Research Article
Doppler Vascularization of Uterine Fibroids According to Location
3182
Views & Citations2182
Likes & Shares
Uterine fibroids are utmost common benign tumors sometimes they affect even after menopause. The estimated incidence of uterine fibroids is 20-40% in reproductive age. There are several factors that attributed to underlie the development and incidence of these common tumors, but this further corroborates their relatively unknown etiology.
Methods: A prospective case control study was carried out, at Sabiry Color Doppler Ultrasound Centre Faisalabad during November, 2015 to March, 2016 on a group of 100 Pakistani females, with clinical suspicions of uterine fibroids. Toshiba Nemio XG & Aplio 500 with 3.5-5 MHztransducer was used. Patients were scanned with adequately full bladder, Fibroids were identified, their sizes and location were evaluated and recorded. RI of the main feeding vessel of fibroids was calculated. The fibroids were divided into three types, according to their perfusion characteristics as follows: Type 1: Perfusion only at the periphery; Type 2: Central perfusion; Type 3: Both central and peripheral perfusion.
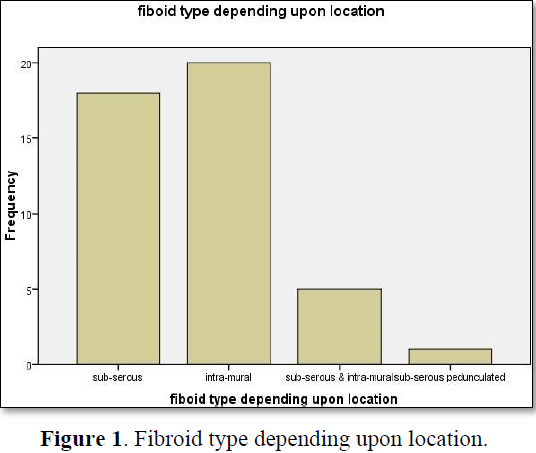
Results: The fibroids were found in 44 patients out of 100 females. They had different types of fibroids depending upon their location. Perfusion grading of the fibroids was done and R.I of feeding vessels was calculated. Both of the variables were correlated. The percentage of uterine fibroids were 18 (40.9%) sub-serous, 20 (45.4%) intra-mural, 5 (11.4%) sub-serous and intra- mural, 1 (2.3%) sub-serous pedunculated. The resistive index of feeding vessel of different fibroids ranges between 0.33-1.00. The perfusion grading of fibroids that is 29 (65.9%) for grade 1, 2 (4.6%) grade 2, 13 (29.5%) for grade 3.
Conclusion: The majority (73%) was found to be in the group of age (22-40) year, (33%), (20-30 years), and (31-40 years), 43% with the higher incidence of uterine fibroid being among the same age group. Among fibroids Intramural fibroids has higher incidence 45.4%. Ultrasound is of high values, useful and practical in evaluation of uterine masses patients. The correlation between grading and R.I is significant.
Keywords: Intra-mural, Perfusion, Resistive index, Sub-serous, Uterine fibroids
INTRODUCTION
Leiomyomas are most common and nonthreatening neoplasms of the uterus. They occur in 20-30% of females over age 30 years [1]. Myomas classified as intramural, confined to the myometrium; submucosal, protrusive into uterine cavity and displacing or distorting the endometrium; or subserosal, bulging from the peritoneal surface of the uterus. Intramural fibroids are more common than Submucosal fibroids, while submucosal myomas produce symptoms most frequently and also associated with infertility. Large size subserosal fibroids might be pedunculated extant as an adnexal mass. Cervical fibroids account forapproximately8% of all fibroids [2]. Fibroids are estrogen dependent, increase in size during an ovulatory cycle, as a result of unopposed estrogen stimulation, In pregnancy, even though about one half of all fibroids show little significant change during pregnancy [3]. In first trimester they are associated with increased risk of pregnancy loss, which is higher in patients with multiple fibroids than in those with a single fibroid [4]. Leiomyomas rarely develop in postmenopausal women, and increase in size in postmenopausal patients who are undergoing HRT; they stabilize or decrease in sizeafter menopause.
