2691
Views & Citations1691
Likes & Shares
Background: Lichen planus is an autoimmune disease affecting the skin and oral mucosa with unclear pathogenesis. The decreased serum lycopene level has been monitored in symptomatic oral lichen planus (OLP) cases. This trial is directed to evaluate the clinical effectiveness of oral lycopene as well as to assess its effect on the salivary MDA expression levels in symptomatic patients affected by OLP.
Methods: 20 patients (Pts) suffering from atrophic/erosive OLP were included in this clinical trial. Lycopene was administered 10 mg/day for 8 successive weeks. Outcome measures included the visual analogue scale and clinical scoring. The un-stimulated salivary MDA was also assayed at baseline, then at weeks: 2, 4 and 8 after therapy.
Results: Lycopene use resulted in a significant reduction at p<0.05 in pain scores and clinical score at weeks: (2, 4 and 8) after administration as well as the mean expression levels of salivary MDA (p<0.05).
Conclusion: Systemic lycopene reduced the salivary MDA expression, and seems be a promising alternative therapy for symptomatic oral lichen planus patients.
Keywords: Oral lichen planus, Lycopene, Anti-oxidant, Pain, Malondialdehyde
INTRODUCTION
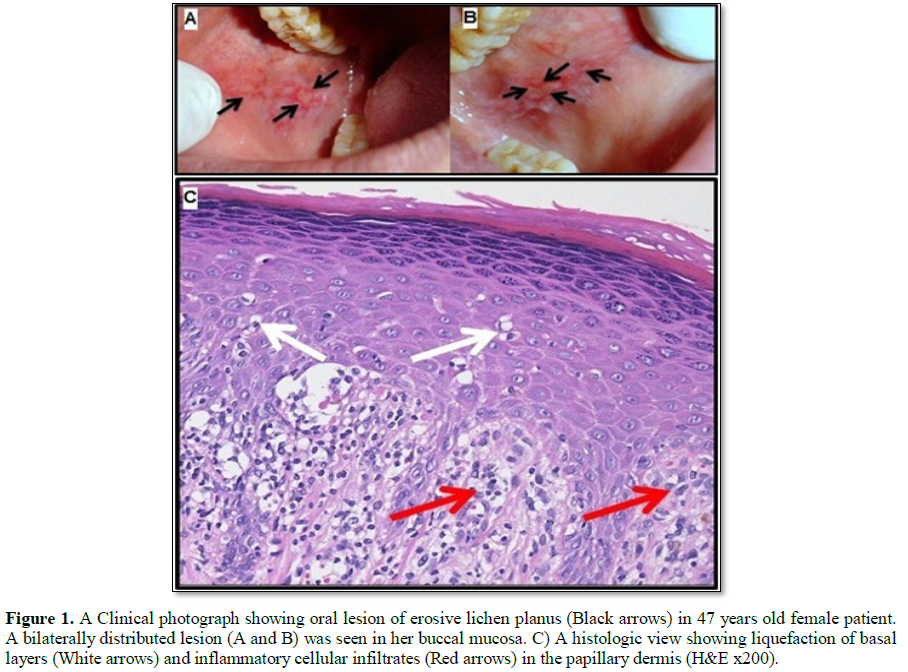
Lichen planus (LP) is an autoimmune chronic inflammatory, disease that is seen in 1-2% of the population. LP affects the skin, mucous membrane, nails and hair [1]. Oral lichen planus (OLP) is the oral mucosal counterpart of LP that affects 0.5-4% of the adult population; with a higher incidence in middle-aged females [2]. OLP may appear as reticular, papular, plaque like, atrophic and bullous-erosive. The erosive and atrophic forms are often symptomatic and need effective therapeutic interference [3], because clinically these categories are related to oral cancer development [4].
However, the basic mechanisms directing OLP toward the development of oral cancer have not been clearly addressed yet. Reactive oxygen species (ROS) were reported as a corner stone in the inflammation-mediated carcinogenesis through their interaction with polyunsaturated fatty acids (PAFs) in membranes or lipoproteins, leading to lipid peroxidation [5].
The management of this disease is aiming at pain alleviation and remission of the symptoms. This could be achieved through the use of different corticosteroids. However, despite the efficacy of corticosteroids, many side effects are to be considered before using it [6].
