3920
Views & Citations2920
Likes & Shares
Due to increasing incidence of breast cancers, it became necessary to develop new, fast and accurate methods for diagnosis. Finding breast cancer early i.e., diagnosing it before symptoms to start is the most important strategy to prevent deaths from breast cancer. Invasive methods such as tissue biopsy, antibody combined with nano markers are cumbersome. Imaging methods are fast, accurate and less cumbersome to the patients. For imaging BC, breast as an organ can be scanned through multiple imaging devices. The existing common imaging modes include mammography, ultrasound and MRI. Ultrasound is good at evaluating superficial lumps, but a mammogram is able to reveal abnormalities deeper in the breast tissue. In digital mammography the X-ray film is replaced by solid-state detectors that convert X-rays into electrical signals and the images can be obtained on computer screen. Newer imaging methods which were not common in medical sciences were modified and made use for imaging for BC. MRI, PET, Gama imaging, Thermography, Ecography, Microwave imaging, Acoustic Aptasensor, etc., recently came in vogue. In this review we analyse these imaging modes and discussed the combination of 2 or more modes for decisive and accurate results.
Keywords: Types of cancer, Risk factors, Mamography, Ultrasound, Acoustic Aptasensor, PEM, Ecography, Microwave imaging
ABBREVIATIONS
BR: Breast cancer; MRI: Magnetic Resonance Imaging; PET: Positron Emission tomography; HRT: Hormone Replacement Therapy; DCIS: Ductal Carcinoma in situ; BSEs: Breast Self-Examination; CBE: Clinical Breast examination; BRCA: Breast Cancer Susceptibility gene; PEM: Positron Emission Mammography; BSGI: Specific Gamma Imaging; FDA: Food and Drug Administration; EM: Electromagnetic Waves; UWB: Ultra-Wideband; TSM: Thick Shear Mode acoustics method; BCSC: Breast Cancer Screening Consortium; PCR: Polymerase Chain Reaction
INTRODUCTION
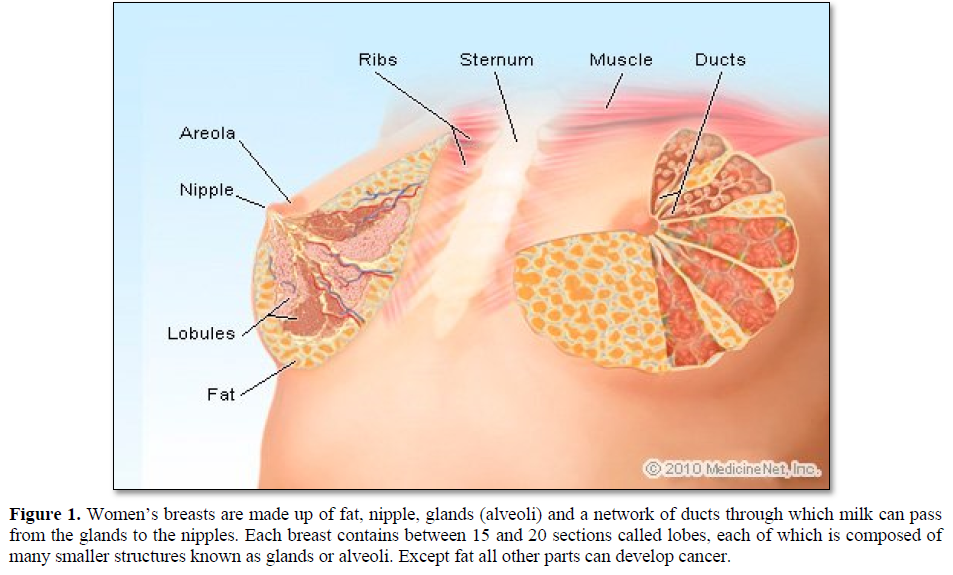
Breast cancer (BC) is the most common cancer in majority of menopausal (50 years or older) women [1]. As puberty sets in, breasts grow in response to the ovarian hormones, oestrogen and progesterone [2]. Breast tissue is composed of milk glands, milk ducts and supportive tissue (dense breast tissue), and fatty tissue [3] (Figure 1). Mammogram can easily differentiate, dense tissue, fatty tissue. The levels of breast density depend on the dense tissue present in the breast and are clearly demarcated in mammograph in the following 4 types [4] (Figure 1):
• the breasts are almost entirely composed of fat. About 1 in 10 women has this result.
