Research Article
Determination of Suitable Needle Length for New Epinephrine auto Injectors
3165
Views & Citations2165
Likes & Shares
Background: Epinephrine auto-injectors are expected to deliver epinephrine IM. The exposed needle length and pressure required to trigger the devices varies among brands and manufactured lots. With the currently available auto-injectors, the high-pressure devices have shorter exposed needle length than the low-pressure devices. As well, injection through clothing affects the depth of tissue delivery.
Objective: To study if it is possible to find a needle length for epinephrine autoinjectors that would avoid intraosseous/periosteal injections in all patients and at the same time not cause subcutaneous injections.
Methods: We assessed the likely location of injections of epinephrine autoinjectors, using ultrasound data from a study of 303 children and adolescents and 99 adults, 2-79 years old. Ultrasound was applied with high and low pressure. The skin to bone and skin to muscle distances were measured. The length of needles with no risk of intraosseous/periosteal injection was estimated. The risk of subcutaneous injection was investigated. The longest needle length not causing intraosseous injection applying low and high pressure on the ultrasound probe and the shortest approved needle for estimation of the risk for subcutaneous injection through naked skin and through thick clothes.
Results: When using high pressure, the risk of subcutaneous injection was very high. Using low pressure, there was a low risk of subcutaneous injection. These results are qualitative and not quantified as the original ultrasound data on pressure were not to the exact product specifications.
Conclusion: With the primary goal of avoiding intraosseous/periosteal injection, low pressure auto-injector delivery is more likely to avoid subcutaneous injection compared to high pressure auto-injectors. This occurs because low pressure does not compress the muscle significantly and the resulting the possible use of longer needles that are more likely to pass through the subcutaneous tissue into muscle.
Keywords: Auto-injector, Epinephrine, Intramuscular, Subcutaneous, Intraosseous, Periosteal, Skin to bone distance, Skin to muscle distance, Clothing, Needle length
Abbreviations: EAI: Epinephrine Auto-injector, HPEAI: High pressure EAI, LPEAI: Low pressure EAI, STMD: Skin to muscle distance, STBD: Skin to bone distance, STMDmin: Skin to muscle distance: low pressure, STMDmax: Skin to muscle distance: high pressure, 8 lb ≈ 35 N; STBDmin: Skin to bone distance, low pressure; STBDmax: Skin to bone distance, high pressure, 8 lb ≈ 35 N; SC: Subcutaneous; IM: Intramuscular; i.o./p: Intraosseous/periosteal.
TO THE EDITOR
Epinephrine is indicated for intramuscular (IM) injection in the treatment of anaphylaxis [1]. For the treatment of anaphylaxis in the community, epinephrine auto injectors (EAI) have been used since the late 1970’s [2]. Recently, we reported on the risk for subcutaneous (SC) and intraosseous/periosteal (i.o/p) injections, using ultrasound for measuring the distance from skin to muscle (STMD) and the skin to bone distance (STBD), respectively [3-9]. There was a risk of SC injection when using high pressure epinephrine auto-injectors (HPEAIs). There is also a risk of SC injection with low pressure EAIs (LPEAIs) in overweight and obese adults. High and low pressure EAIs is defined in the original papers [3-5]. There are two important factors influencing the risk of i.o/p injection and SC injection, the pressure applied to the EAI [8] and the length of the needle penetrating the skin. The variations in the lengths of EAI needles in the production of EAIs also may increase this risk [9]. In a pig cadaver study, Diacono et al. [10] showed that the whole orifice of the needle must pass completely through the fascia and perimysium to enter the muscle.
Duvauchelle et al. [11] confirmed that the injection bolus remained at the tip of the needle in the SC compartment after injection with an HPEAI, Anapen®. Furthermore, we have shown that injecting through thick clothes increases the risk of SC injection and decreased the risk of i.o/p. injection [12].
