3912
Views & Citations2912
Likes & Shares
The improvement of phase and frequency coordination through CDT substantially contributes to the repair in cerebrum and cerebellum injury, spinal cord injury (SCI), cerebral palsy, stroke, myelomeningocele, aging and cancer. Even sportsmen benefit from an improved phase and frequency among neuron firing, because the coordination of arms, legs and trunk improve, to play for example better tennis or football. Since CDT improves the functioning of every CNS, and the CNS is involved in nearly all body functions, the regulations of body functions improve, especially the cardio-vascular performance, to live longer with a better quality of life through 10 to 15 h CDT per week.
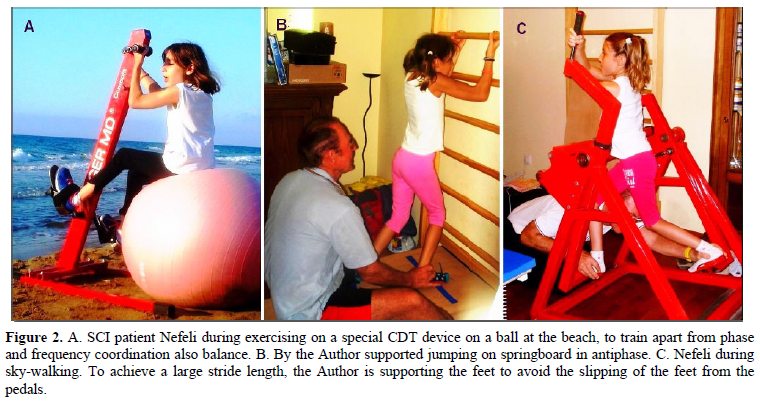
At an age of 9, CDT was started for 4 years. The performed movements included exercising on a special CDT device (Figure 2A), supported jumping on springboard (Figure 2B) and sky-walking (Figure 2C). Further, creeping, crawling, walking and other movements were trained.
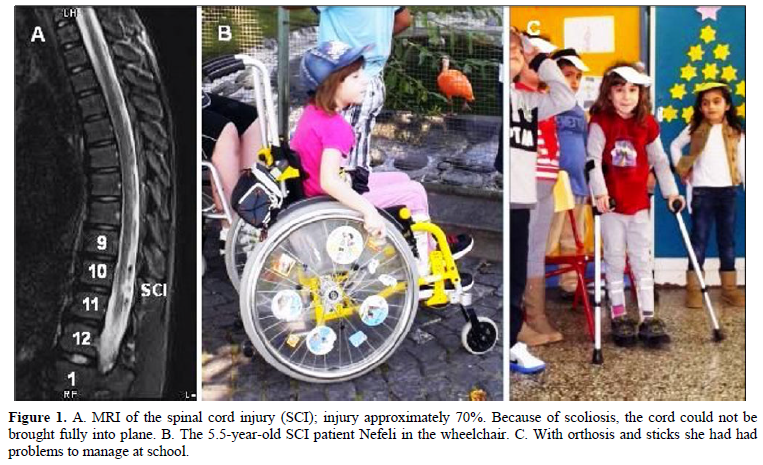
Nefeli relearned walking (with deficits) so that she could walk at school to the whiteboard (Figure 3B) and write there (Figure 3A). The achieved spinal cord repair, including a partial regeneration of the spinal cord [3], allowed her to manage quite well at school, which would have been difficult without CDT (Figure 1C). The urinary bladder functions were mainly repaired through learning transfer [2] from exercising on the special CDT device and jumping on springboard. A bladder repair is most important for patients with SCI. Even a bit of normal cycling she learned (Figure 3C) first time in her life. The feet control was the main problem and not the balance. For details of SCI repair in general and this special SCI repair see [3]. In the Discussion it will be shown that the out-of-date ‘Human Repair-Neurophysiology’ of universities may become a danger to CNS injured patients.
The single-nerve fiber action potential recording method, the surface electromyography and the coordination dynamics recording method are used to analyze premotor spinal oscillators and the phase relations among them in brain-dead humans (HTs) and patients with Parkinson’s disease. Based on the difference between physiologic phase and frequency coordination in HT’s and pathologic coordination in patients, the repair method CDT is developed and used in connection with the plasticity of the human CNS to repair the human nervous system.
METHOD
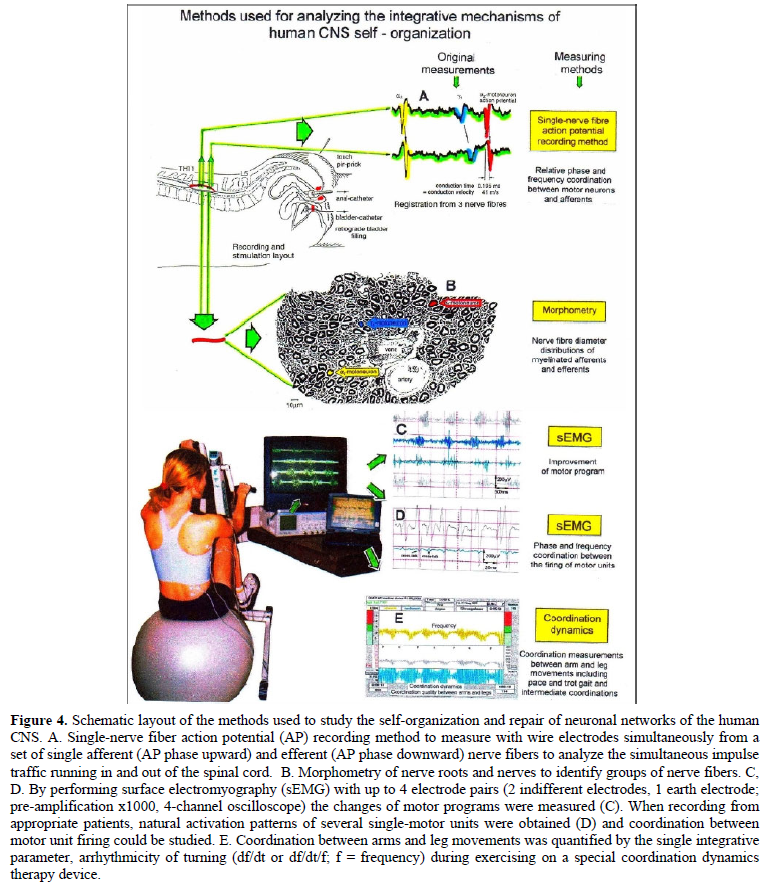
For analyzing phase and frequency coordination and its improvement or repair, mainly four methods were used (Figure 4): The single-nerve fiber action potential recording method (A) [4], the morphometry (B), the surface electromyography (sEMG) (C, D) and the coordination dynamics recording method (E) [5]. The single-nerve fiber action potential recording method and the morphometry were used to classify and identify human peripheral nerve fibers and measure and follow up phase and frequency coordination among a set of single neurons. Surface EMG was used to record the coordinated firing of single motor units and the coordination dynamics recording method was used to measure CNS repair of patients during treatment progress by a single value.