• there are some scattered areas of dense, but the majority of the breast tissue is non dense. About 4 in 10 women have this result.
• there are some areas of non-dense tissue, but that the majority of the breast tissue is dense. About 4 in 10 women have this result.
• nearly all of the breast tissue is dense. About 1 in 10 women has this result.
CHANGES IN HORMONE LEVELS
The ovarian hormones change during the menstrual cycle, pregnancy, breast feeding, and menopause. Imbalance of theses hormones in any of these phases and/or normal adulthood phase could be the causal factor for the breast disorders. Menopause does not cause cancer as such, but the risk of developing ovarian, breast and uterine cancers increase as a woman under goes menopause. With age, due to hormonal changes, many of the breast changes set in and there is also an increasing risk of abnormal growths in the breast [5].
Declining oestrogen levels during menopause make breast tissue dehydrated and less elastic, so the breasts lose their rounded shape and begin to sag. After menopause when the ovaries stop producing the hormone, body's main source of oestrogen is the fat tissue. Having more fat tissue means having higher oestrogen levels, which can increase breast cancer risk. Breast cancer is more common in the left breast than the right. The left breast is 5-10% more likely to develop cancer than the right breast [6]. There are many types of BC that differ in their capability to metastasize to other body tissues. Though BC can be many times symptom free, a change in the size or shape of the breast or nipple is noticed in almost all cases. Generally, BC develops as a lump in the breast or armpit in turn lymph nodes in the neck or armpit also swollen. Remarkable changes in the nipples such as; they invert, develop sore and discharge blood [7].
RISK FACTORS FOR BREAST CANCER
Unequivocal risk factor for this disease is non modifiable age. Women today live nearly 30 years longer than they did a century ago. In modern women menarche starts earlier, first full-term pregnancies later, less number of birth, fewer than one mother in five breast feeds for six months or longer, and some women enter menopause later than average, all of which increase the number of years that breasts tissue is exposed to cancer-promoting hormones [8]. Many risk factors are identified and the list increases with more studies every year [9].
Earlier we proved that light as one of the major risk factors for developing BC and substantiated with a blind menopausal women model. Light in night inhibits the conversion of serotonin into melatonin, the key hormone which regulates ovarian hormones [10-13].
More women today drink alcohol and that too in greater amounts than earlier, and now it has been proven that BC risk increases in direct proportion to how much and how often a woman drinks [14]. Being overweight is associated with increased risk for the disease of BC especially after menopause [15]. Body fat produces oestrogen and also stores environmental pollutants (xenoestrogen) that may disrupt the normal balance of hormones and expose the breast tissue at risk for BC. The use of cosmetics with preservatives such as parabens and use of plastics (bisphenol) is also on the list [16]. Physical activity is associated with a reduced cancer risk, and even though girls and women today are more likely to engage in recreational exercise, for many the ability to be physically active is limited by overwhelming demands of school, work and home life [17]. Personal history of an individual as well as family history has a strong influence on the susceptibility to BC besides other established risk factors [18].
Personal history that matters
• Age: The chances of BC increase as one gets older. The child bearing age is resistant to BC though it is rare.
• Having a BC diagnosed in one breast increases the risk of cancer in the other breast or the chance of an additional cancer in the original breast. Women diagnosed with certain benign (non-cancerous) breast conditions have an increased risk of BC.
• Breast tissue: Women with dense breast tissue (as documented by mammogram) have a higher risk for BC development.
• Menstruation: Early menarche (before 12) or late menopause (after 55) increases the risk.
• Exposure to previous chest radiation or use of diethylstilboestrol is identified as a risk factor.
• Nulliparity or the first child after age 30 increases the risk of BC.
• Breastfeeding for one and a half to two years might slightly lower the risk of breast cancer.
• Obesity increases the risk of developing BC both in pre- and postmenopausal women but at different rates.
• Use of oral contraceptives in the previous 2 decades increases the risk of BC slightly.
• HRT is a risk factor for BC.