In the original trials by Kim et al. [3-5], minimal pressure was applied on the probe, to mimic the pressure applied to low pressure LPEAIs (STBDmin, STMDmin), and higher pressure was applied (about 8 lb or 35 Newtons (N)) (STBDmax, STMDmax) on the probe to mimic the pressure applied to HPEAIs [3-5]. The upper and lower limits for pressure applied to release the needle as reported by the manufacturers of some EAIs are presented in Table 1. At the time of the original trials [3-5], we did not have information on the exact pressure required to trigger the different EAIs brands [7] to release the injection spring, nor the brand-specific variation of pressure to be applied to release the needle. But the 8 lb pressure was expected to reliably trigger the HPEAIs and is the highest pressure needed for the release of Epipen® needles. The minimal pressure on the other hand was very close to the lower limit to trigger the LPEAIs. Furthermore, we had no information on the brand-specific variation in needle length due to inconsistency in production [9]. The variation in needle length accepted for lot release was 5 mm for the HPEAI Epipen®/-EpipenJr®, 2.6 mm for the HPEAI Auvi-Q® and 1.5 mm for the longer needle (0.3 and 0.5 mg epinephrine) and 1.7 mm for the shorter needle (0.15 mg epinephrine) of the LPEAI, Emerade®[9]. High pressure has been shown to exhibit a high risk of i.o./p. injections in small children [4]. To avoid i.o./p. injection in small children, a device with short needle, Auvi-Q 0.1 mg epinephrine, was introduced recently. This device has a low risk of i.o./p. injections but a high risk of SC injections [9].
The ultrasound determinations performed by Kim et al. [3-5] are the basis for our estimations. We use the two pressures on the probe as defined by Kim et al [3-5], and the variation in needle length as declared by the manufacturers of the brand with highest and lowest variation in needle length. In our opinion, it is most important to avoid i.o./p. injection with epinephrine. The aim of this communication to study whether it is possible to find a needle length for EAIs that has no risk of i.o./p injection that consistently results in IM injection.
METHODS
This study, based on previously published ultrasound data on skin to muscle and skin to bone distances using high and minimal pressure on the EAI/probe. The aim was to determine the longest needle length possible to use in EAIs that would not hit the bone and then to investigate the risk of these needle lengths for SC injection.
Food allergic children and adults were consecutively included until approximately 100 patients per age group had been recruited. Four hundred and one (401) consecutive patients with diagnosed food allergy were included in the three original trials [3-5]. As described earlier, 302 children and adolescents (0.2 – 18 years of age, 126 girls and 166 boys) and 99 adults (18 – 72 years of age, 69 women and 32 men) underwent ultrasound investigations using high (8 lb = 35 N) and minimal pressure on the probe. The STBD and STMD at the mid third of the antero-lateral aspect of the right thigh [3-5] were measured. Clinical data and basic statistical analyses have been published [3-5] and the same ultrasound data have been used in previous papers [6,9,12].
There were two groups of children less than 12 years of age: 0-15 kg (n=100) [4] and 15-30 kg (n=102), one group of adolescents >12 years but
The STMD was measured from the skin surface to the outer surface of the facia lata and the STBD from the skin surface to the femur. As in our pevious studies, we used the findings by Diacono et al. [10] who identified that for proper administration of an IM injection, the needle must pass through the fascia and the epimysium into the muscle. This finding was supported by Duvauchelle et al. [11], who published a figure showing the epinephrine bolus staying at the tip of the injection needle (Anapen®, needle length 10.5 mm) after SC injection. The epinephrine remained in the SC space without passing through fascia. Therefore, the full length of the needle’s eye should pass into the muscle tissue for IM delivery. The length of the eye of the EAIs was estimated to be 2 mm [6]. Therefore, the STMD was decreased by 2 mm when estimating the risk for SC injection.
We have previously reported on the variation in needle length and on pressure for lot release, Table 1 [7,9]. In this study we used the shortest and longest possible needle lengths based on the greatest range allowed by manufacturers. The pressures applied in the original study were approximately 35 N, “high pressure” and the minimal pressure applied, “low pressure”.