The uncontrolled production of lipid peroxides may lead to oxidative stress, with notable destruction to cell integrity. Numerous markers have been recommended to observe the lipid peroxidation process. Malondialdehyde (MDA) is a common product of PAFs peroxidation that is increased secondary to oxidative stress [7].
Currently, the ability of oxidative stress coupled with the diminished antioxidant enzyme expression has been suggested in the pathogenesis of OLP [8]. Moreover, decreased serum carotenoid's levels have been reported in patients with LP [9]. In 2011, a notable lower serum level of lycopene was monitored in symptomatic OLP cases [10].
Lycopene is a red-colored carotenoid that has a variety of therapeutic properties like inhibition of cancer cell proliferation, antioxidant activity, inducing phase II, interference with growth factor stimulation, control of transcription and restoration of gap junctions. Lycopene has also a unique antioxidant activity via its physical and chemical quenching ability of singlet oxygen [11]. However; the direct evaluation of this anti-oxidant role was not assessed. The present study evaluated the salivary malondialdehyde (MDA) as a quantitative indicator for the anti-oxidant capacity of the treatment as well as evaluation of the effectiveness of oral lycopene in the management of symptomatic OLP-patients.
SUBJECTS AND METHODS
Study design
A non-controlled clinical trial was applied on 20 patients suffering from symptomatic OLP. The design of this trial (16CCT20-311) was accepted by the Ethics Committee of Al-Azhar University (Girls Branch). This treatment protocol was applied in accord with the ethical fundamentals described in the Declaration of Helsinki.
Participants
Eligibility criteria:
Inclusion criteria: Patients suffering from symptomatic OLP (atrophic/erosive). The included cases were having oral lesions of at least 10 mm in their widest dimension. Patients were free from any systemic disease except for OLP [12].
Exclusion criteria: Lichenoid reactions, malignant lesion or infective oral involvement. In addition, patients who received topical therapy for OLP in the last 2 weeks or systemic therapy in the last 4 weeks [13] or any lichenoid inducing drugs. Pregnant or breast feeding women were also excluded.
Study setting: The participating patients were recruited in a consecutive order from the Oral Medicine clinic, Faculty of Oral and Dental Medicine, Al-Azhar University (Girls Branch), Cairo, Egypt. This study was performed between March 2017 and June 2018.
Intervention
Pre-treatment measures: A detailed history was taken from each patient including: the systemic condition, duration as well as disease and drug history using diagnostic chart. Giving verbal and written oral hygiene instructions. The aim of the study, the steps, and the treatment plan were explained and clarified for each participant. Oral mucosal incisional biopsies measuring 5-7 mm; including a healthy and integrate tissue zone, were taken from the most representative lesion area. The definite diagnosis of OLP cases (Figure 1) was assessed in accordance with the modified criteria of OLP [14].
Application of intervention: Patients received systemic treatment with lycopene (LYCOPENE 10 mg 60 Soft gels, Biovea, Egypt), twice daily for eight consecutive weeks.
Outcomes assessment
Each patient was evaluated immediately before active treatment (T0), then during the treatment course, after 2 weeks (T1), 4 weeks (T2), and 8 weeks (T3) [15]. After completion of therapy, the following scores were used in this study:
Primary outcome:
Clinical scoring (CS): The representative lesion was characterized by being the most severe and clear lesion, and diagnosed by inspection using CS [16] as following:
· 0 means no lesion/normal mucosa.
· 1 means mild white striae⁄no erythematous area.
· 2 means white striae with atrophic area 2.
· 3 means white striae with atrophic area more than 1 cm2.
· 4 means white striae with erosive area 2.
· 5, white striae with erosive area more than 1 cm2.
Secondary outcomes:
Visual analogue scale (VAS): It consisted of a 10 cm horizontal line marked 0-10 (0 no pain; 10 most severe pain experienced). Each patient was asked to mark the scale at the follow-up time points [17].
Saliva collection for malondialdehyde (MDA) assessment: Five milliliter of unstimulated whole salivary samples was expectorated in dry plastic vials. Salivary samples underwent the following steps:
· Centrifuged at 2000 g for 10 min.
· The supernatants were stored at -70°C until further analysis.
· Salivary samples were taken at the same time of day (10-12 AM) and at least 2 h after the last food or drink intake.
Measurement of saliva MDA: The salivary levels of MDA were determined by a method based on reaction with thiobarbituric acid (TBA) at 90-100°C according to Esterbauer and Cheeseman [18] and expressed as ng/ml.