• A recent meta-analysis reviewing the research on alcohol use and breast cancer concluded that all levels of alcohol use are associated with an increased risk for BC. This includes even light drinking.
• Exercise seems to lower the risk of BC.
Family history
• Race: White women have a higher risk of developing the disease, but African-American women tend to have more tumours that are aggressive when they do develop BC.
• Having a close relative with the disease (sister, mother, and daughter) doubles a woman's risk.
• Genetic risk factors: The most common causes are mutations in the BRCA1 and BRCA2 genes (breast cancer and ovarian cancer genes). Inheriting a mutated gene from a parent means that one has a significantly higher risk of developing breast cancer.
Types of breast cancer
There are many types of breast cancer. Some are more common than others, and there are also combinations of cancers [19]:
• Ductal carcinoma in situ: Non-invasive BC is ductal carcinoma in situ (DCIS) is common.
• The most common type of Invasive ductal carcinoma: Cancer starts in the milk ducts of the breast and grows into other parts of the surrounding tissue. About 80% of invasive breast cancers are invasive ductal carcinoma.
• Invasive lobular carcinoma: This disease starts in the milk-producing glands of the breast. Approximately 10% of invasive breast cancers are invasive lobular carcinoma.
• Mucinous carcinoma is formed from mucus-producing cancer cells. Mixed tumours contain a variety of cell types.
• Medullary carcinoma is an infiltrating BC that presents with well-demarked boundaries between the cancerous and noncancerous tissue.
• Inflammatory BC: This cancer makes the skin of the breast appear red and feel warm (giving it the appearance of an infection). These changes are due to the blockage of lymph vessels by cancer cells.
• Triple-negative BC: This is a subtype of invasive cancer with cells that lack oestrogen and progesterone receptors and have no excess of a specific protein (HER2) on their surface. It tends to appear more often in younger women and African-American women.
• Paget's disease of the nipple: This cancer starts in the ducts of the breast and spreads to the nipple and the area surrounding the nipple. It usually presents with crusting and redness around the nipple.
• Adenoid cystic carcinoma: These cancers have both glandular and cystic features. They tend not to spread aggressively and have a good prognosis.
• Lobular carcinoma in situ: This is not a cancer but an area of abnormal cell growth. This pre-cancer can increase the risk of invasive BC later in life.
• Papillary carcinoma, Phyllodes tumour, Angiosarcoma, Tubular carcinoma are uncommon types of BC.
BREAST CANCER SCREENING METHODS
Globally, BC is a major reason for female mortality. Limitations of current clinical imaging to detect the disease in an earlier stage, researchers are encouraged to explore alternative and complementary tool efficiency. Earlier randomized trials have shown that mammographic screening of all women who are between 50 and 70 years of age can reduce mortality from BC by about 25 percent which is proven not true now [20,21]. Although these findings were recently disputed, there is a consensus among clinicians that breast-cancer screening of women in this age group is effective. On the other hand, there is no consensus about the value of BC screening among women who are 40 to 49 years old, one reason being the difficulty in detecting tumours by mammographic screening in younger women, who have denser breasts than postmenopausal women. It is not clear why some women have a lot of dense breast tissue and others do not, nevertheless at younger age breast tissue is denser and tends to become less dense on ageing. Breast density is divided into 4 levels based upon the amount of glandular tissue and stroma compared to the amount of fatty tissue. Fatty breasts have a protective effect on breast cancer risk. Dense breasts make it more difficult to detect breast cancer on mammograms [22].
Self-examinations (BSES)
Breast cancer cells have to divide 30 times before it can be felt as new identity in the breast. Up to the 28th cell division, cannot be detected by hand and with most breast cancers each division takes one to two months, so by the time it is felt as a cancerous lump, the cancer has been in the body for two to five years. Unfortunately, scientific studies designed to measure the efficacy of BSEs have not found that women who perform BSEs are any less likely to die of breast cancer than women who do not perform them.
Examination by a health care provider
A clinical breast examination (CBE) is a physical examination of the breasts by a trained health care provider which is happens during a routine annual check-up. There is no evidence that CBE alone reduces breast cancer death rates, since there have been no trials that have looked at the use of only CBE, without mammography. If a lump is discovered, better to seek a clinical breast examination; further diagnostic testing is essential [23]. The feel of a breast lump depends on its cause, location, and growth. They can vary greatly from painful, hard, and immobile to soft, painless, and easily moveable. According to BreastCancer.org, lumps are most likely to be cancerous if they do not cause pain, are hard, unevenly shaped, and immobile.