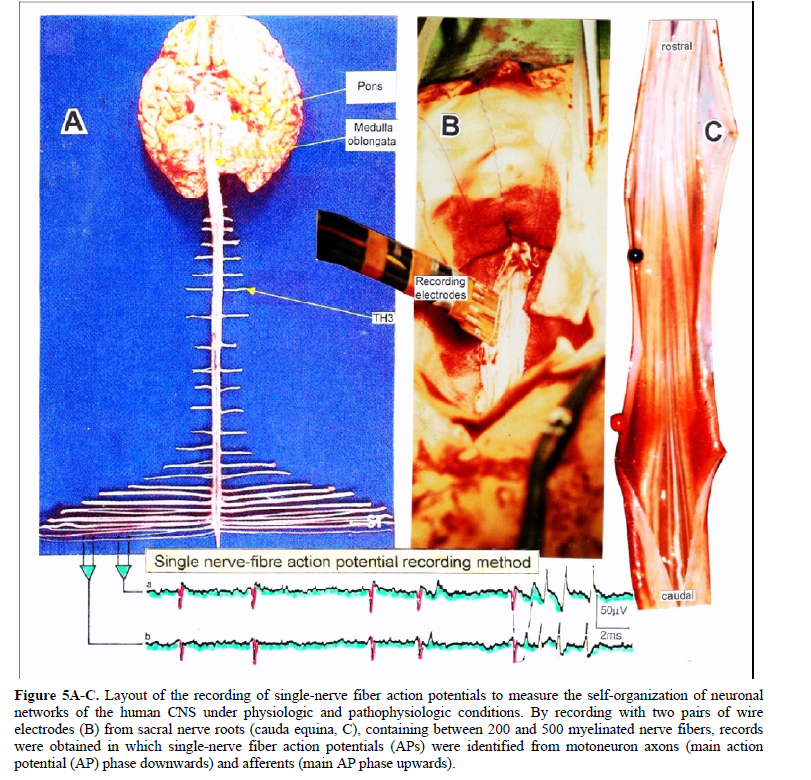
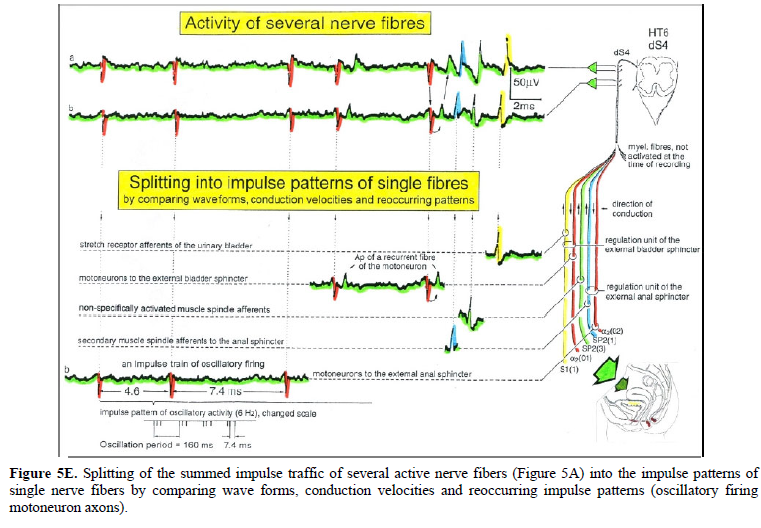
By recording with two pairs of platinum wire electrodes from sacral nerve roots containing between 200 and 500 myelinated nerve fibers (of which a few ones are only active), records were obtained in which single-nerve fiber action potentials (APs) could be identified from motoneuron axons (main AP phase downwards) and afferents (main AP phase upwards) (Figure 5A-5C). By measuring the conduction times and with the known electrode pair distance (10 mm) conduction velocities could be calculated.
By comparing wave forms, conduction times/velocities and reoccurring action potential patterns, the summed impulse traffic of several active nerve fibers could be splitted into the impulse patterns of single nerve fibers (Figures 5D and 5E).
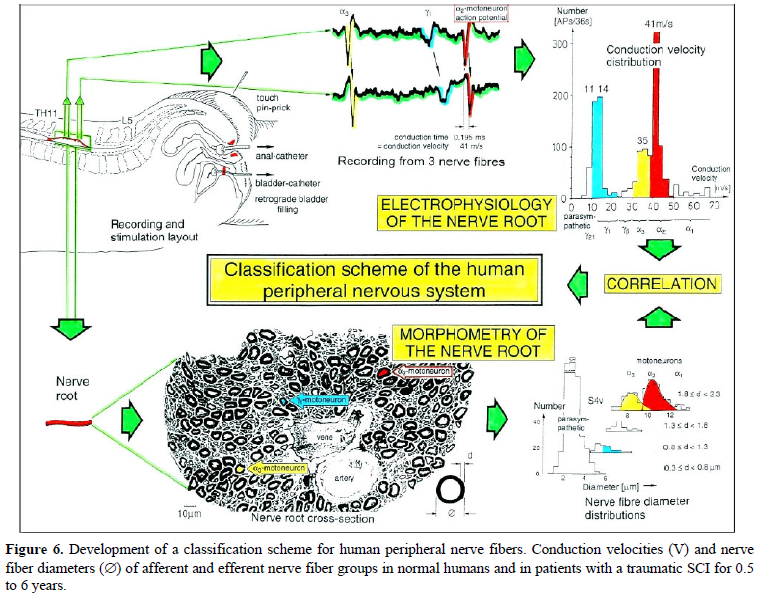
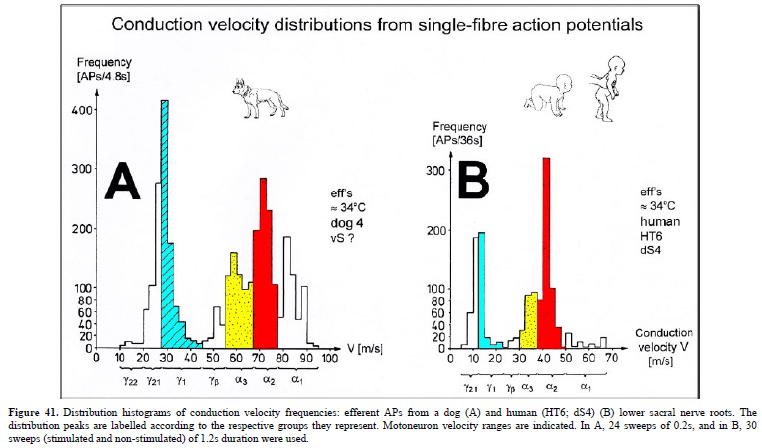
Conduction velocity distribution histograms were constructed in which the myelinated nerve fiber groups larger than 4mm could be characterized by group conduction velocity value. Recording was followed by morphometry. Distributions of nerve fiber diameters were constructed and nerve fiber groups were characterized by the peak values of asymmetrical distributions [6,7].
By correlating the peak values of the velocity distributions with those of the diameter distributions obtained for the same root, a classification scheme was constructed of the human peripheral nervous system (Figures 6 and 7) [7-9]. The group conduction velocities and group nerve fiber diameters had the following pair-values at 35.5°C:
Spindle afferents: SP1(65ms-1/13.1µm), SP2(51/12.1); touch afferents: T1(47/11.1), T2(39/10.1), T3(27/9.1), T4(19/8.1); urinary bladder afferents: S1(41ms-1/-), ST (35/-); a-motoneurons: a13 (-/14.4), a12 (65ms-1/13.1µm), a11 (60?/12.1)[FF], a2 (51/10.3)[FR], a3 (41/8.2)[S]; g-motoneurons: gb(27/7.1), g1(21/6.6), g21(16/5.8), g22(14/5.1); preganglionic parasympathetic motoneurons: (10ms-1/3.7µm). Schematically the values are summarized in Figure 7.
With respect to electrical stimulation, it was found that the primary spindle afferents likely have the lowest threshold upon electrical nerve root stimulation, followed by α1-motoneurons (FF), secondary muscle spindle afferents, α2-motoneurons (FR), α3-motoneurons (S), gb, g1 (dynamic), g21 (static), g22 (static), and parasympathetic motoneurons.
This classification and identification scheme represents a solid basis for classifying and identifying nerve fiber groups in the human peripheral nervous system (PNS) and analyzing central nervous system (CNS) functions at the single-neuron level, even though it is incomplete and holds so far only for nerve fiber diameters larger than approximately 3.5µm.
With this classification and identification scheme it became possible to record natural impulse patterns simultaneously from identified single afferent and efferent nerve fibers and analyze the self-organizing mechanism “phase and frequency coordination” of the human CNS under physiologic and pathologic conditions and find therapy to repair the human CNS.
The drawing back of the single-nerve fiber action potential recording method is that it is an invasive recording method. But with surface electromyography (sEMG) one can record non-invasively coordination among motoneurons via the motor units if one records from suitable patients, like incomplete spinal cord injury patients, when a certain muscle is only still innervated by a few motoneurons. With the sEMG the tremor of patients with Parkinson’s disease will be analyzed.
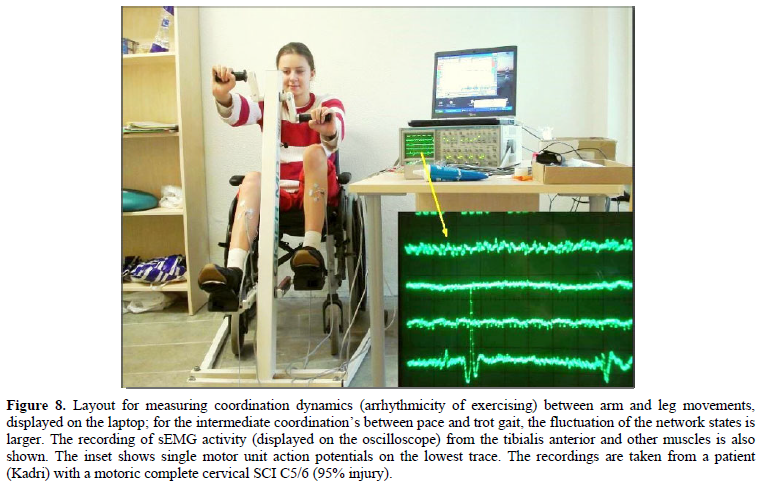
The improvement of CNS functioning will be measured with the coordination dynamics values. With a single value the organization of the human CNS is characterized. Figure 8 shows a spinal cord injury patient during exercising on the special CDT device in the sitting position. The arrhythmicity of exercising, the coordination dynamics, of the patient is measured. Simultaneously sEMG is performed.
