3070
Views & Citations2070
Likes & Shares
Epidermal nevus is a benign, congenital malformation or hamartoma of epidermal cells derived from embryonic ectoderm. Epidermal nevus is classified into specific variants with pertinent, incriminated epidermal structure besides clinical appearance, degree of involvement and distribution of lesions. Adjunctive categories of epidermal nevus are comprised of malformations of squamous epithelium which layers various cutaneous adnexa and are designated as “organoid” variant of epidermal nevus. Inflammatory linear verrucous epidermal nevus was initially scripted by Unna in 1896 and is contemplated as an exceptional variant of verrucous epidermal nevus. The condition commonly implicates the female population and is essentially refractory to therapy.
Keywords: Verrucous, Epidermal nevus, Inflammatory
DISEASE CHARACTERISTICS
Linear verrucous epidermal nevus commonly appears as linearly configured, verrucous papules or plaques distributed along Blaschko’s lines which are embryonic lines situated along the planes of ectodermal cleavage. Linear verrucous epidermal nevus demonstrates a unilateral lesion as cogitated with nevus unis lateralis or extensive bilateral incrimination as enunciated with ichthyosis hystrix. Linear verrucous epidermal nevus is associated with aberrations of central nervous system and/or skeletal deformities. However, oral mucosal lesions are infrequent. Linear verrucous epidermal nevus appears with the incidence of an estimated 1 to 3 neonates per 1000 live births. The condition is commonly congenital, can appear in children or adolescents and is equally distributed amongst males and females. Alternative clinical representation of epidermal nevus is contingent to linear, unilateral and extensive variants. Epidermal nevus is frequently localized although extra-cutaneous anomalies can concur. Specific syndromes can be associated with epidermal nevus such as keratitis, erythrokeratoderma and sensorineural deafness (KID syndrome), congenital hemidysplasia, ichthyosiform erythroderma and limb defects (CHILD syndrome) and Gardner’s syndrome. Epidermal nevus can clinically and histologically simulate lesions of psoriasis [1,3]. Epidermal nevus syndrome exemplifies the association of epidermal nevus with various skeletal, neurological and ophthalmic anomalies. Extensive epidermal nevi and concurrent systemic anomalies can indicate the appearance of an epidermal nevus syndrome. Inflammatory linear verrucous epidermal nevus is an infrequent variant of epidermal nevus which commonly appears at birth, infancy or early childhood although adults can be implicated. Inflammatory verrucous epidermal nevus generally enunciates as a sporadic disorder although familial instances can occur and the condition demonstrates a female predominance with a female to male ratio of 4:1. Classic clinical criterions for diagnosing inflammatory linear verrucous epidermal nevus were described by Altman and Mehregan in 1971 [1] with subsequent modifications from Morag and Metzker in 1985 [2]. Criterion are specified as early age of disease onset, predominance in females, frequent incrimination of the left lower limb, pruritus, distinctive psoriasis-like appearance and resistance to therapy. Features such as unilateral, pruritic linear epidermal nevus, bifid uvula, hypoplasia of enamel, alopecia cutis and skeletal anomalies as cogitated with spina bifida, intracranial lipomas and calcification are suggestive of inflammatory linear verrucous epidermal nevus. Bilateral lesions can occur. Inflammatory linear verrucous epidermal nevus should be contemplated with the appearance of extensive verrucous and pruritic epidermal nevi with or without co-existent systemic abnormalities. Inflammatory linear verrucous epidermal nevus is a potentially premalignant condition with an estimated one fifth (15% to 20%) persons depicting malignant transformation. Thus, a preliminary surgical elimination of lesions is recommended [3,4].
DISEASE PATHOGENESIS
Inflammatory linear verrucous epidermal nevus is engendered by somatic mutations as a component of genetic mosaicism and is associated with enhanced production of interleukins 1 and 6, tumor necrosis factor alpha (TNFα) and intercellular adhesion molecules. Exposure to ultraviolet radiation and chemical carcinogens accounts for predilection for malignant transformation which can manifest as rapid growth or superficial ulceration of lesion. Basal cell carcinoma, keratoacanthoma and squamous cell carcinoma can exceptionally develop in cutaneous linear epidermal nevus. Bowen’s disease, verrucous carcinoma and adnexal carcinoma can also originate within an epidermal nevus [4,5].