Mammography
Mammograms is an x-ray picture of the breast, a screening tool to detect early BC without symptoms as well as diagnose breast disease such as a lump, pain, skin dimpling or nipple discharge. This screening tool initiated in the 1980s accounted for much of the increase in BC diagnosis in the subsequent years following. Mammograms can miss more than 25 percent of all breast cancers, known as “false negatives” or “false positive” results can occur which on research has shown that as many as 75 percent of all post-mammogram biopsy results turn out to be benign lesions [20-22]. Everyone experiences mammograms differently; only a few may feel pain during the procedure. Most women feel some discomfort during the actual X-ray process due to the pressure against the breasts from the testing equipment. Digital images can be enlarged and the contrast adjusted, enabling radiologists to concentrate on suspicious areas to detect tumours even in dense breast tissue. Digital images can also be stored and transmitted electronically, making it easier to consult with experts at a distance. In digital mammography the X-ray film is replaced by solid-state detectors that convert X-rays into electrical signals and the images can be obtained on computer screen, Women with dense breast tissue are benefitted since in digital technique the, glandular tissue appears white, while fatty tissue appears grey. Digital mammography is a highly accurate tool for breast cancer detection, having a positive predictive value of 89%.
In general, regular mammograms are not recommended for women who are at average risk for BC if they are under 40 years old, since breast tissue tends to be denser in young women. Moreover, there is a fear to the negative effect of radiation. In addition, women who are exposed to radiation for other purposes or women who are carriers of the BRCA (breast cancer susceptibility) gene are at an even higher risk of experiencing adverse effects of radiation [20,21].
Guidelines for mammography differ depending on the organization making recommendations. According to, the American Cancer Society yearly mammograms for women aged 45-54 for women at average risk for BC and once in two years for women aged 55 and older, who should also have the option to continue yearly screening is recommended.
For women under age 50, women who are premenopausal or perimenopausal, and women who have dense breasts, digital mammography may work better, but for most women over age 50, the use of digital mammography does not seem to catch cancers earlier or improve outcomes.
New data documented that for a woman beginning annual mammographic screening at age 50 and continuing until age 75 the benefit exceeds the radiation risk by a factor of almost 100%. Even for a woman who begins annual screening at age 35 and continues until age 75, the benefit of reduced mortality is projected to exceed the radiation risk by a factor of more than 25%.
Ultrasound (Sonography)
This technique excels in distinguishing solids from liquids, so differentiating solid tumours from fluid-filled cysts, which are benign, to decide becomes easy. Evaluation of abnormalities that appear on regular mammograms and to guide needle biopsies is possible. According to Johns Hopkins Medicine, a breast ultrasound is not recommended as the only screening method as is does not always detect early signs of cancer, such as microcalcifications (tiny calcium deposits) which can be a sign of breast cancer. While an ultrasound often is used in conjunction with a mammogram to further test abnormalities is a good combo. Automated whole-breast ultrasound devices have been proved significantly to improve cancer detection in women with dense breasts, compared with mammography alone [24-26].
Magnetic Resonance Imaging (MRI)
MRI is quite effective in detecting invasive breast cancer, but differentiation between benign and malignant lesions is not possible. Breast MRI is useful for women at high risk group for developing the disease and rarely recommended to screen with an average risk of BC [27]. As in sonography calcium deposits cannot be detected in MRI too [24].
Positron Emission Mammography (PEM)
PEM uses gamma rays to detect “hot spots” of rapidly growing cells. A computer analyses the image to determine the size, shape and location of the mass. PEM is a supporting document in addition to mammography to identify small invasive cancers and ductal carcinoma in situ; cancer that is confined to the milk ducts [20,28].
Specific Gamma Imaging (BSGI)
Breast-specific gamma imaging employs a radioactive tracer to identify cancer cells. Like PEM, it is used along with mammography and not instead of it [29].