For treatment, especially for severely injured patients, the exercising on a special CDT in the lying position is suitable Figures 8 and 9)
Figure 8. Layout for measuring coordination dynamics (arrhythmicity of exercising) between arm and leg movements, displayed on the laptop; for the intermediate coordination’s between pace and trot gait, the fluctuation of the network states is larger. The recording of sEMG activity (displayed on the oscilloscope) from the tibialis anterior and other muscles is also shown. The inset shows single motor unit action potentials on the lowest trace. The recordings are taken from a patient (Kadri) with a motoric complete cervical SCI C5/6 (95% injury).
Figure 9. The 11-year-old Nefeli with an incomplete SCI during exercising coordinated arm, leg and trunk movements to improve the coordinated firing of neurons and sub-neural networks. This special CDT device for measuring and therapy (int.pat.) is produced by the firm: Giger Engineering, Martin Giger dipl.Ing.ETH/SIA, Herrenweg 1, 4500 Solothurn, Switzerland, www.g-medicals.ch.
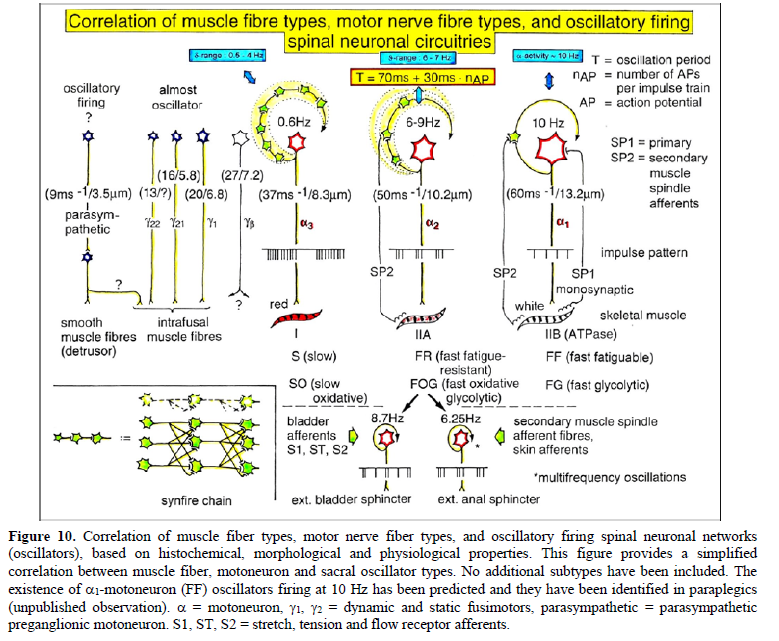
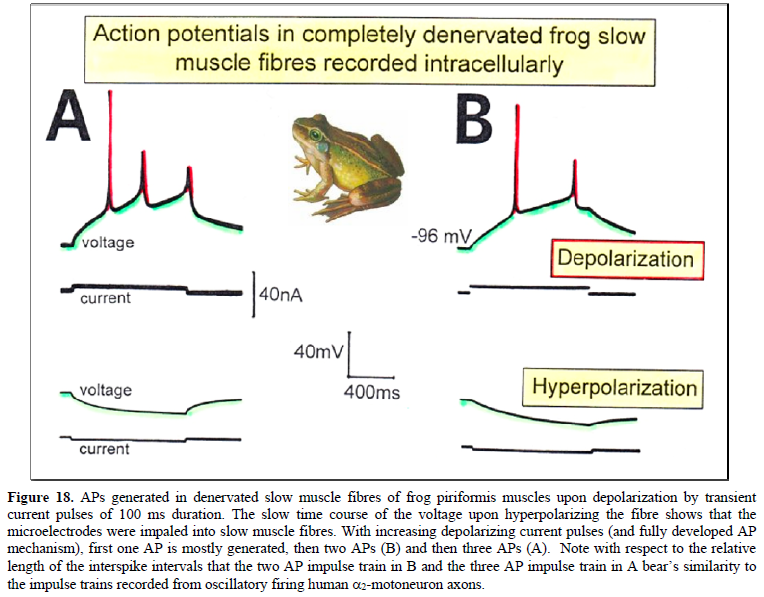
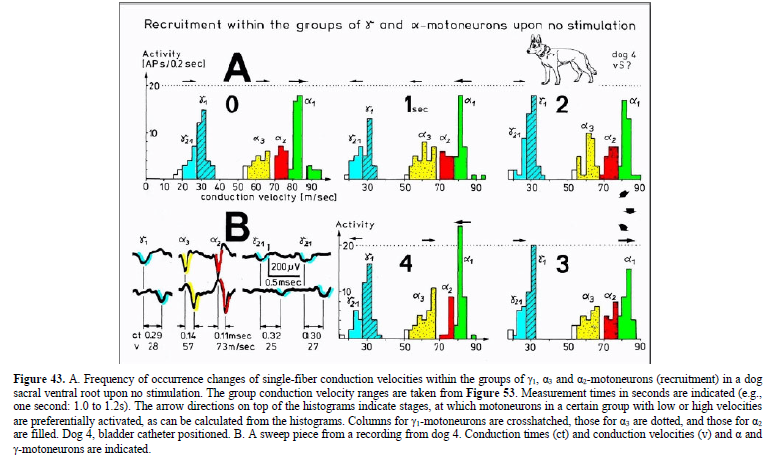
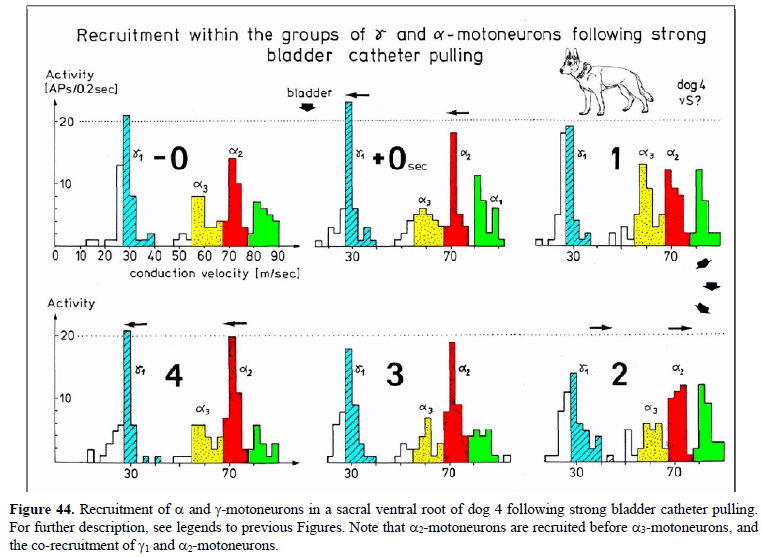
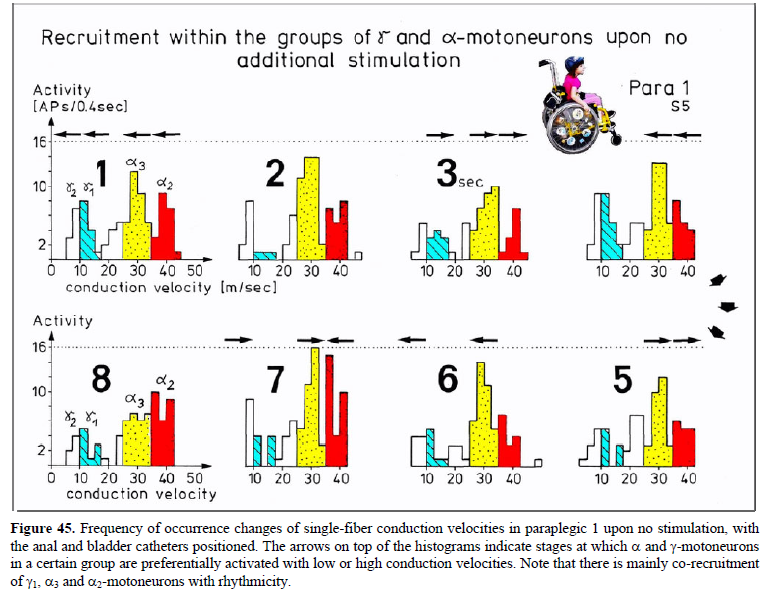
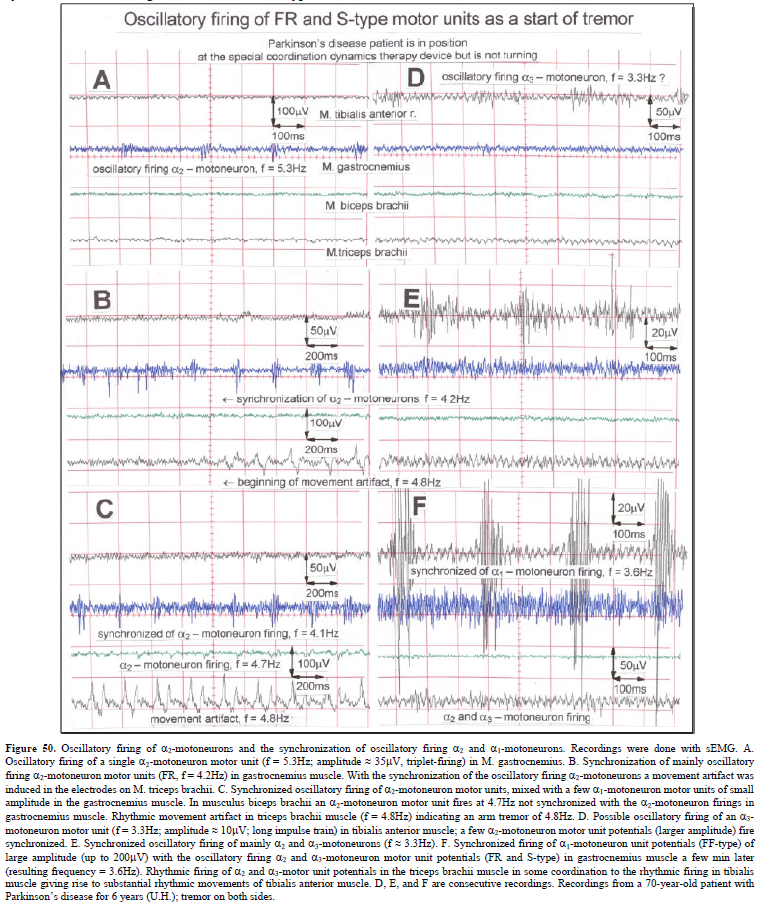
RESULTS OSCILLATORY FIRING OF HUMAN MOTONEURONS FOR HIGH ACTIVATION Oscillatory Firing of α1, α2, and α3-Motoneurons The most important finding with the single-nerve fiber action potential recording method is that nerve cells in the human CNS are organizing themselves through phase and frequency coordination. In nerve fibers, this phase and frequency coordination can easily be measured, because the three motoneuron types fire for high activation oscillatory [10-13] and offer in this way a structure to which the timed firing of neurons can be related to. Since the α2-motoneuron oscillations are most stable, firing phases of neurons will be related mostly to the α2- motoneuron firings. It is started with the oscillatory firing of the three motoneuron types. Figure 10 shows schematically the oscillatory firing patterns of the three kinds of motoneurons and the muscle fiber types they innervate. The differences between the three premotor oscillator types will be considered in more detail.
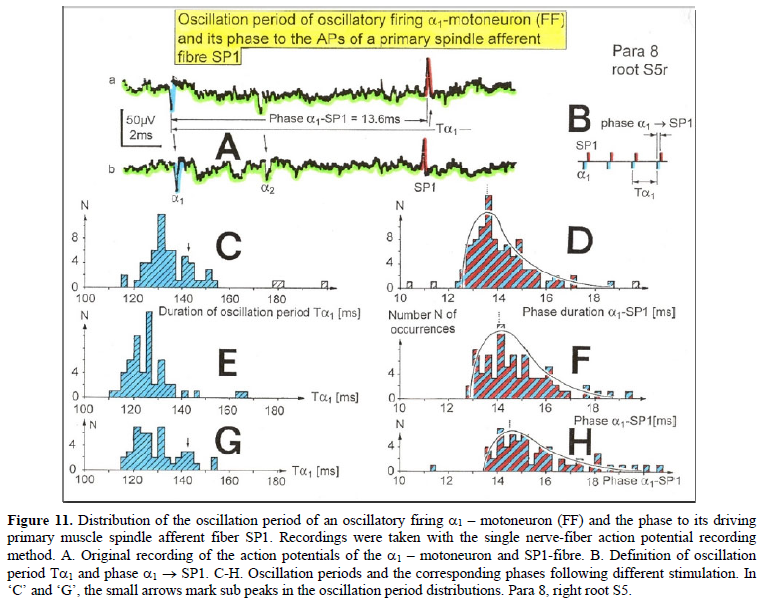
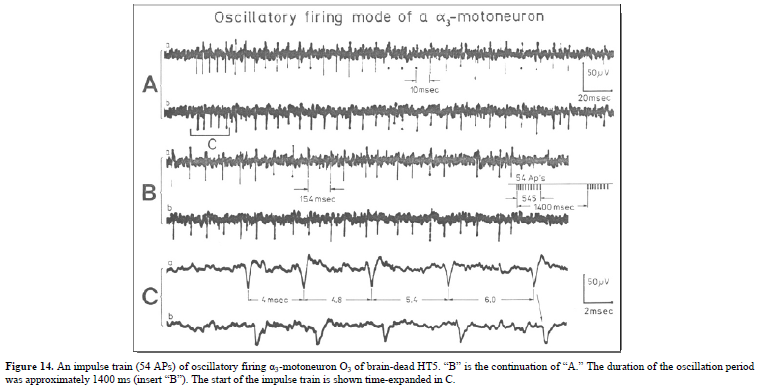
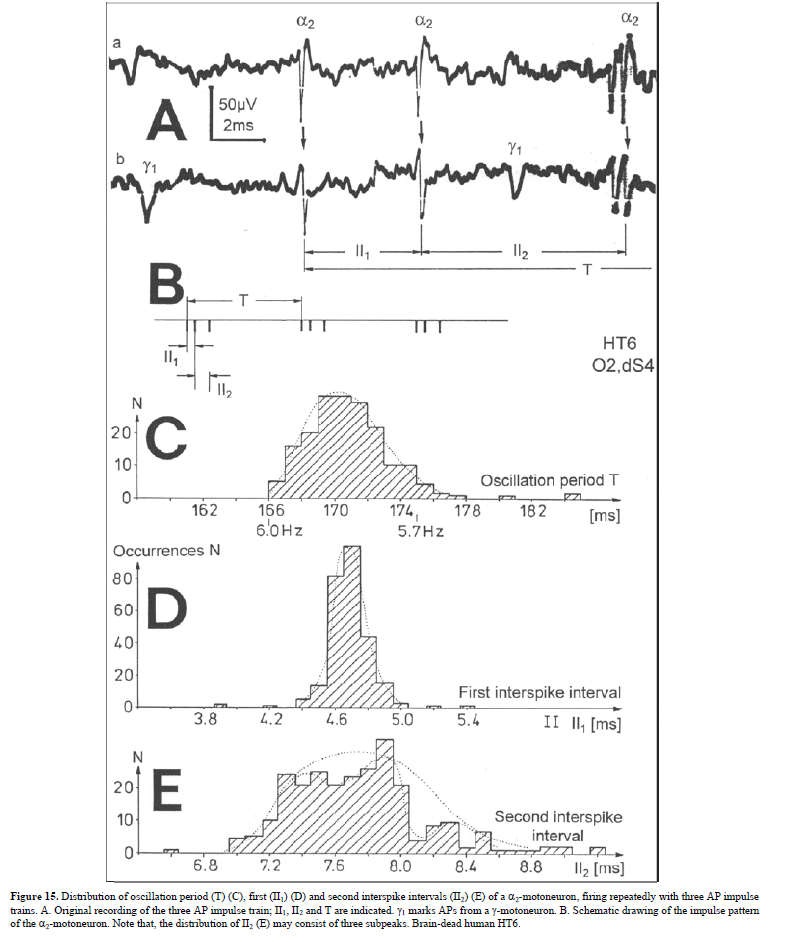
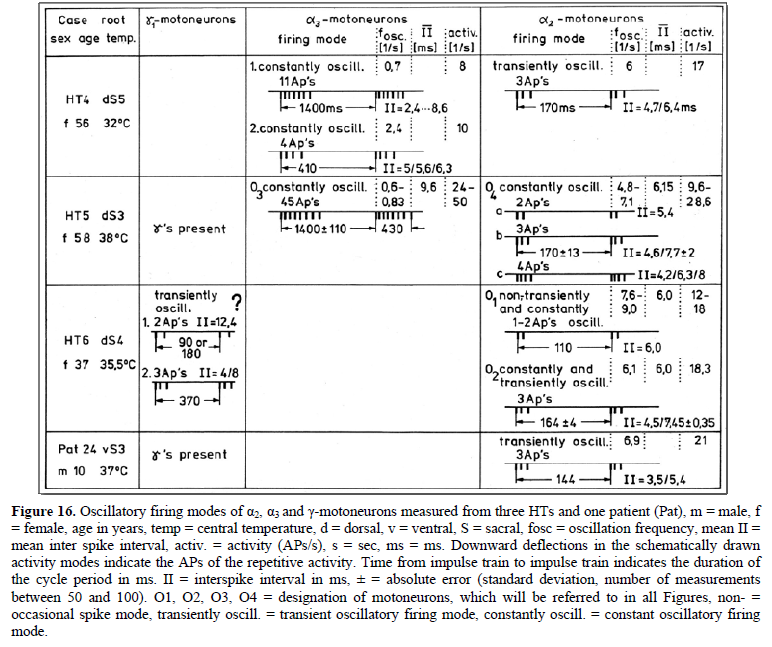
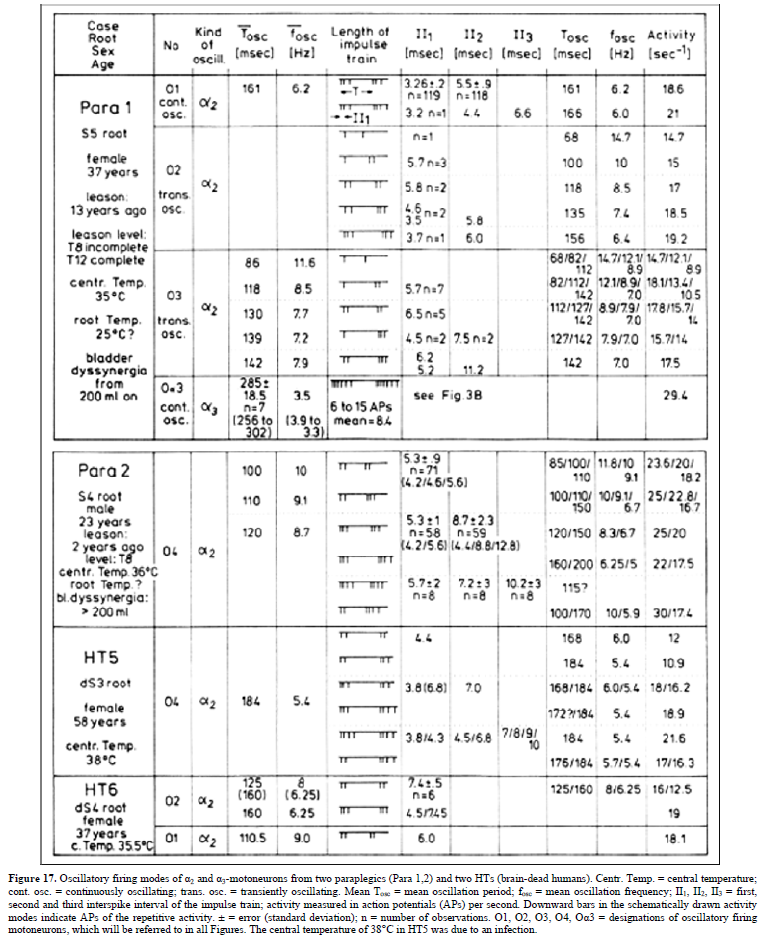
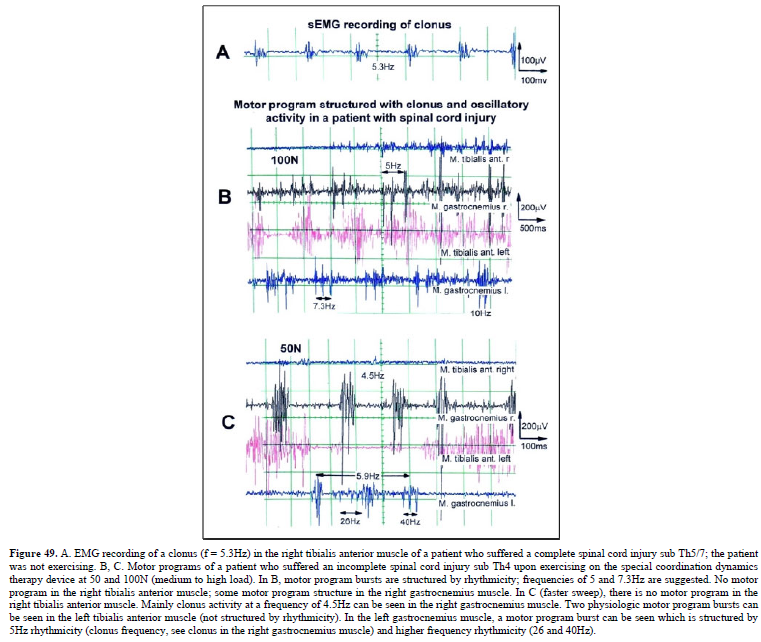
Figure 10. Correlation of muscle fiber types, motor nerve fiber types, and oscillatory firing spinal neuronal networks (oscillators), based on histochemical, morphological and physiological properties. This figure provides a simplified correlation between muscle fiber, motoneuron and sacral oscillator types. No additional subtypes have been included. The existence of 1-motoneuron (FF) oscillators firing at 10 Hz has been predicted and they have been identified in paraplegics (unpublished observation). = motoneuron, 1, 2 = dynamic and static fusimotors, parasympathetic = parasympathetic preganglionic motoneuron. S1, ST, S2 = stretch, tension and flow receptor afferents. α1-oscillators: The 1-motoneurons fire oscillatory with one action potential at 10 Hz and higher and innervate fast fatigue (FF) muscle fibers. The stability of oscillatory firing is low, and they are driven by primary muscle spindle afferents. Figure 11 shows an original recording of an 1-motoneuron action potential (AP), activated by aFigure 11. Distribution of the oscillation