Tamoxifen has also been reported to cause growth in leiomyomas [5]. Sonographically, myomas have variable appearances, most often hypoechoic or heterogeneous in echo texture. Calcification may occur in fibroids, frequently appearing as focal areas of increased echogenicity with shadowing, or as a curvilinear echogenic rim, which may simulate the outline of a fetal head [6]. When fibroids undergo rapid growth, they tend to outgrow their blood supply, leading to degeneration, necrosis and producing areas of decreased echogenicity or cystic spaces within the fibroid [7]. This often during pregnancy, affecting about 7-8% of pregnant females with fibroids, it presents with pain over this area [8]. Submucosal fibroids are usually broad-based solid hypoechoic masses with an overlying layer of echogenic endometrium. Sonographically, the finding of a highly echogenic, attenuating mass within the myometrium is virtually diagnostic of this condition [9]. It is important to identify the lesion within the uterus so as not to confuse it with the more common, similar-appearing, fat-containing ovarian dermoid. Lipomatous uterine tumors are usually asymptomatic, they do not require surgery [10]. In this study we divided the fibroids into three types, according to their perfusion characteristics: Type 1: Perfusion only at the periphery; Type 2: Central perfusion; Type 3: Both central and peripheral perfusion.
The aim of this study was to aid in investigation and management of fibroids. Malignancies of uterine fibroids may be difficult to distinguish clinically and on grey scale, Doppler sonography is reliable now days. The diagnosis of uterine fibroids on color Doppler sonography makes it possible to differentiate between benign and malignant.
METHODOLOGY
This prospective case control study was carried out at Sabiry color Doppler ultrasound Centre Faisalabad between November 2015 to March 2016. The study was approved by the institutional review board and all women were informed about its purpose and gave their written consent to participate. One hundred females with clinical suspicion of uterine fibroids were included in this study. All ultrasound examinations were performed by the same examiner using a Toshiba Nemio XG & Aplio 500 with 3.5-5 MHz transducer. Myoma characteristics, such as location (submucous, intramural or subserous), topographic site (mostly anterior or posterior) were identified. In women with multiple fibroids, each myoma was measured and reference to its specific location. Demographic characteristics of patients including age, infertility, parity and body mass index (BMI) and menstrual cycle bleed were recorded. Statistical analysis was performed using the SPSS 20 software package. The normal distribution of quantitative variables age, parity, menstrual cycle bleed, BMI, size and blood flow in myoma were verified. Ultrasound examinations of myomas diameter were normally distributed and are reported as mean ±SD. Qualitative variables (location of myoma, topographic site of myoma) were expressed. The fibroids were divided into three types, according to their perfusion characteristics as follows:
Type-1: Perfusion only at the periphery;
Type-2: Central perfusion;
Type-3: Both central and peripheral perfusion.
RI of the main feeding vessel of fibroids was calculated.
RESULTS
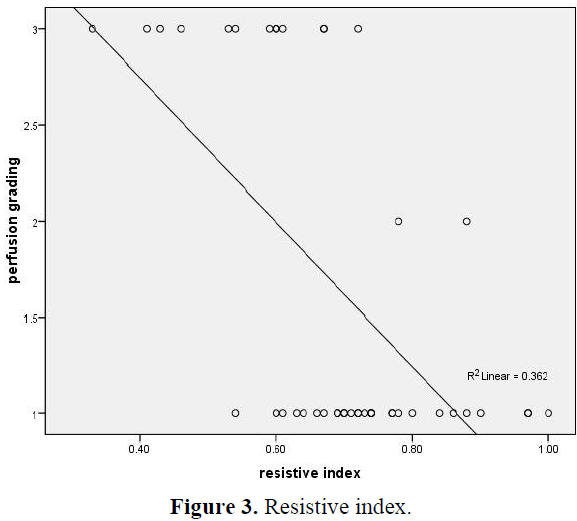
The age of Patients having fibroids 22-59.29 (65.9%) married and 15 (34.1%) unmarried females out of 100 patients. Table 1 showed the fibroid type depending upon location which is 18 (40.9%) sub-serous, 20 (45.4%) intra-mural, 5 (11.4%) sub-serous and intra-mural, 1 (2.3%) sub-serous pedunculated (Figure 1). Table 1 showed resistive index of feeding vessels of different fibroids ranges between 0.33-1.00. Table 2 showed the perfusion grading of fibroid which is 29 (65.9%) for grade 1, 2 (4.6%) grade 2, 13 (29.5%) for grade 3 (Figures 2 and 3).