Outcome variables
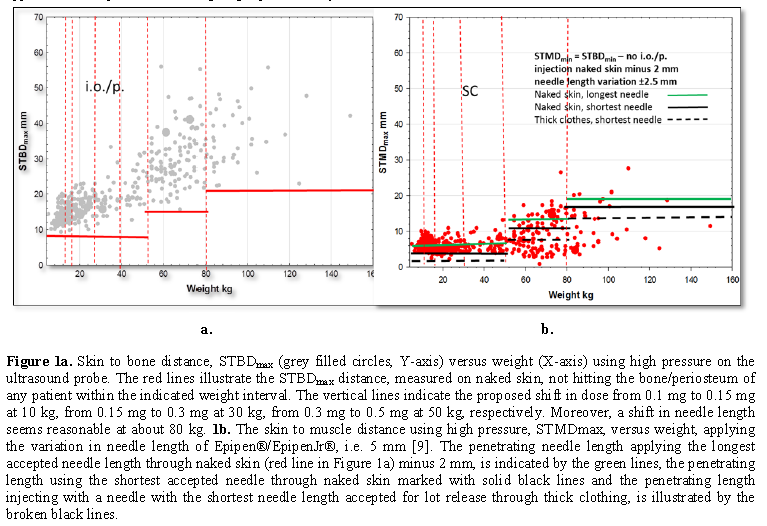
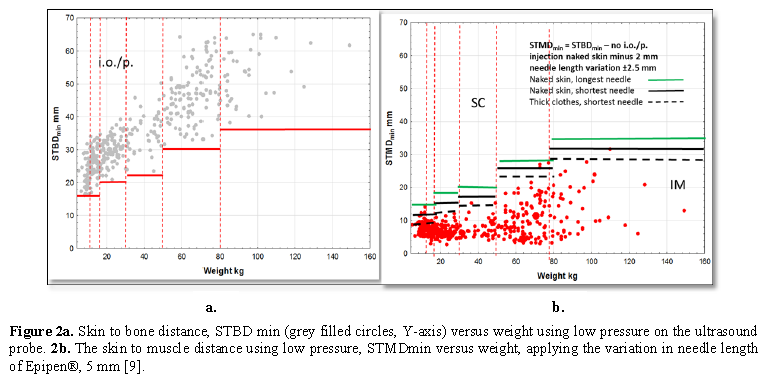
We wanted to identify whether we could find a needle length that caused no i.o./p. injections and at the same time avoided SC injections. We started by estimating the longest needle that would not cause an i.o./p when high pressure was applied to the probe mimicking high pressure injection (Figure 1a for high pressure, STBDmax) and mimicking low pressure (Figure 2a for minimal pressure, STBDmin). In the next step, we identified which needle length would cause a SC injection in the same patients (Figure 1b and 2b, respectively). First, we indicated the longest needle not causing an i.o./p. injection on the STMDmax (Figure 1b) and STMDmin data. (Figure 2b). Furthermore, based on the variation in needle lengths, we indicated the shortest needle when applying the variation in needle length as reported by the manufacturers of Epipen® HPEAI (5 mm variation, Figure 1b) and Emerade® LPEAI (1.5 mm variation, Figure 2b), black lines, in Figures 1b and 2b as well as the calculated needle length when injecting through thick clothing (minus 3 mm), black broken lines. Figures 1b and 2b illustrate the number of subjects receiving a subcutaneous injection The possible risk is illustrated using the accepted maximum variation of needle length by the manufacturers., Table 1.
We estimated compressed thick winter clothing to be 3 mm thick (12). This was based on meassurment of three pairs of winter trousers. Therefore, we decreased needle length by 3 mm when estimating the risks for SC injection wearing thick clothes. After taking the needle eye factor (2 mm) and winter clothing into account, we subtrated 5 mm when calculating the distance for IM injection. The needle length (STBD) and adjusted STMD used for calculation of the risk of i.o./p. and SC injection, respectively, are given in Table 2.