period of an oscillatory firing 1 – motoneuron (FF) and the phase to its driving primary muscle spindle afferent fiber SP1. Recordings were taken with the single nerve-fiber action potential recording method. A. Original recording of the action potentials of the 1 – motoneuron and SP1-fibre. B. Definition of oscillation period T1 and phase 1 SP1. C-H. Oscillation periods and the corresponding phases following different stimulation. In ‘C’ and ‘G’, the small arrows mark sub peaks in the oscillation period distributions. Para 8, right root S5. α2-oscillators: The 2-motoneurons fire oscillatory with an action potential train of 2 to 4 APs at a frequency around 6 Hz and innervate fatigue resistant muscle fibers (FR). An original recording of oscillatory firing is shown in Figure 12A. It could be shown for a sphincteric urinary bladder 2-motoneuron that with bladder filling the 2- motoneurons fired occasional, transient oscillatory and continuous oscillatory Figure 12D). The characteristics of 2-oscillators are in between those of the 1 and the 3- oscillators. They are self-organized by the adequate afferent input patterns from several kinds of receptors. During oscillatory firing the 2-motoneurons showed relative phase correlation to the driving secondary muscle spindle afferents and bladder afferents (Figures 12B and 13). α3-oscillators: An original recording from an oscillatory firing α3-motoneuron is shown in Figure 14. The insert “C” shows a schematic layout of the firing. The duration of the interspike intervals of the impulse train increases regularly at the beginning of the impulse train and does alternate between long and short values at the end of the impulse train. This long-short alternation of interspike intervals may be due to a coupling phenomenon between oscillators. Alternating firing with short and long oscillation periods has also been observed for α2- oscillators and may be connected with the existence of antagonistic inhibition of network structures similar to half-center oscillators. Regular Oscillatory Firing of 2-Motoneurons in Brain-Dead Human Figure 15 shows an impulse train triplet of an oscillatory firing 2-motoneuron (O2) of the brain-dead individual HT6 [14] to secure bowel continence. The firing pattern is schematically drawn in Figure 15B. Distributions of the oscillation period and of the first and the second interspike intervals are plotted in Figure 15C-15E. SciTech Central Inc. Int J Med Clin Imaging (IJMCI) 335 Int J Med Clin Imaging, 7(2): 324-399 Schalow G Figure 12. Self-organization of premotor spinal α2-oscillator O1, which innervates the external urinary bladder sphincter (skeletal muscle). Brain-dead human HT6; recording from a dorsal S4 nerve root. A. Recordings from α2-motoneurons O1 and O2, firing in the oscillatory mode with impulse trains of 2 (upper recording) and 3 (lower recording) action potentials (APs). The durations of the oscillation periods were 110 (O1) and 164ms (O2). The interspike intervals of the impulse trains were 5.9ms (O1) and 4.6 and 7.4ms (O2). Motoneuron O1 conducted at 36 m/s; its recurrent fiber conducted at 21 m/s. The measurement layout is shown schematically. The inserts show the oscillatory firing modes; they have not been drawn to scale. B. Impulse patterns of oscillatory firing α2-motoneuron O2 innervating the external anal sphincter, in relation to the muscle spindle afferent activity SP2(1 to 3), activated by the stretch of the anal sphincter by the anal catheter, and impulse patterns of oscillatory firing α2-motoneuron O1 innervating the external urethral sphincter, in relation