CLINICAL ELUCIDATION
Lesions are initially miniature, squamoid, pruritic and extend gradually to incriminate adjacent regions. However, lesions can be stationary and devoid of progression.
Clinical evaluation demonstrates enlarged, erythematous, well defined keratotic or verrucous patches with foci of hemorrhagic crust and multiple, confluent plaques and papules. Localized, verrucous papules or plaques which appear on the extremities, trunk or head and neck are denominated. Epidermal nevus can emerge as elevated, painless, erythematous and papillary lesion or delineate multiple, unilateral, patches and plaques recapitulating a wart-like appearance. Accompanying lesions appear as painless, linear and yellowish macules depicting a focal blaschkoid and linear distribution [5,6]. Inflammatory linear verrucous epidermal nevus characteristically exhibits unilateral, recurrent, inflammatory manifestations with features of chronic eczema or psoriasis associated with severe pruritus is exemplified. Typical clinical symptoms demonstrate intensely pruritic, inflammatory, erythematous papules which coalesce to configure well defined verrucous plaques. Aforesaid lesions emerge on the extremities along the distribution of Blaschko’s lines and infrequently in a curvilinear arrangement on the trunk. Dark tinged or brownish, mossy, pruritic papules with a linear configuration are elucidated on the neck, lumbar region, axilla, chest, shoulders and upper extremities. Verrucous papules can appear on the face, cheek, external ear, pre-auricular region, forehead and scalp with linear extensions. Intraoral papules appear as diffuse, sessile, linear and