No formal statistical analyses were performed. The figures are intended to qualitatively find needle lengths that would avoid i.o./p. injection and have low risk of SC injection.
RESULTS
The results are illustrated in Figures 1 and 2. The STBDmax is illustrated in Figure 1a, and the STBDmin in Figure 2a (red horizontal lines). They have been constructed to approximately show how long the penetrating part of an EAI needle can be without hitting the bone.
The next step was to describe the influence of needles of the above-mentioned lengths using the brand-specific data on the variation of needle length. Using needles of the lengths in Figures 1a and 2a (minus 2 mm) for injection into the muscle using the variation of needle length as illustrated in Figures 1b for HPEAIs (Epipen®/ Epipen Jr® and Auvi-Q®), and in Figure 3b for, the LPAI: s (Emerade®). The length is reduced by 2 mm to adjust for the fascia findings by Diacono et al. (10).
In Figures 1b and 2b is illustrated the proportion of patients likely to receive a SC injection. Using the variation in needle length accepted by the manufacturer of Epipen®/Epipen Jr®, it appears that even the longest accepted needles would cause SC injection in many patients and the shortest accepted needles would cause SC injection in all patients weighing less than 40 kg wearing winter clothes.
In Figure 2b is illustrated the effect of using low pressure on the optimal needle length not causing any i.o./p. injection, injected with the shortest needle approved and through thick clothing, IM injection will be achieved in most patients.
In summary, starting out with the ideal to not cause any i.o./p. injection, then using the longest needle accepted for lot release for IM injection many patients will get SC injections if using HPEAIs. The LPEAIs will compress the muscle less and have longer needles leading to most patients receiving IM injection.
DISCUSSION
In recent years the risk of SC or i.o./p injection of epinephrine using EAIs has been widely discussed. This paper is based on data from the three original studies completed by Dr. Harold Kim’s group [3-5]. These studies represent the most extensive investigations published on the relationships between the STMD, and the STBD versus weight. Distances were determined by ultrasound at the mid anterolateral aspect of the thigh, the recommended area for IM injection of epinephrine using EAIs [13].
In the original series of studies [3-5], we used 8 pounds or about 35 Newtons (N) as “high pressure” and applied a minimal pressure, referred to as “low pressure”, to mimic the pressure required to release the needle of HPEAIs and LPEAIs, respectively. The declared variation in pressure that is accepted by companies for release of new lots of EAIs has been presented elsewhere [7] and summarized in Table 1. This data was not known by us at the time of the original studies. There are instruments available that can apply a specified pressure to the ultrasound probe [14,15] that should be used in all future trials using ultrasound imaging for the study of STBD and STMD. Also, the pressure applied to the probe was not sufficiently well defined in the original studies. Therefore, we did not present any risk data for any specific EAIs currently available. Instead, we illustrated the principle that by avoiding i.o./p. injection in all patients, by using short enough needles, there would be SC injections occurring frequently when using HPEAIs and less often if using LPEAIs.
Our results suggest that the risk of SC injection is very high if using a needle length that would definitely not cause i.o./p. injection if using high pressure on the probe/auto injector. This risk is reduced when requiring a lower pressure to trigger the auto injector as the lower pressure would allow a longer needle to be used. This occurs because less pressure compressing the muscle will provide a greater STBD and therefore allow the use of longer needles. Then, we believe more patients will receive IM injections. Using this qualitative approach to this important issue, we were able to estimate the needle lengths for EAIs that would deliver the epinephrine to the IM compartment. These findings should be useful in future studies and future EAI designs.
The findings from our study should be confirmed in well-designed prospective studies. With future larger studies, the chance of avoiding i.o./p. injection could be calculated by z-scores for different weight classes, age classes, pubertal stages etc. in children and adolescents and z-scores for weight classes and possibly BMI classes, in adults. Currently, 15 kg and 30 kg are the weight thresholds most often used for EAI prescribing. But we propose the intervals of 10, 50 and 80 kg be used for estimating successful IM injection based on our data.