to the stretch receptor afferent activity (S1(1)) of the urinary bladder, activated by 750 ml bladder filling. Phase relations between APs of SP2(2) and O2 and between APs of S1(1) and O1 are indicated by the small arrows. C. Three series of successive interspike intervals of the 2-stretch receptor afferent fibers S1(1) and S1(2) activated by retrograde bladder filling. The oscillation period of oscillatory firing motoneuron O1, activated only by bladder filling is shown. D. The firing in the occasional spike mode, the transient and the constant oscillatory firing mode of α2-motoneuron O1 in response to filling of the bladder. In the ‘activity pattern’ column changing durations of oscillation periods are given. The oscillation frequencies in the brackets give the frequencies at the moment of oscillation for the transient oscillatory mode. Downward deflections are schematized APs. Interspike intervals of the close APs ≈ 6.0ms (A). E. Activity levels of stretch (S1) and tension (ST) and flow receptor afferents (S2) (E) and of sphincter α2-motoneuron O1 (F) in response to retrograde filling of the bladder. The activity values of the S1, ST and S2 afferents are taken from histograms like the one in G. Filling of the bladder was stopped once between 600- and 650-ml. F. The small dotted lines represent mean activity (APs/s) and oscillation frequency (impulse trains/s) of α2-motoneuron O1 if bladder filling were not stopped in between. Note that the mean activity increases continuously with the filling of the bladder from 550 to 650 ml, even though motoneuron O1 started to fire in the oscillatory mode from 620 ml on (D). Note further that the oscillatory firing motoneuron O2 (frequency of firing with impulse trains is shown) is nearly not affected by the filling of the bladder and by the start of the oscillatory firing of motoneuron O1. G. Conduction velocity frequency distribution histogram of stretch, tension and flow receptor afferent activity at 750 ml. The activities of afferents S1, ST and S2 are quantified by counting the afferent conduction velocities under the peaks (open plus hatched part), with the conduction velocity limits given in the insert. The counts (27, 33, 59) are given below the peak labeled S1, ST and S2 and plotted into E for the afferent activity at 750 ml. H. Schematic drawing of the anatomical arrangement of the afferents and the motoneuron O1. SciTech Central Inc. Int J Med Clin Imaging (IJMCI) 336 Int J Med Clin Imaging, 7(2): 324-399 Schalow G Figure 13. Absolute and relative correlation quantified by phase relations between the α1 (FF) and α2-motoneurons (FR) and their driving primary (SP1) and secondary (SP2) muscle spindle afferent fibers. A, B. Definition of the different phases. C. Distribution of the phases between α1-motoneuron and the secondary muscle spindle afferent fiber SP2(1). Note that the phase distribution α1 SP2(1) is approx. 40 times wider than that of the α1 SP1 distribution (phase α1 SP1 taken from Fig.65). D. Distribution of the phases between α2-motoneuron and the secondary muscle spindle afferent fiber SP2(2). Note that the phase distribution α2 SP2(2) is similar to that of α1 SP2(1) (approx. 4 times wider). E, F. Note that every SP1-action potential is accompanied by a time-locked α1-motoneuron action potential (AP). Para 8, root S5r. Figure 14. An impulse train (54 APs) of oscillatory firing α3-motoneuron O3 of brain-dead HT5. “B” is the continuation of “A.” The duration of the oscillation period was approximately 1400 ms (insert “B”). The start of the impulse train is shown time-expanded in C. SciTech Central Inc. Int J Med Clin Imaging (IJMCI) 337 Int J Med Clin Imaging, 7(2): 324-399 Schalow G Figure 15. Distribution of oscillation period (T) (C), first (II1) (D) and second interspike intervals (II2) (E) of a 2-motoneuron, firing repeatedly with three AP impulse trains. A. Original recording of the three AP impulse train; II1, II2 and T are indicated. 