CONCLUSION
The majority 73% was found in the age group of 22-40 y, 33% in 20-30 y of age, and 43% in 31-40 y with the higher incidence of uterine fibroid being among the same age group. The correlation between grading and R.I is significant. Among fibroids, intramural fibroids have higher incidence 45.4%. Color Doppler values are useful in evaluation of uterine fibroids. Early detection of fibroids on ultrasonography can be treated by HRT (hormonal replacement therapy).
This is a potential cause of concern when fibroids are left untreated or managed conservatively, they can transform into malignancy.
DISCUSSION
Uterine fibroids are the benign tumors of the uterus and the most common indication for hysterectomy [11]. The most functional imaging modality for evaluating uterine fibroids is ultrasonography and the vascularity changes of fibroids can be easily assessed by color Doppler sonography [12]. Sonographically, the finding of a highly echogenic attenuating mass within the myometrium is virtually diagnostic of this condition [8]. Uterine fibroids can also be assessed by MRI but it is expensive, so majority of the patients with symptomatic uterine fibroids undergo sonographic evaluation [13]. It is important to identify the lesion within the uterus so as not to confuse it with the more common similar-appearing fat-containing ovarian dermoid. Aslipomatous uterine tumors are usually asymptomatic they do not require surgery [10]. The incidence of fibroids according to location in our study was 45.4% intramural, 40.9% subserosal, 11.4% subseroal and intramural and 2.3% subserosal pedunculated. Nieuwenhuis et al. [14] found 66 women having uterine fibroids with 5 Submucosal, 32 Intramural, and 29 Subserosal mean age 42 years. Ukwenya et al. found 7.8% subserosal, 5.1% submucosal, 4.5% pedunculated and 74.4% intramural. Sarkodie et al., reported 57.8% intramural, 4.4% submucosal and 55.6% in more than one layer [15,16]. The mean age of fibroid presentation in our study was 34 years and in studies and was 42, 31 and 31 years respectively [14,16,17]. In our study, 65.9% fibroids were in grade1 having peripheral flow, 4.6% grade-2 having central flow & 29.5% grade-3 having peripheral and central flow. 56 of U/S finding were found to be normal. The morphological criteria of “peripheral vascularity” were seen in 44 cases, being the criterion showing highest prevalence. Kurjak et al. [18] have shown increased blood velocity and decreased RI and PI in both uterine arteries in patients with uterine fibroids. Sladkevicius et al. [19] found that uterine myomas substantially affect blood flow velocity in the uterine arteries resulting PI values commonly
REFERENCES
-
Kurman RJ (2013) Blaustein's pathology of the female genital tract. Springer Science & Business Media.
-
Kirchner L, Eng KA, Hudelist G (2018) Hysteroscopy, pp : 375-386.
-
Sampat K, Alleemudder DI (2018) Fibroids in pregnancy: Management and outcomes. Obstet Gynaecol 20 : 187-195.
-
Benson CB, Chow JS, Lee WC, Hill IIIJA, Doubilet PM (2001) Outcome of pregnancies in women with uterine leiomyomas identified by sonography in the first trimester. J Clin Ultrasound 29 : 261-264.
-
Dilts PV, Hopkins MP, Chang AE, Cody RL (1992) Rapid growth of leiomyoma in patient receiving tamoxifen. Am J Obstet Gynecol 166 : 167-168.