In closing, if we have the primary goal of avoiding i.o./p injections, an LPEAI design for EAIs is more likely to result in successful IM drug delivery. Therefore, when designing future EAIs or modifying currently available EAIs, we recommend devices that require less force to release the needle and drug. This lower force will allow for the designers to find the proper needle lengths for different weight ranges that will deliver the epinephrine properly.
- Simons FE, Gu X & Simons KJ (2001) Epinephrine absorption in adults: Intramuscular versus subcutaneous injection. J Allergy Clin Immunol 108: 871-873.
- Lockey SD (1980) A new method of administering aqueous epinephrine: the EpiPen, an automatic syringe. J Asthma Res 17: 153-155.
- Tsai G, Kim L, Nevis IF, Dominic A, Potts R, et al. (2014) Auto-injector needle length may be inadequate to deliver epinephrine intramuscularly in women with confirmed food allergy. Allergy Asthma Clin Immunol 10: 39.
- Kim L, Nevis IF, Tsai G, Dominic A, Potts R, et al. (2014) Children under 15 kg with food allergy may be at risk of having epinephrine auto-injectors administered into bone. Allergy Asthma Clin Immunol 10: 40.
- Dreborg S, Wen X, Kim L, Tsai G, Nevis I, et al. (2016) Do epinephrine auto-injectors have an unsuitable needle length in children and adolescents at risk for anaphylaxis from food allergy? Allergy Asthma Clin Immunol 12: 11.
- Dreborg S, Kim L, Tsai G, Kim H (2018) Epinephrine auto-injector needle lengths: Can both subcutaneous and periosteal/intraosseous injection be avoided? Allergy Asthma Clin Immunol 120: 648-653.
- Dreborg S, Kim H (2018) Authors' response. Allergy Asthma Clin Immunol 121: 644-645.
- Dreborg S, Kim H (2019) Tissue compression and epinephrine deposition. J Allergy Clin Immunol Pract 7: 2096-2097.
- Dreborg S, Tsai G, Kim H (2019) Implications of variation of epinephrine auto-injector needle length. Allergy Asthma Clin Immunol 123: 89-94.
- Diacono D, Pumphrey RS, Sharma V, Arkwright PD (2015) The deep fascia of the thigh forms an impenetrable barrier to fluid injected subcutaneously by auto injectors. J Allergy Clin Immunol Pract 3: 297-299.
- Duvauchelle T, Robert P, Donazzolo Y, Loyau S, Orlandini B, et al. (2018) Bioavailability and cardiovascular effects of adrenaline administered by anapen auto injector in healthy volunteers. J Allergy Clin Immunol Pract 6: 1257-1263.
- Dreborg S, Tsai G, Kim H (2020) Epinephrine auto-injector needle length: The impact of winter clothing. Allergy Asthma Clin Immunol 16: 24.
- Simons FE, Ardusso LR, Bilo MB, Cardona V, Ebisawa M, et al. (2014) International consensus on (ICON) anaphylaxis. World Allergy Organ J 7: 1-19.
- Andersen HH, Lundgaard AC, Petersen AS, Hauberg LE, Sharma N, et al. (2016) The lancet weight determines wheal diameter in response to skin prick testing with histamine. PLoS One 11: e0156211.
- Kim H, Dinakar C, McInnis P, Rudin D, Benain X, et al. (2017) Inadequacy of current pediatric epinephrine autoinjector needle length for use in infants and toddlers. Ann Allergy Asthma Immunol 118: 719-725.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Ageing and Restorative Medicine (ISSN:2637-7403)
- International Journal of Medical and Clinical Imaging (ISSN:2573-1084)
- Journal of Psychiatry and Psychology Research (ISSN:2640-6136)
- Journal of Pathology and Toxicology Research
- International Journal of Diabetes (ISSN: 2644-3031)
- Advance Research on Alzheimers and Parkinsons Disease
- International Journal of Radiography Imaging & Radiation Therapy (ISSN:2642-0392)