STATISTICAL ANALYSIS
Data management and statistical analysis were done using Statistical Package for Social Sciences, Version 21.0 (SPSS, IBM) for Windows. Comparisons between the base line and after treatment at different study time’s interventions and over time were done by paired T-test. For pain score as well as clinical score, both were expressed as median and range and differences between the 2 groups was tested by the Student-T test was used for changes overtime. Adjustments of the p-value for multiple testing were performed using Bonferroni method. P value
RESULTS
The present study included a sample size of 20 patients. None of the study patients discontinued the applied protocol or the follow up visits. All the collected data were applied for statistical analysis.
Clinical improvement was assessed as primary outcome using clinical score (CS) immediately before the treatment administration, then during the treatment course, at 2, 4 and 8 weeks after completion of therapy. The secondary outcomes were pain was assessed using VAS and the salivary assessment of MDA.
Demographic data
The age ranged from 46 to 62 years with a mean of 52.1 ± 4.2. Regarding the gender, 12 (60%) out of the 20 patients enrolled in the test group were females and 8 patients (40%) were males.
Clinical score results
A statistically significant difference was noticed comparing the clinical score values before and 2 weeks after treatment and through all the follow up time 4 and 8 weeks after treatment as mentioned in Table 1. The highest mean of clinical score was reported before treatment 3.6 ± 0.88 while the lowest mean of clinical score was noted in week 8 after treatment 1.15 ± 0.74 (Table 2).
Visual analogue score results
A statistically significant difference in VAS values was found comparing before and 2 weeks after treatment and through all the follow up time 4 and 8 (Table 2). The highest mean value for pain score was reported before treatment 7.65 ± 0.74. The least mean values of pain score were on week 8 after treatment 2.95 ± 0.82 (Table 3).
Salivary malondialdehyde (MDA)
There was a statistically significant difference in MDA values before and 2 weeks after treatment and through all the follow up time 4 and 8 weeks, 4 and 8 weeks after treatment (Table 4).
The least mean values were on week 8 after treatment 312.95 ± 51.4. The highest mean was reported before treatment 395.55 ± 7.8 (Table 5).
DISCUSSION
Among many alternative treatments used to spare corticosteroids in OLP, herbal medicine could be of value as palliative treatment to help decrease the symptoms of the disease and overcome the side effects of corticosteroids [19]. Given that the oxidative stress is believed to have a crucial effect in the pathogenesis of OLP, the use of anti-oxidants would be implied to induce remission in OLP patients [20].
Lycopene is a plant extract that has been used to treat many diseases with oxidative stresses playing a role in their pathogenesis [21], including OLP [15]. Therefore, the current study evaluates the use of 10 mg of lycopene per day for 8 weeks in management of symptomatic OLP cases. Clinical score was measured before, 2, 4 and 8 weeks after treatment to evaluate the remission of oral signs. Visual analogue scale of pain was evaluated at the same time intervals; along with the salivary malondialdehyde (MDA) as a quantitative indicator for the anti-oxidant capacity of the treatment. The reaction of malondialdehyde with thiobarbituric acid has been widely employed in the spectrophotometric detection of malondialdehyde in several biological samples [22].
The present study reported reduction in clinical score of oral mucositis and pain in 2 weeks after the use of lycopene. These results are in accordance with the findings of Saawarn et al. [23]. The authors compared lycopene 8 mg per day for 8 weeks to identical placebo on a sample of 30 symptomatic OLP patients and reported that lycopene has a valuable effect in treating OLP. They also added that oxidative stress may have a potential role in disease pathogenesis.
Moreover, the results of Shekhawat et al. [15] were in line with our findings. Their study included 50 symptomatic OLP patients that were randomly assigned into two groups; one received 8 mg lycopene per day while the other group had levamisole in a dose of 50 mg 3 times/day for 3 successive days per week; for 8 weeks. Pain was assessed 2, 4 and 8 weeks after treatment and lycopene has shown a more potent and faster therapeutic effect.
Kushwaha et al. [24] used oral lycopene capsules (4 mg/day) for 8 successive weeks with symptomatic OLP patients. The assessment was done at 0, 2, 4, 6 and 8 weeks intervals to record the clinical and symptomatic improvement in OLP lesions. The authors reported as an effective therapy in relieving patients’ symptoms.