1 marks APs from a -motoneuron. B. Schematic drawing of the impulse pattern of the 2-motoneuron. Note that, the distribution of II2 (E) may consist of three subpeaks. Brain-dead human HT6. SciTech Central Inc. Int J Med Clin Imaging (IJMCI) 338 Int J Med Clin Imaging, 7(2): 324-399 Schalow G The distributions of the oscillation period and of the first interspike interval are smooth and show no obvious subpeaks. The distribution of the second II shows subpeaks suggesting interactions with other oscillatory firing circuitries. The narrow distribution of the oscillation period, and the interspike intervals of the impulse train show that the oscillatory firing network fired very regularly. Other spinal oscillators in brain-dead individuals fired just not as regularly as the one shown in Figure 15. It is concluded that in brain-dead and normal individuals, the neuronal networks, driving the motoneurons, fire regularly with some variation. The impulse patters show little variation, are stable and can easily identified (Figure 16). It will be shown below that this regularity is lost following spinal cord injury. Figure 16. Oscillatory firing modes of α2, α3 and γ-motoneurons measured from three HTs and one patient (Pat), m = male, f = female, age in years, temp = central temperature, d = dorsal, v = ventral, S = sacral, fosc = oscillation frequency, mean II = mean inter spike interval, activ. = activity (APs/s), s = sec, ms = ms. Downward deflections in the schematically drawn activity modes indicate the APs of the repetitive activity. Time from impulse train to impulse train indicates the duration of the cycle period in ms. II = interspike interval in ms, ± = absolute error (standard deviation, number of measurements between 50 and 100). O1, O2, O3, O4 = designation of motoneurons, which will be referred to in all Figures, non- = occasional spike mode, transiently oscill. = transient oscillatory firing mode, constantly oscill. = constant oscillatory firing mode. Irregular Oscillatory Firing of 2-Motoneurons Following Spinal Cord Injury Following spinal cord injury, mean values of oscillation period and interspike intervals for oscillatory firing motoneurons show much more variation (Figure 17) than do normal values (HTs, Figure 16) [10,14]. The most regular oscillatory firing 2-motoneuron in a paraplegic patient fired nearly as regularly as the 2- motoneuron of the brain-dead individual HT6 (Figure 15). On the other hand, some oscillatory firing 2- motoneurons of paraplegics fired extremely irregularly so that it was difficult or even impossible to identify the kind of the motoneuron from the impulse patterns (Figure 17). The distributions of the oscillation period and interspike intervals of the impulse train from an 2-motoneuron in paraplegic 8 showed more scatter than those obtained for HT6 (Figure 15). For further, see Chapter III of [7].
-
Schalow G (2005) Phase and frequency coordination between neuron firing as an integrative mechanism of human CNS self-organization. Electromyogr Clin Neurophysiol 45: 369-383.
-
Schalow G (2010) Scientific basis for learning transfer from movements to urinary bladder functions for bladder repair in patients with spinal cord injury. Electromyogr Clin Neurophysiol 50: 339-395.