papillary lesions on the dorsum of tongue. Gingival inflammation is generalized and accompanied by gingival desquamation, enamel hyperplasia and dental caries. Subjects can delineate hemorrhagic features such as bleeding gums, skeletal abnormalities as delineated with scoliosis, asymmetrical lower extremities with anterior bowing and alopecia cutis [3,4].
HISTOLOGICAL ELUCIDATION
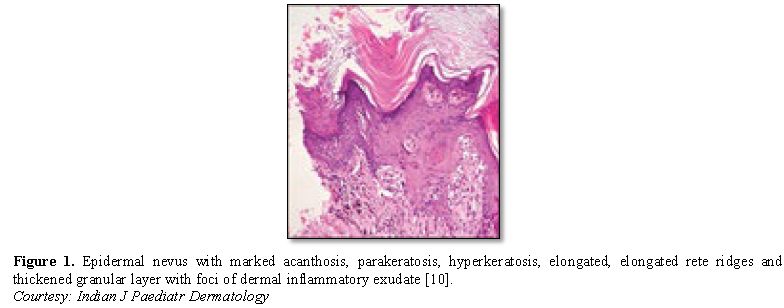
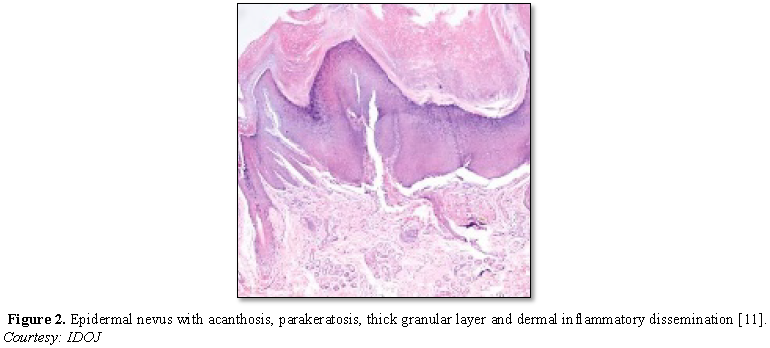
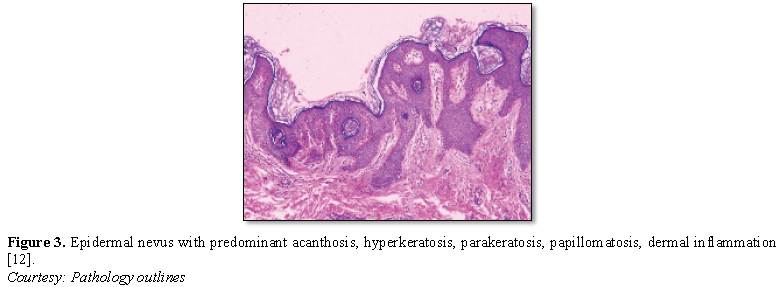
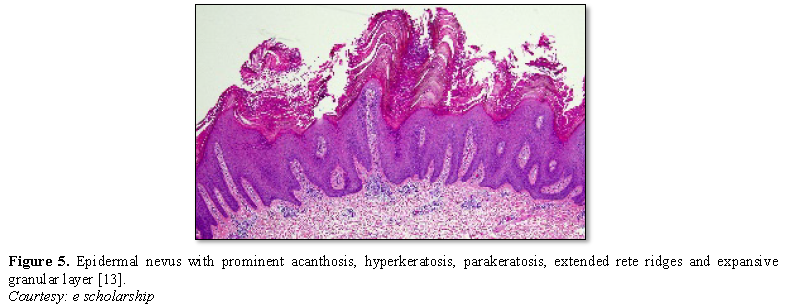
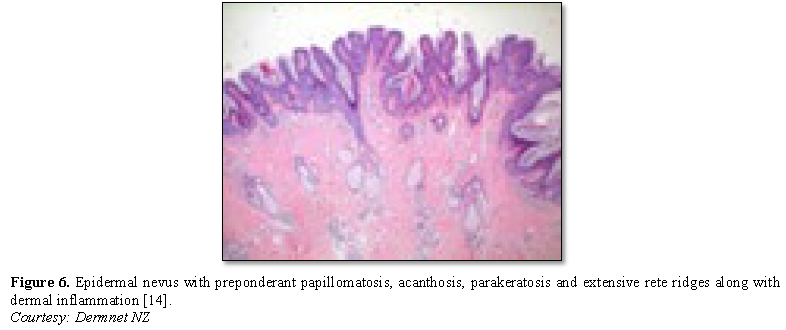
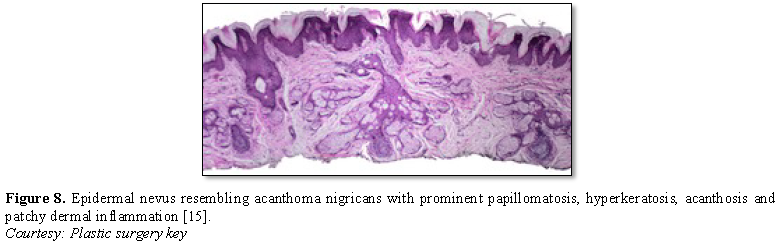
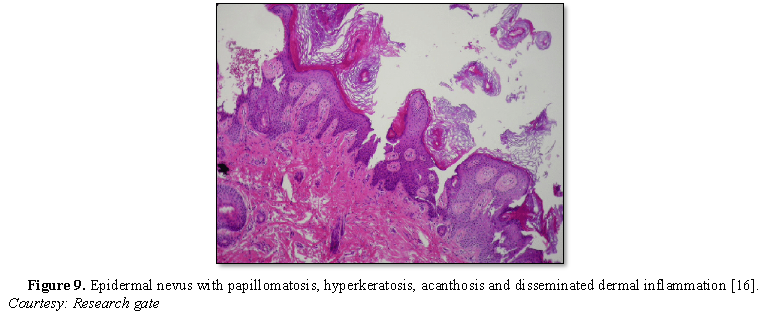
Epidermal nevus displays moderate hyperkeratosis, acanthosis, papillomatosis with a psoriasiform, inflammatory infiltration of the dermis or occurrence of Munro’s micro-abscesses. Elongated rete ridges and focal thickening of granular epithelial layer are accompanied by expansive columns of parakeratosis. Basal layer can occasionally depict mildly enhanced melanin pigment, thereby generating typical hues of tan or brownish lesions. Also, perimeter of the lesion is microscopically well demarcated from surrounding epithelium. Acanthosis and hyperkeratosis are accompanied by thick, shallow papillomatosis. Morphology often simulates the papillomatous variant of seborrheic keratosis. Occasionally, epidermolytic hyperkeratosis can arise within the lesion.
Certain exceptional histological patterns can originate in the epidermal nevus such as configurations recapitulating acanthosis nigricans, Hailey-Hailey disease, acantholytic dyskeratosis or associated conditions [5,6] (Figures 1-9).
