parasite samples (often called the ‘specimen sign’ or ‘matchbox sign’) to their appointment. They also recorded whether the patient had any physical exam findings (primary or secondary skin lesions). Demographic data was abstracted from the electronic medical record, including age and gender.
Data Analysis
Descriptive analysis was performed for all variables collected. In order to assess predictors of a diagnosis of a delusional disorder, chi-square testing and univariate logistic regression analysis was performed. A multivariate analysis was also conducted to determine whether or not predictors identified on univariate analysis were independently associated with presence of DI when adjusted for other risk factors. If the survey was completed at multiple visits, only the first visit survey was included in the data analysis. Standardized cut-offs were used for determining if a PHQ15 score was minimal (4 or less) or significant (5 or greater) [12]. When determining cut-offs for our continuous variables such as quality of life effects, the authors (JR and AJP) came up with numbers they felt were a logical cut-off point (ex: 1-5 and 6-10 for quality of life split, 3 or greater for number of doctors seen). Statistical analysis was performed using Stata 15 software (StataCorp. 2017. Stata Statistical Software: Release 15. College Station, TX: StataCorp LLC). 56(58) + 76(55)/56+76. 3248+4180=7428.
RESULTS
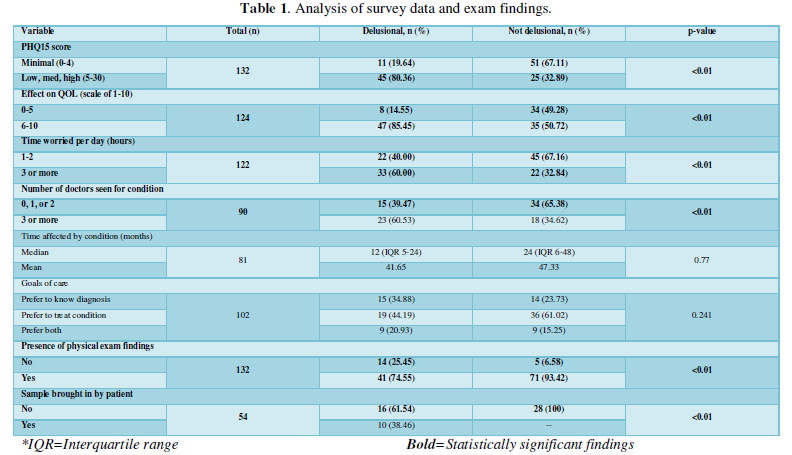
A total of 132 patients participated in this study. Fifty-six patients (42%) were given a diagnosis of D, and 76 were given a diagnosis of ND. There were 91 female patients and 41 male patients with a mean age of 56 years. Table 1 lists the results of the survey data and exam findings in patients diagnosed with D vs. those diagnosed as ND. Six variables were more likely to be associated with a diagnosis of DI: PHQ15 score ≥5 (80.36% D, 20.64% ND; p<0.01); effect of symptoms on QOL ≥6 on scale of 1-10 (85.45% D, 14.55% ND; p<0.01); worrying about condition ≥3 hours per day (60.00% D, 32.84% ND; p<0.01); having seen ≥3 doctors for condition (60.53% D, 34.62% ND; p<0.01); presence of physical exam findings (74.55% D, 93.42% ND; p<0.01); presence of a sample or ‘specimen sign’ (38.46% D, 0% ND; p<0.01). Some participants left parts of the survey blank which explains the difference in sample size for each variable studied.