-
Schalow G (2019) Regeneration of the human spinal cord via coordination dynamics therapy. Peertechz Publications. eBook. pp: 97.
-
Schalow G, Lang G (1987) Recording of Single Unit Potentials in Human Spinal Nerve Roots: A New Diagnostic Tool. Acta Neurochir 86: 25-29.
-
Schalow G (2006) Surface EMG- and coordination dynamics measurements-assisted cerebellar diagnosis in a patient with cerebellar injury. Electromyogr Clin Neurophysiol 46: 371-384.
-
Wattig B, Schalow G, Heydenreich F, Warzok R, Cervos-Navarro J (1992) Nucleotides enhance nerve fiber regeneration after peripheral nerve crush damage - Electrophysiologic and morphometric investigation. Arzneimittel-forschung 42(9): 1075-1078.
-
Schalow G (2013) Human Neurophysiology: Development and Repair of the Human Central Nervous System. Nova Science Publishers, Inc, Hauppauge NY, USA. pp: 734.
-
Schalow G (2015) Repair of the Human Brain and Spinal Cord. Nova Science Publishers, Inc, Hauppauge NY, USA. pp: 525.
-
Schalow G (2015) Neural network learning in human. Nova Science Publishers, Inc, Hauppauge NY, USA. pp: 324.
-
Schalow G (1991) Oscillatory firing of single human sphincteric a2 and a3-motoneurons reflexly activated for the continence of urinary bladder and rectum. Restoration of bladder function in paraplegia. Electromyogr Clin Neurophysiol 31: 323-355.
-
Schalow G (1993) Action potential patterns of intrafusal g and parasympathetic motoneurons, secondary muscle spindle afferents and an oscillatory firing a2-motoneuron, and the phase relations among them in humans. Electromyogr Clin Neurophysiol 33: 477-503.
-
Schalow G, Zäch GA (1996) Mono and polysynaptic drive of oscillatory firing a1 (FF) and a2-motoneurons (FR) in a patient with spinal cord lesion. Gen Physiol Biophys 15: 57-74.
-
Schalow G, Bersch U, Zäch GA, Warzock R (1996) Classification, oscillatory and alternating oscillatory firing of a1 (FF) and a2-motoneurons (FR) in patients with spinal cord lesion. Gen Physiol Biophys 15: 5-56.
-
Schalow G (1993) Spinal oscillators in man under normal and pathologic conditions. Electromyogr Clin Neurophysiol 33: 409-426.
-
Schalow G (2009) The classification and identification of human somatic and parasympathetic nerve fibers including urinary bladder afferents is preserved following spinal cord injury. Electromyogr Clin Neurophysiol 49: 263-286.
-
Hess WR (1981) Biological order and human society. In: Akert, K. (Ed.), Biological Order and Brain Organization - Selected Works of W.R. Hess, Springer-Verlag, Berlin. pp: 3-15.
-
Schalow G, Zäch GA (1996) Reflex stimulation of continuously oscillatory firing a and g-motoneurons in patients with spinal cord lesion. Gen Physiol Biophys 15: 75-93.
-
Schalow G (2009) Partial cure achieved in a patient with near-complete cervical spinal cord injury (95% injury) after 3 years of coordination dynamics therapy. Electromyogr Clin Neurophysiol 49: 199-221.
-
Schöner G, Zanone PG, Kelso JA (1992) Learning as change of coordination dynamics: Theory and experiment. J Mot Behav 24: 29-48.
-
Schalow G (2019) Brain repair and general health improvement through human neurophysiology and repair physiology (Review of Coordination Dynamics Therapy (CDT)). Clin Med Rep 2(2): 1-68.
-
Sperry RW (1945) The problem of central nervous system reorganization and muscle transposition. Quart Rev Biol 20: 311-369.
-
Sperry RW (1947) Effect of crossing nerves to antagonistic limb muscles in the monkey. Arch Neurol Psychiat (Chicago) 58: 452-473.
-
Weiss P, Brown PF (1941) Electromyographic study on coordination of leg movements in poliomyelitis patients with transposed tendons. Proc Soc Exper Biol Med 48: 384-387.
-
Schalow G, Zäch GA (2000) Reorganization of the Human CNS, Neurophysiologic measurements on the coordination dynamics of the lesioned human brain and spinal cord. Theory for modern neurorehabilitation (31 case reports). Gen Physiol Biophys 19: 1-244.
-
Boyd IA (1980) The isolated muscle spindle. Trends in Neurosciences 3: 258-265.
-
Gladden MH (1985) Efferent control of human muscle spindles. In: Boyd, I.A. and Gladden, M.H. (Eds.), The Muscle Spindle, Stockton Press, New York. pp: 243-254.
-
Schalow G (1992) Recruitment within the groups of g1, a2 and a3-motoneurons in dogs and humans following bladder and anal catheter pulling. Gen Physiol Biophys 11: 101-121.
-
Bawa B, Binder MD, Ruenzel P, Henneman E (1984) Recruitment of motoneurons in stretch reflexes is highly correlated with their axonal conduction velocity. J Neurophysiol 52: 410-420.
-
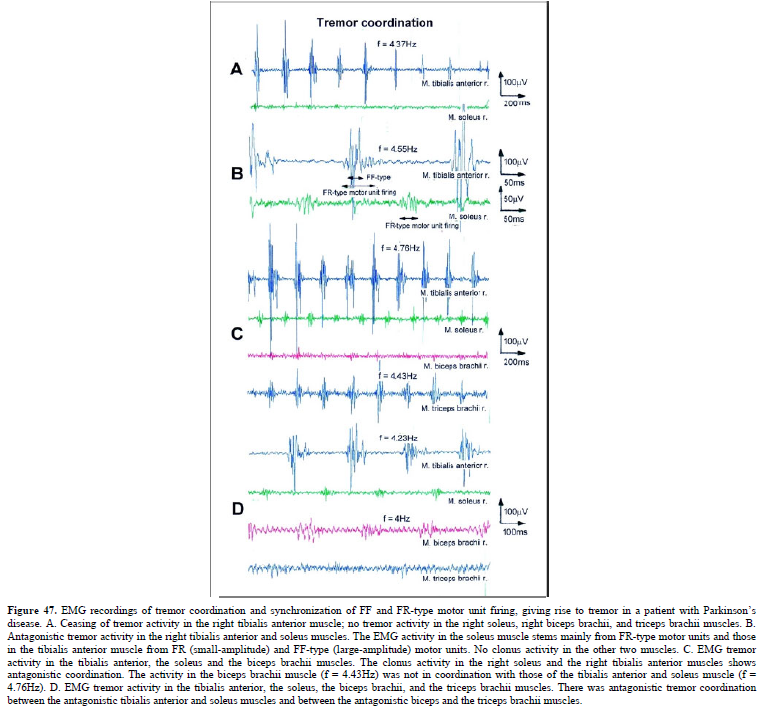
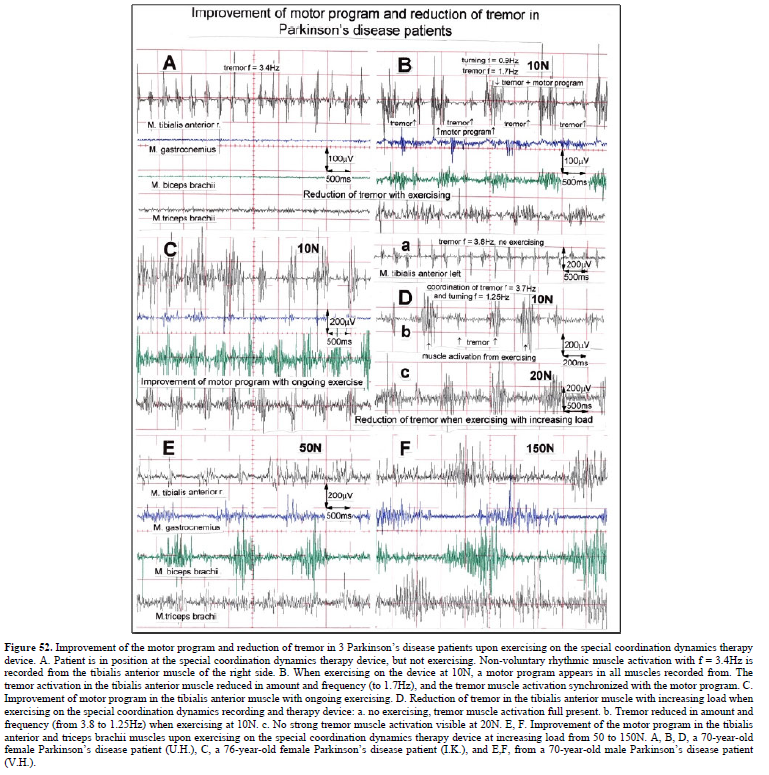
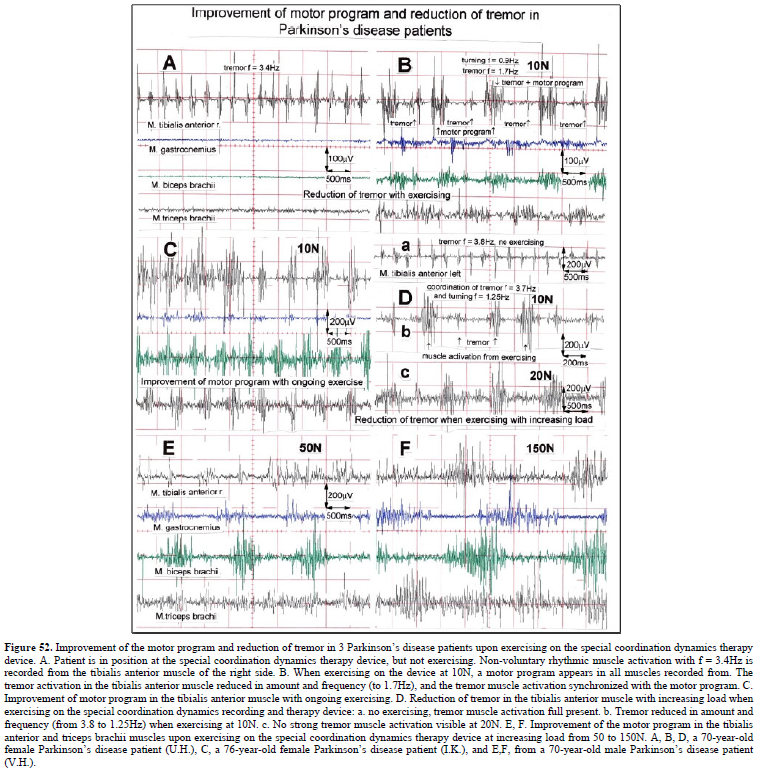
Schalow G, Pääsuke M, Jaigma P (2005) Integrative re-organization mechanism for reducing tremor in Parkinson’s disease patients. Electromyogr Clin Neurophysiol 45: 407-415.