Moreover, the use of lycopene in our study revealed a notable decrease in the monitored salivary MDA levels along the duration of study. This result can be explained in the light of its ability to scavenge free radicals [25]. It is worth noting that this was the first clinical report to provide data about the effect of systemic lycopene administration and the oxidative stress biomarker in treating OLP patients. Given that lycopene deficiency has been reported in symptomatic OLP cases (9) and lycopene supplementation significantly decreased the salivary MDA parallel with clinical improvement, a decreased lycopene levels are suggested to have a role in the etio-pathogenesis of OLP for future research. Additionally, lycopene can be successfully used as a sole treatment to manage OLP cases, and as a corticosteroid sparing as recommended by various researchers [6,21].
Nevertheless, a special concern should be paid to the used dose of lycopene to be used as a corticosteroid sparing line. In this regard, Kushwaha et al. [26] have compared the use of lycopene (4 mg/day) with the systemic prednisolone (40 mg/day) in OLP patients for eight consecutive weeks. The authors reported a significant difference in pain reduction in the prednisolone group. This finding might be attributed to the used dose of lycopene (4 mg), in their model.
In accord, Devaraj et al. [27] examined the immune-modulatory effects of different doses (0, 6.5, 15 or 30 mg lycopene/day for 8 weeks) of purified lycopene supplementation on the oxidative stress biomarkers in healthy volunteers. The authors concluded that purified lycopene delivered a decrease in DNA oxidative damage at the high dose. The aforementioned speculations can explain the good clinical and biochemical results of our study; with further confirmation of the used regimen of lycopene (10 mg/day for 8 weeks); owing to its dose dependent effect.
A possible limitation can be addressed of our study; the relatively short evaluation period. However, this time frame was taken by various researchers [15,23,24]. Indeed, longer follow up clinical trials are needed to further understand the effectiveness of various therapeutic protocols in treating OLP cases, as recommended [28].
CONCLUSION
According to our data; the oral use of lycopene has a promising clinical advantage in treating symptomatic OLP patients via its immune-modulatory effect on the expression levels of the salivary MDA. This finding indirectly substantiates the hypothesis of the potential role of oxidative stress in the pathogenesis of lichen planus. Further studies should be conducted to assess the maintenance effects of oral lycopene.
ACKNOWLEDGEMENT
The authors acknowledge the financial support of the Faculty of Oral and Dental Medicine, Al-Azhar University by providing us with the necessary materials used in this research in addition to the technical support.
CONFLICTS OF INTEREST
Authors certify that they have no involvement in any organization or entity with any interest in the discussed protocol in this work.
1. Farhi D, Dupin N (2010) Pathophysiology, etiologic factors and clinical management of oral lichen planus. Part I: Facts and controversies. Clin Dermatol 28: 100-108.
2. Axell T, Rundquist L (1987) Oral lichen planus - A demographic study. Community Dent Oral Epidemiol 15: 52-56.
3. Gorsky M, Raviv M (1992) Efficacy of etretinate (Tigason) in symptomatic oral lichen planus. Oral Surg Oral Med Oral Pathol 73: 52-55.
4. Mignogna MD, Fedele S, Lo Russo L, Lo Muzio L, Bucci E (2004) Immune activation and chronic inflammation as the cause of malignancy in oral lichen planus: Is there any evidence? Oral Oncol 40: 120-130.
5. Gue¢rin P, El Mouatassim S, Me¢ne¢zo Y (2001) Oxidative stress and protection against reactive oxygen species in the pre-implantation embryo and its surroundings. Hum Reprod Update 7: 175-189.
6. Gonzalez-Moles MA, Scully C (2005) Vesiculo-erosive oral mucosal disease management with topical corticosteroids: Fundamental principles and specific agents available. J Dent Res 84: 294-301.
7. Marnett LJ (1999) Chemistry and biology of DNA damage by malondialdehyde. IARC Sci Publ 150: 17-27.
8. Agha-Hosseini F, Mirzaii-Dizgah I, Mikaili S. Abdollahi M (2009) Increased salivary lipid peroxidation in human subjects with oral lichen planus. Int J Dent Hyg 7: 246-250.
9. Rollman O, Vahlquist A (1985) Vitamin A in skin and serum-studies of acne vulgaris, atopic dermatitis, ichthyosis vulgaris and lichen planus. Br J Dermatol 113: 405-413.