Thermography
Thermography is a radiation-free imaging technique that creates a picture based on the temperature differences in the tissue being examined. Rich blood supply and higher metabolic rate (malignant tissue) gives a higher temperature than normal tissue; however, some cancerous cells do not give off additional heat, and some that do, are often located too deep within the tissue to register in the thermography screening. These factors render a high number of both false positive and false negative thermographic readings. At present there is no evidence to support the use of thermography as a screening or diagnostic tool according to FDA for BC screening. Nevertheless, some individuals at a high risk for developing breast cancer may choose to use thermography in conjunction with mammography for maximum detection [30].
Echography
Libertario Demi, Ruud van Sloun and Massimo Mischi [31] have now developed a variant of the echography method that is suitable for breast cancer detection. This method is known as “Dynamic Contrast Specific Ultrasound Tomography”. When echo scanner is put on due to its vibration’s microbubbles are produced in the blood and at the same frequency as the sound produced by the scanner, the image is formed. By capturing the vibration, the bubbles are located. But body tissue also generates harmonics which disturbs the observation. Mischi with Demi investigated and modified to visualisation method by creating difference in timing for production of bubbles and image. By measuring this delay, the air bubbles could be localized without any disturbance because the harmonic generated by the body tissue is not delayed, and is therefore discernible. So, this method is perfectly suited to organs that can be approached from both sides [31].
Microwave Imaging System
Microwave imaging is a science is used for detecting/locating radars, this technique can evaluate hidden or embedded objects in a structure (or media) using electromagnetic (EM) waves (i.e., ~300 MHz-300 GHz). It has remarkable effects on efficiency and gain. For experimental validation, a heterogeneous breast phantom was developed that contains dielectric properties identical to real breast tissues with the inclusion of tumours. The radiated backscattered signals are collected by antenna from the targeted tissue are analysed to reveal the changes in dielectric properties in tissues. The dielectric constants of tumorous cells are higher than that of normal tissues due to their higher water content. The developed UWB (Ultra-Wideband) antenna based MWI has been able to perform the detection of tumorous cells in breast phantom that can pave the way to saving lives [32].
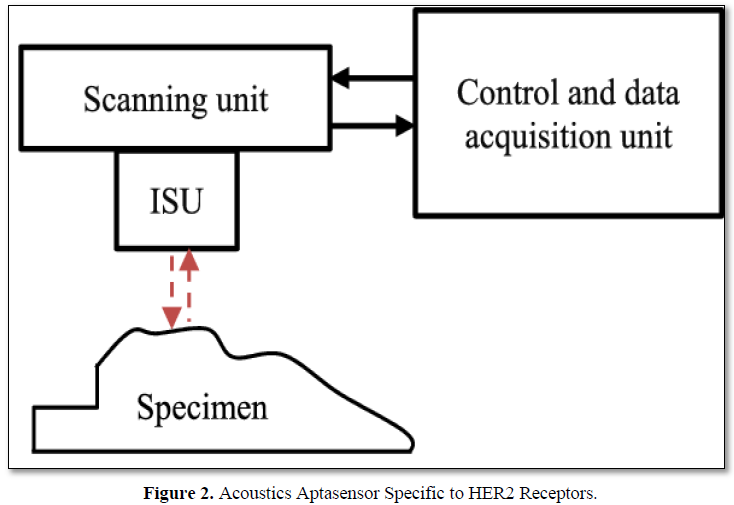
Thick Shear mode acoustics method (TSM) has been applied to detect SK-BR-3 BC cells using DNA aptamers specific to HER2 positive membrane receptors (Figure 2). Biotinylated aptamers were immobilized at the neutravidin layer chemisorbed at gold surface of TSM transducer. Addition of the cells resulted in decrease of resonant frequency fs, and in increase of motional resistance, Rm. Using gold nanoparticles modified by aptamers it was possible to improve the limit of detection that reached 550 cells/mL, while without amplification the sensitivity of the detection of SK-BR-3 cells was 1574 cells/mL. HER2 negative cell line MDA-MB-231 did not resulted in significant changes of fs. The viability studies demonstrated that cells are stable at experimental conditions used during at least 8 h [33]. The viability studies demonstrated that cells are stable at experimental conditions used during at least 8 h. AuNPs were not toxic on the cells up to concentration of 1 μg/mL [34].