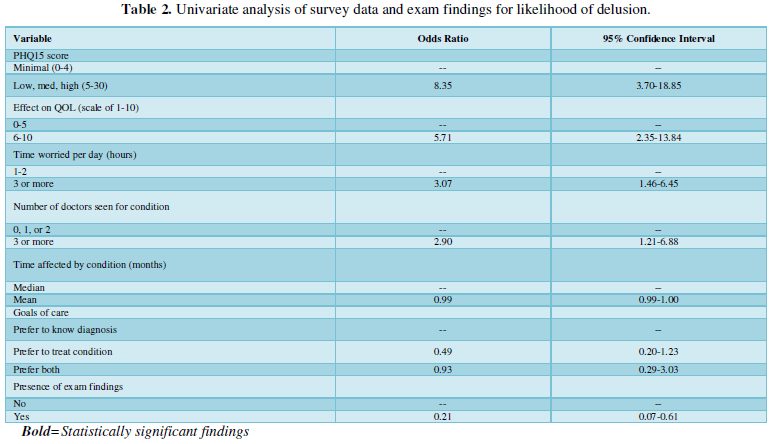
A univariate analysis of the data was also performed (Table 2). Factors associated with a higher likelihood of being diagnosed with DI were: PHQ15 score ≥5 (OR 8.35, 95% Confidence Interval (CI) 3.70-18.85); effect of symptoms on daily life ≥6 on scale of 1-10 (OR 5.71, 95% CI 2.35-13.84); worrying about condition ≥3 hours per day (OR 3.07, CI 1.46-6.45); having seen ≥3 doctors for condition (OR 2.90, 95% CI 1.21-6.88); presence of physical exam findings (OR 0.21, 95% CI 0.07-0.61).
Upon further examination using a multivariate analysis of the data, two variables remained statistically significant: a PHQ15 score ≥5 (OR 6.20, 95% CI 2.63-14.66) and effect of symptoms on daily life ≥6 on scale of 1-10 (OR 4.38, 95% CI 1.69-11.35).
The presence of a sample, or ‘specimen sign’, was found to be 38.46% sensitive and 100% specific for being in the D group. As a result, the univariate and multivariate analyses could not be performed for this variable because no patients in the ND group brought in samples.
DISCUSSION
The goal of this study was to identify patient predictors or exam findings associated with a diagnosis of DI. This study found that two factors are independently associated with DI: high impact of disease on QOL and a high PHQ15 score. The presence of a ‘specimen sign’ on initial presentation was




highly specific for DI but the data analysis could not show an independent association. It is well documented that patients with skin disease can experience a significant impact on their QOL [13-15]. However, this study demonstrates that a more severe effect of skin disease on QOL can predict a final diagnosis of DI. Similarly, patients with DI appear to have a higher score on the PHQ15, which screens for somatic signs of depression. In this clinic’s experience, the key factor in patients with DI is when the skin problem’s effect on QOL or somatic symptoms is out of proportion to the skin findings on exam. These patients are likely to have a subtype of DI previously referred to as “somatoform preoccupation” [2].
The ‘specimen sign’ was also found to have a specificity of 100% for DI. Originally named the ‘matchbox sign’ when first described in 1983 [16] the ‘specimen sign’ has commonly been reported in patients with DI [16-18]. Prior to this study there has been no data directly comparing the presence of the specimen sign in both delusional and not delusional patients.
There are several limitations to this study that should be noted. First, this was a retrospective study with a small sample size. While a standard PHQ15 was used, portions of this survey have not been validated previously. Additionally, the study population may not be reflective of the general population seen by general dermatologists, as these patients were seen in a tertiary referral clinic. Another limitation is that some patients could not complete all the survey questions due to their disorganized behavior (most common in the delusional patients). Finally, this study only shows associations and causation cannot be determined based upon the study design.
When a patient presents with concerns of bugs on their skin, it is important for the clinician to perform a thorough evaluation to look for signs of infestation as well as secondary causes of delusional disorder such as medications. The authors recommend that providers ask about the impact of a patient’s disease on their quality of life and screen for somatic symptoms of depression. It may indicate a delusional disorder if the patient’s level of dysfunction exceeds that which the provider would expect for their clinical exam. If a patient presents with specimens, this may help signal a mental disorder as well. By offering these tools, we hope to expedite a diagnosis of DI and guide treatment options. Since DI is a special population of psychocutaneous disorders, these patients often benefit from referral to specialists or psychodermatology clinics which continue to grow in number [19].
ACKNOWLEDGEMENTS
We would like to thank Adewole Adamson, MD, MPP for his help with reviewing this manuscript and providing feedback on the format of the paper.