10. Nagao T, Warnakulasuriya S, Ikeda N, Fukano H, Yamamoto S, et al. (2001) Serum antioxidant micronutrient levels in oral lichen planus. J Oral Pathol Med 30: 264-267.
11. Levy J, Sharoni Y (2004) The functions of tomato lycopene and its role in human health. Herbalgram 62: 49-56.
12. Abramson J (1996) The cornel medical index as an epidemiological tool. Am J Pub Health 56: 287-298.
13. Ibrahim SS, Hazzaa HH (2012) Topical pimecrolimus effect on Fas inducing apoptosis in oral lichen planus: A clinical immunohistochemical study. J Oral Pathol Med 41: 315-321.
14. van der Meij EH, van der Waal I (2003) Lack of clinicopathologic correlation in the diagnosis of oral lichen planus based on the presently available diagnostic criteria and suggestions for modifications. J Oral Pathol Med 32: 507-512.
15. Shekhawat KS, Deepak TA, Srivastava C (2016) Assessment of lycopene and levamisole in management of oral lichen planus - A comparative study. Oral Surg Oral Med Oral Pathol Oral Radiol 2: 4-10.
16. Thongprasom K, Luengvisut P, Wongwatanakij A, Boonjatturus C (2003) Clinical evaluation in treatment of oral lichen planus with topical fluocinolone acetonide: A 2 year follow-up. J Oral Pathol Med 32: 315-322.
17. Scott J, Huskisson EC (1976) Graphic representation of pain. Pain 2: 175-184.
18. Esterbauer H, Cheeseman KH (1990) Determination of aldehydic lipid peroxidation products: Malonaldehyde and 4-hydroxynonenal. Methods Enzymol 186: 407-421.
19. Ghahremanlo A, Boroumand N, Ghazvini K, Hashemy SI (2019) Herbal medicine in oral lichen planus. Phytother Res 33: 288-293.
20. Ergun S, Troşala ŞC, Warnakulasuriya S, Özel S, Önal AE, et al. (2011) Evaluation of oxidative stress and antioxidant profile in patients with oral lichen planus. J Oral Path Med 40: 286-293.
21. Lowe GM, Graham DL, Young AJ (2018) Lycopene: Chemistry, metabolism and bioavailability. In: Lycopene and Tomatoes in Human Nutrition and Health. CRC Press, pp: 1-20.
22. Fogarasi E, Croitoru MD, Fülöp I, Nemes-Nagy E, Tripon RG, et al. (2016) Malondialdehyde levels can be measured in serum and saliva by using a fast HPLC method with visible detection. Revista Romana de Medicina de Laborator 24: 319-326.
23. Saawarn N, Shashikanth MC, Saawarn S, Jirge V, Chaitanya NC, et al. (2011) Lycopene in the management of oral lichen planus: A placebo-controlled study. Indian J Dent Res 22: 639-643.
24. Kushwaha RP, Rauniar GP, Rimal J (2019) Clinical assessment of the effects of lycopene in the management of oral lichen planus. Int Dent Med J Adv Res 5: 1-5.
25. Sander CS, Cooper SM, Ali I, Dean D, Thiele JJ, et al. (2005) Decreased antioxidant enzyme expression and increased oxidative damage in erosive lichen planus of the vulva. BJOG 112: 1572-1575.
26. Kushwaha RP, Rauniar GP, Rimal J (2017) Comparative study of the efficacy of lycopene versus prednisolone in the management of oral lichen planus - A randomized, double blind clinical trial. World J Pharm Pharm Sci 6: 1103-1115.
27. Devaraj S, Mathur S, RD, Jialal I (2008) A dose-response study on the effects of purified lycopene supplementation on biomarkers of oxidative stress. J Am Coll Nutr 27: 267-273.
28. Gupta S, Ghosh S, Gupta S (2017) Interventions for the management of oral lichen planus: A review of the conventional and novel therapies. Oral Dis 23: 1029-1042.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Cancer Science and Treatment (ISSN:2641-7472)
- Journal of Psychiatry and Psychology Research (ISSN:2640-6136)
- International Journal of Radiography Imaging & Radiation Therapy (ISSN:2642-0392)
- Journal of Nursing and Occupational Health (ISSN: 2640-0845)
- Journal of Blood Transfusions and Diseases (ISSN:2641-4023)
- International Journal of Medical and Clinical Imaging (ISSN:2573-1084)
- International Journal of Diabetes (ISSN: 2644-3031)