Using the approach for BC screening, Optimal risk cut off for two different risk models were estimated: The Breast Cancer Screening Consortium (BCSC) model and a hypothetical model with much better discriminatory accuracy.
STAGING SYSTEM
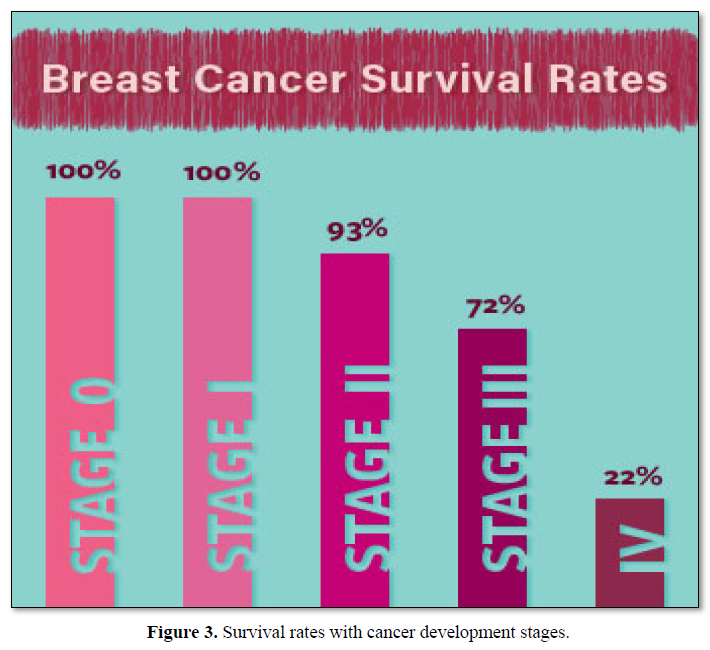
The staging system is essential to determine the most appropriate treatment for the type of cancer after ascertaining the stage of development of BC. Medical professionals developed a new eighth edition of the staging system for 2018 [35] that includes results of testing for certain biomarkers, including the HER2 protein and the results of gene expression assays. The information gained from the imaging tests in addition, uses the results from surgical procedures. After surgery, a pathologist observes the cells from the BC as well as from the lymph nodes. This information gained is incorporated into the staging, as it tends to be more accurate than the physical exam and X-ray findings alone [21]. The survival rate of the patients is ascertained depending up the stage of the disease condition (Figure 3). TNM staging. This system uses letters and numbers to describe certain tumour characteristics in a uniform manner.
T: This describes the size of the tumour. A number from 0 to 4 follows. Higher numbers indicate a larger tumour or greater spread:
· TX: Primary tumour cannot be assessed
· T0: No evidence of primary tumour
· Tis: Carcinoma in situ
· T1: Tumour is 2 cm or less across
· T2: Tumour is 2 cm-5 cm
· T3: Tumour is more than 5 cm
· T4: Tumour of any size growing into the chest wall or skin
· N: represents the spread to lymph node near the breast. A number from 0 to 3 follows.
· NX: Nearby lymph nodes cannot be assessed (for example if they have previously been removed).
· N0: There has been no spread to nearby lymph nodes. In addition to the numbers, this part of the staging is modified by the designation "i+" if the cancer cells are only seen by immunohistochemistry (a special stain) and "mol+" if the cancer could only be found using PCR (special detection technique to detect cancer at the molecular level).
· N1: Cancer spreads to one to three axillary lymph nodes (underarm lymph nodes) or medical professionals find tiny amounts of cancer in internal mammary lymph nodes (lymph nodes near breastbone).
· N2: Cancer has spread to four to nine axillary lymph nodes or the cancer has enlarged the internal mammary lymph nodes.
· N3: Any of the conditions below
· Cancer has spread to 10 or more axillary lymph nodes with at least one cancer spread larger than 2 mm.
· Cancer has spread to lymph nodes under the clavicle with at least area of cancer spread greater than 2 mm.
· M: This letter is followed by a 0 or 1, indicating whether the cancer has spread to other organs.
· MX: Medical professionals cannot assess metastasis.
· M0: Health care providers find no distant spread on imaging procedures or by physical exam.
· M1: Spread to other organs is present.