-
Early HM, Gahan JPM, Scoutt LM, Revzin M, Lamba R, et al. (2016) Pitfalls of sonographic imaging of uterine leiomyoma. Ultrasound Q32 : 164-174.
-
Caoili EM, Hertzberg BS, Kliewer MA, DeLong D, Bowie JD (2000) Refractory shadowing from pelvic masses on sonography: A useful diagnostic sign for uterine leiomyomas. AJR Am J Roentgenol 174 : 97-101.
-
Dodd GD, Budzik RF (1990) Lipomatous uterine tumors: Diagnosis by ultrasound, CT and MR. J Comput Assist Tomogr 14: 629-632.
-
Serafini G, Martinoli C, Quadri P, Speca S, Crespi G, et al. (1996) Lipomatoustumorsof the uterus: Ultrasonographic findings in 11 cases. J Ultrasound Med 15 : 195-199.
-
Hertzberg BS, Kliewer MA, George P, Donald JWM, Carroll BA (1995) Lipomatousuterine masses : Potential to mimic ovarian dermoids on endovaginal sonography. J Ultrasound Med 14 : 689-692.
-
Stewart EA (2001) Uterine fibroids. Lancet 357 : 293-298.
-
Muniz CJ, Fleischer AC, Donnelly EF, Mazer MJ (2002) Three-dimensional color doppler sonography and uterine artery arteriography of fibroids. J Ultrasound Med 21 : 129-133.
-
Exacoustos C, Romanini ME, Amadio A, Amoroso C, Szabolcs B, et al. (2007) Can gray-scale and color Doppler sonography differentiate between uterine leiomyosarcoma and leiomyoma? J Clin Ultrasound 35 : 449-457.
-
Nieuwenhuis LL, Keizer AL, Stoelinga B, Twisk J, Hehenkamp W, et al. (2018) Fibroid vascularisation assessed with three-dimensional power Doppler ultrasound is a predictor for uterine fibroid growth: A prospective cohort study. Int J Obstet Gynaecol 125 : 577-584.
-
Ukwenya V, Maduemezia N, Afolayan O, Alese O, Thomas W (2015) Prevalence of uterine fibroid in a South-Western Nigerian population : A sonographic study. J Exp Clin Anat 14 : 24.
-
Sarkodie BD, Botwe BO, Ofori EK (2016) Uterine fibroid characteristics and sonographic pattern among Ghanaian females undergoing pelvic ultrasound scan : A study at 3-major centres. BMC Womens Health16 : 10.
-
Exacoustòs C, Rosati P (1993) Ultrasound diagnosis of uterine myomas and complications in pregnancy. Obstet Gynecol 82 : 97-101.
-
Kurjak A, Urek SK, Miric D (1992) The assessment of benign uterine tumor vascularization by transvaginal color Doppler. Ultrasound Med Biol 18 : 645-649.
-
Sharma K, Bora MK, Venkatesh BP, Barman P, Roy SK, et al. (2015) Role of 3D ultrasound and doppler in differentiating clinically suspected cases of leiomyoma and adenomyosis of uterus. J Clin Diagn Res 9 : QC08-QC12.
-
Gupta KP, Jain SK (2016) Role of ultra-sonography and color doppler to diagnosis of pelvic masses and its correlation with histopathological findings. Int J Sci Study 4 : 147-153.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- International Journal of Internal Medicine and Geriatrics (ISSN: 2689-7687)
- Journal of Otolaryngology and Neurotology Research(ISSN:2641-6956)
- International Journal of Radiography Imaging & Radiation Therapy (ISSN:2642-0392)
- Chemotherapy Research Journal (ISSN:2642-0236)
- Journal of Carcinogenesis and Mutagenesis Research (ISSN: 2643-0541)
- Advance Research on Endocrinology and Metabolism (ISSN: 2689-8209)
- Journal of Pathology and Toxicology Research