DISEASE VARIANTS
Aplasia cutis congenita is a cutaneous deformity commonly arising in midline scalp and can be associated with epidermal nevus syndrome (ENS). Aplasia cutis congenita typically displays hairless, solitary cutaneous lesions with a distinct margin. Skeletal anomalies are cogitated in one half to two third (50% to 66%) individuals. Predominant primary osseous manifestations are described as incomplete bony development at various sites, vertebral defects, camptodactyly, clinodactyly, aberrant clavicles, rib asymmetry, shortened bones of extremities, posterior luxation of ankle joint and asymmetry of skull [3,5]. Secondary bony manifestations include kyphoscoliosis and limb hypertrophy. Radiographic assessment of skeletal bones demonstrate expansile bony lesions, thickening of bone cortex, sclerosis, bony trabeculae with designated radiolucent areas a uniform ground glass appearance and bony deformities. Central nervous system (CNS) manifestations of epidermal nevus syndrome occur in around half to three fourths (50% to 70%) subjects and are commonly defined by seizures and mental retardation. Associated neurologic features are denominated by hypotonia, hyperkinesia, hemiparesis, hemiplegia, cranial nerve palsies, hydrocephalus, hemi-megalencephaly, cortical lesions, cortical atrophy, ventricular abnormalities and intracerebral calcification [7,8].
Intra-spinal and intracranial lipomas, ocular defects such as astigmatism, choristomas, colobomas of eyelid, iris, choroid and retina, cortical blindness, nystagmus, oculomotor dysfunction, optic nerve dysfunction, ptosis and strabismus are additional complications of epidermal nevus syndrome. Oral lesions exceptionally appear in epidermal nevus syndrome and define unilateral or midline papules or nodules. Lesions with papillary or verrucous superficial surface appear on the lips, tongue, buccal mucosa, hard palate, tonsils, pharynx, soft palate and gingiva. Tooth anomalies include missing tooth, impacted tooth, and tooth of atypical magnitude, tooth hypoplasia, odontodysplasia and anomalous spacing between teeth. Intra-oral lesions can appear as wart-like, condylomata, mammilated or verrucous lesions and demonstrate various hues as of normal oral mucosa, yellow-white, tan, dark brown or grey [4,6].
DIFFERENTIAL DIAGNOSIS
Inflammatory linear verrucous epidermal nevus necessitates a distinction from various dermatoses such as adjunctive variants of epidermal nevus, Darier’s disease, linear porokeratosis, linear lichen planus, linear psoriasis and lichen striatus. Lichen striatus is a self-limiting condition which can clinically simulate inflammatory linear verrucous epidermal nevus and can be distinguished on account of early disease onset and lack of spontaneous regression. Segregation of epidermal nevus is required from acanthosis nigricans, a condition which usually depicts lesser quantities of hyperkeratosis and papillomatosis than a typical epidermal nevus. However, acanthosis nigricans-like variant of epidermal nevus can ensue. Confluent and reticulated papillomatosis of Gougerot and Carteaud (CARP) also necessitates a distinction from epidermal nevus. Papillomatous articulations within lesions of CARP are subtle and can be absent [7,8]. Epidermal nevus can recapitulate associated conditions with epidermal proliferation and papillomatosis within the epidermal arrangements. Adequate clinical information is mandated in cogent instances for appropriate distinction. Seborrheic keratosis often demonstrates an identical histology. However, epidermal nevus morphologically resembling seborrheic keratosis is often enunciated in children or young individuals or can emerge as congenital lesions. Verruca vulgaris can simulate epidermal nevus [9].
INVESTIGATIVE ASSAY
Dermoscopic examination of epidermal nevus typically reveals filiform zones exemplifying irregular and linear blood vessels with peripheral accentuation superimposed upon an erythematous backdrop. Few centroidal, enlarged, brownish spheres and circles can accompany the yellowish, homogenous foci. Histological assessment and a bi-annual clinical examination are necessitated to further elucidate an epidermal nevus. Dental radiographs can depict an absence of teeth or deviated nasal septum. Axial computerized tomography (CT) of the brain displays miniature, calcific foci enunciated within the frontal or parietal regions, radio-dense adipose tissue deposits accumulated upon bilateral cistern and cerebellopontine angle is indicative of intracranial lipomas. Lumbar radiographs delineate mild scoliosis with congenital schmorl’s node and spina bifida. Anterior bowing of lower extremity can occur with sclerosis of tibia [7,9].