FUNDING SOURCES
None
CONFLICTS OF INTEREST
None declared
- Fried R, Heymann WR (2016) Considering the Lexicon of psychocutaneous disorders: The time for change has come. Dermatologist 24(4): 15-16.
- Brown GE, Sorenson E, Malakouti M, Leon A, Reichenberg JS, et al. (2014) The Spectrum of Ideation in Patients with Symptoms of Infestation: From Overvalued Ideas to the Terminal Delusional State. J Clin Exp Dermatol Res 5: 241.
- Lyell A (1983) The Michelson Lecture. Delusions of parasitosis. Br J Dermatol 108: 485-499.
- Bourgeois M, Rager P, Peyré F, Nguyen-Lan A, Etchepare JJ (1986) Incidence and aspects of the Ekbom syndrome. Study involving French dermatologists (about 150 cases). Ann Med Psychol (Paris) 144(6): 659-668.
- Foster AA, Hylwa SA, Bury JE, Davis MD, Pittelkow MR, et al. (2012) Delusional infestation: Clinical presentation in 147 patients seen at Mayo Clinic. J Am Acad Dermatol 67(4): 673.e1-673.e10.
- Freudenmann RW, Lepping P (2009) Delusional infestation. Clin Microbiol Rev 22: 690-732.
- Wong JW, Koo JY (2013) Delusions of parasitosis. Indian J Dermatol 58: 49-52.
- Lepping P, Noorthoorn EO, Kemperman PMJH, Harth W, Reichenberg JS, et al. (2017) An international study of the prevalence of substance use in patients with delusional infestation. J Am Acad Dermatol 77(4): 778-779.
- Santillan MR, Taylor DL, Reichenberg JS (2018) Delusional infestation in psychodermatology. G Ital Dermatol Venereol 153(4): 497-505.
- Reichenberg JS, Kroumpouzos G, Magid M (2018) Approach to a psychodermatology patient. G Ital Dermatol Venereol 153(4): 494-496.
- Romanov DV, Lepping P, Bewley A, Huber M, Freudenmann RW, et al. (2018) Longer Duration of Untreated Psychosis is Associated with Poorer Outcomes for Patients with Delusional Infestation. Acta Derm Venereol 98(9): 848-854.
- Kroenke K, Spitzer RL, Williams JBW (2002) The PHQ-15: Validity of a new measure for evaluating the severity of somatic symptoms. Psychosom Med 64(2): 258-266.
- Hong J, Koo B, Koo J (2008) The psychosocial and occupational impact of chronic skin disease. Dermatol Ther 21: 54-59.
- Choi J, Koo JY (2003) Quality of life issues in psoriasis. J Am Acad Dermatol 49: 57-61.
- Fowler JF, Ghosh A, Sung J, Emani S, Chang J, et al. (2006) Impact of chronic hand dermatitis on quality of life, work productivity, activity impairment and medical costs. J Am Acad Dermatol 54: 448-457.
- Lee WR (1983) The matchbox sign. Lancet 2(8344): 261.
- Bhushan P, Thatte SS, Kalra K (2015) Matchbox sign: Look before you label! Indian J Dermatol Venereol Leprol 81(5): 507-509.
- Freudenmann RW, Lepping P, Huber M, Dieckmann S, Bauer-Dubau K, et al. (2012) Delusional infestation and the specimen sign: A European multicentre study in 148 consecutive cases. Br J Dermatol 167(2): 247-251.
- Patel A, Jafferany M (2020) Multidisciplinary and holistic models of care for patients with dermatologic disease and psychosocial comorbidity: A systematic review. JAMA Dermatol 156(6): 686-694.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Ageing and Restorative Medicine (ISSN:2637-7403)
- Journal of Nursing and Occupational Health (ISSN: 2640-0845)
- Journal of Rheumatology Research (ISSN:2641-6999)
- Journal of Blood Transfusions and Diseases (ISSN:2641-4023)
- International Journal of Medical and Clinical Imaging (ISSN:2573-1084)
- Archive of Obstetrics Gynecology and Reproductive Medicine (ISSN:2640-2297)
- Chemotherapy Research Journal (ISSN:2642-0236)