-
Windhorst U (1988) How Brain-Like is the Spinal Cord? Springer Verlag, Berlin.
-
Schalow G, Pääsuke M, Ereline J, Gapeyeva H (2004) Improvement in Parkinson’s disease patients achieved by coordination dynamics therapy. Electromyogr Clin Neurophysiol 44: 67-73.
-
Schalow G (2017) Breast cancer growth inhibition via Coordination Dynamics Therapy. In: “Horizons in Cancer Research. Editor: Hiroto S. Watanabe. Nova Science Publishers, Inc, Hauppauge NY, USA. Vol 68, pp: 125-151.
-
Schalow G (2010) Cure of urinary bladder functions in severe (95%) motoric complete cervical spinal cord injury in human. Electromyogr Clin Neurophysiol 50: 155-179.
-
Schalow G (1990) Feeder arteries, longitudinal arterial trunks and arterial anastomoses of the lower human spinal cord. Zentralbl Neurochir 51: 181-184.
-
Schalow G, Pääsuke M (2003) Low-load coordination dynamics in athletes, physiotherapists, gymnasts, musicians and patients with spinal cord injury, after stroke, traumatic brain lesion and with cerebral palsy. Electromyogr Clin Neurophysiol 43: 195-201.
-
Schalow G, Pääsuke M, Kolts I (2003) High-load coordination dynamics in athletes, physiotherapists, gymnasts, musicians and patients with CNS injury. Electromyogr Clin Neurophysiol 43: 353-365.
-
Colvis CM, Pollok JD, Goodman RH, Impey S, Dunn J, et al. (2005) Epigenetic mechanisms and gene networks in the nervous system. J. Neurscience 25(45): 10379-10379.
-
Bird A (2007) Perceptions of epigenetics. Nature 447: 396-398.
-
Covic M, Karaca E, Lie DC (2010) Epigenetic regulation of neurogenesis in the adult hippocampus. Heredity 105: 122-134.
-
Brown JC, Winters-Stone K, Schmitz KH (2012) Cancer, physical activity, and exercise. Compr Physiol 2: 2775-2809.
-
Christensen JF, Jones LW, Andersen JL, Daugaard G, Rorth M, et al. (2014) Muscle dysfunction in cancer patients. Ann Oncol 25: 947-958.
-
Schalow G (2002) Stroke recovery induced by coordination dynamic therapy and quantified by the coordination dynamic recording method. Electromyogr Clin Neurophysiol 42: 85-104.
-
Schalow G (2002) Improvement after traumatic brain injury achieved by coordination dynamic therapy. Electromyogr Clin Neurophysiol 42: 195-203.
-
Schalow G, Jaigma P (2006) Improvement in severe traumatic brain injury induced by coordination dynamics therapy in comparison to physiologic CNS development. Electromyogr Clin Neurophysiol 46: 195-209.
-
Schalow G (2002) Recovery from spinal cord injury achieved by 3 months of coordination dynamic therapy. Electromyogr Clin Neurophysiol 42: 367-376.
-
Schalow G (2003) Partial cure of spinal cord injury achieved by 6 to 13 months of coordination dynamic therapy. Electromyogr Clin Neurophysiol 43: 281-292.
-
Schalow G, Jaigma P, Belle VK (2009) Near-total functional recovery achieved in partial spinal cord injury (50% injury) after 3 years of coordination dynamics therapy. Electromyogr Clin Neurophysiol 49: 67-91.
-
Schalow G (2010) Cure of urinary bladder functions in severe (95%) motoric complete cervical spinal cord injury in human. Electromyogr Clin Neurophysiol 50: 155-179.
-
Schalow G (2006) Cerebellar injury improvement achieved by coordination dynamics therapy. Electromyogr Clin Neurophysiol 46: 433-439.
-
Schalow G, Jaigma P (2005) Cerebral palsy improvement achieved by coordination dynamics therapy. Electromyogr Clin Neurophysiol 45: 433-445.
-
Schalow G (2006) Hypoxic brain injury improvement induced by coordination dynamics therapy in comparison to CNS development. Electromyogr Clin Neurophysiol 46: 171-183.
-
Schalow G, Nyffeler T (2001) Coordination dynamics therapy: Myelomeningocele (spina bifida). Physical Therapy.
-
Schalow G, Nyffeler T (2000) Coordinate dynamics therapy: Scoliosis. Physiotherapy.
-
Schalow G (2019) Permanent coma patient re-learned to speak via Coordination Dynamics Therapy. Arch Clin Med Case Rep 3(2): 33-50.
-
Schalow G (2020) Anaplastic oligodendroglioma WHO III brain cancer-patient recovered following operation, radiation and chemotherapy through Coordination Dynamics Therapy, which is also a Covid-19 treatment without ventilator. Int J Med Clin Imaging 5(2): 165-210.
-
Schalow G (2020) From brain repair to Covid-19 treatment via Coordination Dynamics Therapy. (Video film contains therapy movements of patient’s recent publications). Available online at: https://www.scitcentral.com/article/62/1656/From-Brain-Repair-to-COVID-19-Treatment-via-Coordination-Dynamics-Therapy-(CDT) Video conference on: August 19, 2020. (Download from www.coordination-dynamics-therapy.com).
-
Schalow G (2020) Classification and Identification of Human Peripheral Nerve Fibers by Conduction Velocity, Nerve Fiber Diameter and Natural Firing Patterns with Consequences for CNS Repair and Covid-19 Infection Treatment. Int J Med Clin Imaging 5(3): 231-314.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Pathology and Toxicology Research
- Advance Research on Endocrinology and Metabolism (ISSN: 2689-8209)
- Journal of Rheumatology Research (ISSN:2641-6999)
- Journal of Otolaryngology and Neurotology Research(ISSN:2641-6956)
- Journal of Neurosurgery Imaging and Techniques (ISSN:2473-1943)
- Journal of Cancer Science and Treatment (ISSN:2641-7472)
- Advance Research on Alzheimers and Parkinsons Disease