Once the T, N and M categories have been determined, physicians combine them into staging groups. There are five major staging groups, stage 0 to stage IV, which are subdivided into A and B, or A and B and C, depending on the underlying cancer and the T, N, and M scale.
The recently developed imaging methods [29-34] which are fast and accurate in comparison to even nanotechnological methods [36] have brought revolution in diagnostics of BC. In a series of research papers, we have suggested that the BC can be prevented/ treated with epigenetic factors such as light, food, exercise, life style (regulating biological clock), sleep, etc. [36-39].
CONCLUSION
Data on mammography and MRI test score distributions were drawn from the Magnetic Resonance Imaging Screening Study Group. In the near future, as less invasive, more effective approaches are sought for early diagnosis and treatment of the disease. Newer imaging technologies that look at breast cancer at the cellular level may become more widely used if there is clear evidence of their effectiveness as screening tools. Treatment of breast cancer depends on the type of cancer and its stage (0-IV) and may involve surgery, radiation, or chemotherapy.
1. Pushkala K, Gupta PD (2009) Prevalence of breast cancer in menopausal blind Women. Int J Med Medic Sci 1 : 425-431.
2. Gupta PD, Pushkala K (2019) Menarche: The essential event for motherhood. J Age Resto Med 2 : 84.
3. Gupta PD (2010) Mothering a cause. Oxford & IBH Publications Pvt. Ltd. New Delhi.
4. Harbeck N, Penault-Llorca F, Cortes J, Gnant M, Houssami N, et al. (2019) Breast cancer. Nat Rev Dis Primers 5 : 66.
5. Johnson KC, Houseman EA, King JE, Christensen BC (2017) Normal breast tissue DNA methylation differences at regulatory elements are associated with the cancer risk factor age. Breast Cancer Res 19 : 81.
6. Tulinius H, Sigvaldason H, Olafsdóttir G (1990) Left and right sided breast cancer. Pathol Res Pract 186 : 92-94.
7. Parthasarathy V, Rathnam U (2012) Nipple discharge: An early warning sign of breast cancer. Int J Prev Med 3 : 810-814.
8. Dupont WD, Page DL (1985) Risk factors for breast cancer in women with proliferative breast disease. N Engl J Med 312 : 146-151.
9. Pushkala K, Gupta PD (2011) Dark side of the night light (Monograph). First edition. Germany. LAMBERT Academic Publishing, GmbH & Co. KG, Saarbrücken, Germany.
10. Gupta PD, Usha N, Pushkala K (2010) Dark side of the night light : Implication in breast cancer. J Cell Tissue Res 10 : 2173-2184.
11. Gupta PD, Pushkala K (2016) Increased incidence of breast cancer due to long exposure of light. J Analy Oncol 5 : 146-152.
12. Pushkala K, Gupta PD (2016) Light and breast cancer : Is there any relationship. BAOJ Cancer Res Ther 2 : 26.
13. Pushkala K, Gupta PD (2017) Missing dark in modern life aids in developing breast cancer. J Sci Discov 1 : jsd17003.
14. Zeinomar N, Knight JA, Jeanine M, Genkinger JM, Phillips K, et al. (2019) Alcohol consumption, cigarette smoking, and familial breast cancer risk: Findings from the prospective family study cohort (ProF-SC). Breast Cancer Res 21 : 128.
15. Agurs-Collins T, Ross SA, Dunn BK (2019) The many faces of obesity and its influence on breast cancer risk. Front Oncol 9 : 765.
16. Gupta PD, Pushkala K (2019) Increasing woman’s health concern due to xenoestrogens and parabens: A review. J Cell Tissue Res 19 : 6829-6832.
17. Niehoff NM, Nichols HB, Zhao S, White AJ, Sandler DP (2019) Adult physical activity and breast cancer risk in women with a family history of breast cancer. Cancer Epidemiol Biomarkers Prev 28 : 51-58.
18. Katz Jessica (2020) Breast Cancer Risk Factors. Available online at : https//CDC govt/genomics/diseases/breast/ovarian-cancer/
19. Sharma GN, Dave R, Sanadya J, Sharma P, Sharma KK (2010) Various types and management of breast cancer : An overview. J Adv Pharm Technol Res 1 : 109-126.