THERAPEUTIC OPTIONS
Therapeutic modalities for managing epidermal nevus are diverse although a singular, consistently successful therapeutic option is lacking. Surgical eradication of the lesions is not a preferable mode of treatment on account of extensive scarring and relapsing lesions. Several treatment modalities can be applied in the management of inflammatory linear verrucous epidermal nevus. Pertinent methodologies include administration of intra-lesional and topical steroids, topical and systemic retinoids, topical 5-fluorouracil, podophyllin, dermabrasion, cryosurgery and surgical extermination of the lesions. However, an incomplete eradication of lesions or extensive scarring can ensue. Lasers can be employed to manage epidermal nevus and several options such as Argon, Erbium-YAG, pulsed dye and carbon dioxide laser are available. The contemporary pulsed, scanned carbon dioxide laser is a preferential mode of treatment [8,9].
Therapeutic response to the anti-psoriatic drug calcipotriol and anti TNF antibody such as etanercept is enunciated. Inflammatory linear verrucous epidermal nevus is markedly refractory to therapeutic intervention. It can be managed with topical glucocorticoids, intra-lesional corticosteroids, combined tretinoin and 5% fluorouracil. Besides, agents such as anthralin, tar, analogues of vitamin D3 can be adopted. Surgical extermination, cryotherapy with liquid nitrogen and carbon dioxide laser therapy is recommended and beneficial. However, a singular therapeutic option has not been consistently advantageous or deemed superior. However, as treatment of inflammatory linear verrucous epidermal nevus is controversial and invasive, an accurate determination of the condition is required [8,9].
1. Altman J, Mehregan AH (1971) Inflammatory linear verrucosae epidermal nevus. Arch Dermatol 104: 385-389.
2. Morag C, Metzker A (1985) Inflammatory linear verrucous epidermal nevus - Report of seven new cases and review of literature. Pediatr Dermatol 3: 15-18.
3. Senhaji G, Jouari OE, Gallouj S, Mernissi FZ (2018) Dermoscopy of inflammatory linear verrucous epidermal nevus syndrome (ILVEN). Cosmetol 2: 000113.
4. Kumar CA, Yelluri G, Raghav N (2012) Inflammatory linear verrucous epidermal nevus syndrome with its polymorphic presentation - A rare case report. Contemp Clin Dent 3: 119-122.
5. Avcioglu SN, Altinkaya SO, Küçük M, Yüksel H, Demircan-Sezer S, et al. (2013) Vulvar and perianal condyloma superimposed inflammatory linear verrucous epidermal nevus: a case report and review of literature. Case Rep Dermatol Med 261574.
6. Gianfaldoni S, Tchernev G, Gianfaldoni R, Wollina U, Lotti T (2017) A case of inflammatory linear verrucous epidermal nevus(ILVEN) treated with CO2 laser ablation. Open Access Macd J Med Sci 5: 454-457.
7. Behera B, Devi B, Nayak BB, Sahu B, Singh B (2013) Giant inflammatory linear verrucous epidermal nevus: Successfully treated with full thickness excision and skin grafting. Indian J Dermatol 58: 461-463.
8. Cabanillas M, Aneiros A, Monteagudo B, Santos-García D, Suárez-Amor O, et al. (2009) Epidermal nevus syndrome associated with polyostotic fibrous dysplasia, CNS lipoma and aplasia cutis. Dermatol Online J 15: 7.
9. Canyigit M, Oguz KK (2006) Epidermal nevus syndrome with internal carotid artery occlusion and intracranial and orbital lipomas. Am J Neuroradiol 27: 1559-1561.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- International Journal of Surgery and Invasive Procedures (ISSN:2640-0820)
- Journal of Alcoholism Clinical Research
- Journal of Cardiology and Diagnostics Research (ISSN:2639-4634)
- Stem Cell Research and Therapeutics (ISSN:2474-4646)
- Oncology Clinics and Research (ISSN: 2643-055X)
- International Journal of Anaesthesia and Research (ISSN:2641-399X)
- Journal of Immunology Research and Therapy (ISSN:2472-727X)