20. Ballal HS, Gupta PD (2009) Peeping inside the body. Capricorn Publishing House, Jaipur.
21. Gupta PD (2019) Mammography: Boon or Bane. J Women Health Care Issues 2.
22. Ingrid S (2009) Dense breast tissue as an important risk factor for breast cancer and implications for early detection. Breast Care (Basel) 4 : 89-92.
23. Provencher L, Hogue JC, Desbiens C, Poirier B, Poirier E, et al. (2016) Is clinical breast examination important for breast cancer detection? Curr Oncol 23 : e332-e339.
24. Gokhale S (2009) Ultrasound characterization of breast masses. Indian J Radiol Imaging 19 : 242-247.
25. Van Sloun R, Pandharipande A, Mischi M, Demi L (2015) Compressed sensing for ultrasound computed tomography. IEEE Trans Biomed Eng 62 : 1660-1664.
26. SG Schalk, J Huang, J Li, L Demi, H Wijkstra, et al. (2018) 3-D quantitative dynamic contrast ultrasound for prostate cancer localization. Ultrasound Med Biol 44 : 807-814.
27. Radhakrishna S, Agarwal S, Parikh PM, Kaur K, Panwar S, et al. (2018) Role of magnetic resonance imaging in breast cancer management. South Asian J Cancer 7 : 69-71.
28. Berg WA, Weinberg IN, Narayanan D, Lobrano ME, Ross E, et al. (2006) High-resolution fluorodeoxyglucose positron emission tomography with compression (“positron emission mammography”) is highly accurate in depicting primary breast cancer. Breast J 12 : 309-323.
29. Brem RF, Floerke AC, Rapelyea JA, Teal C, Kelly T, et al. (2008) Breast-specific Gamma Imaging as an adjunct imaging modality for the diagnosis of breast cancer. Radiology 247 : 651-657.
30. Acharya UR, Ng EY, Tan JH, Sree SV (2012) Thermography based breast cancer detection using texture features and support vector machine. J Med Syst 36 : 1503-1510.
31. Demi L, Sloun R van, Mischi M (2016) Towards dynamic contrast specific ultrasound tomography. Sci Rep 6 : 34458.
32. Kwon S, Lee S (2016) Recent advances in microwave imaging for breast cancer detection. Int J Biomed Imaging 2016 : 5054912.
33. Poturnayová A, Dzubinová L, Buríková M, Bízik J, Hianik T (2019) Detection of breast cancer cells using acoustics aptasensor specific to HER2 receptors. Biosensors (Basel) 9 : 72.
34. Kalli S, Semine A, Cohen S, Naber SP, Makim SS, et al. (2018) American Joint Committee on Cancer’s Staging System for Breast Cancer, Eighth Edition : What the radiologist needs to know. Radiographics 38 : 1921-1933.
35. V Jaishree, Gupta PD (2012) Nanotechnology: A revolution in cancer diagnosis. Indian J Clin Biochem 27 : 214-220.
36. Gupta PD (2019) Nanotechnological Approaches for Cancer Diagnosis and Treatment: A Review. J Cell Tissue Res 19 : 6613-6626.
37. Gupta PD, Pushkala K (2012) Prevention and treatment of breast cancer by light and food in : Natural products and their active compounds on disease prevention. Chapter 17. Editors : Essa MM, Manickavasagan A, Sukumar E. Nova Science Publishers. C.G.C Press USA, 153-159.
38. Pushkala K, Gupta PD (2013) Epigenetic effect of food for cancer management. Int J Med Sci Biotech 1 : 1-11.
39. Gupta PD, Pushkala K (2020) Cancer management through epigenetics. J Cell Tissue Res 20 : 6847-6852.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Otolaryngology and Neurotology Research(ISSN:2641-6956)
- International Journal of Diabetes (ISSN: 2644-3031)
- Journal of Oral Health and Dentistry (ISSN: 2638-499X)
- Journal of Psychiatry and Psychology Research (ISSN:2640-6136)
- BioMed Research Journal (ISSN:2578-8892)
- Journal of Neurosurgery Imaging and Techniques (ISSN:2473-1943)
- Journal of Carcinogenesis and Mutagenesis Research (ISSN: 2643-0541)