9
Views & Citations10
Likes & Shares
In that study, the lipid moiety of this conjugate was found to correspond to monolaurin. When adenosine-monolaurin (A-M) compounds were prepared, artificial cells could be produced (4). Subsequent analysis revealed that the outer membrane of artificial cells are coated with DNA, resulting in the designation of these cells as “DNA Crown Cells” in 2016 (4).
DNA crown cells, which have been shown to develop into fully self-replicating cells after incubation in egg white of artificial cell seeds or synthetic DNA crown cells which can be prepared with sphingosine, DNA, adenosine, and monolaurin, all of which are commercially available compounds. A comprehensive procedure for generating DNA crown cells with synthetic DNA crown cells was reported in 2017 (5), and numerous DNA crown cells and associated cell strains have been produced to date (6–14). These cells have been prepared and stored stably at 4°C for nearly a decade.
When cultured in agar with egg white, monolaurin, salt or microorganisms, synthesized DNA crown cells formed assemblies or diverse morphological objects (15–20).
Antibiotic-producing cells were also isolate from beer produced using co-cultures of DNA crown cells and yeast (21). Based on these findings, egg white powder enclosing different DNA crown cells in conjunction with different substances has been prepared, and numerous antibiotic-producing cells have been produced using agar cultures of these powders (22–27). Such a culture system (i.e., using egg white powder) can be regarded as being equivalent to an egg-white-based culture system, which obviates the need to use eggs for culture.
Despise these extensive observations, the mechanism by which such self-replicating antibiotic-producing cells arise remains unknown.
Therefore, to better understand the emergence of self-replicating antibiotic-producing cells, this study examined whether gas production occurs when egg white powder enclosing DNA (bovine) crown cells with a blueberry extract is cultured.
MATERIALS AND METHODS
Materials
DNA (bovine meat) crown cells that had been prepared previously and stored in a refrigerator at approximately 4°C were used in this study (5, 22). However, the methods are described here again for clarity.
The materials used in the present study were the same as those employed in previous studies (5): Sph (Tokyo Kasei, Japan), DNA (from bovine meat), adenosine (Sigma-Aldrich; Wako, Japan), monolaurin (Tokyo Kasei), and adenosine-monolaurin (A-M), a compound synthesized from a mixture of adenosine and monolaurin (5). Sorbent tubes (SIBATA, Japan). Monolaurin solutions were prepared to a final concentration of 0.1 M in distilled water. Agar plates; agar medium (SMA) (AS ONE, Japan). Blueberries were obtained from a local market.
Gas chromatography-mass spectrometry (GC-MS) and gas chromatography (GC) analyses were performed using a GCMS-TQ8030, a GC-2010 Plus and NIST11 Mass Spectral Libray, respectively (Shimazu, Japan).
Methods
Preparation of DNA (bovine meat) crown cells (6, 22).
Step 1 A total of 180 µL of Sph (10 mM) and 50 µL of DNA (1.7 µg/µL) were combined, and the mixture was heated and cooled twice.
Step 2. A-M solution (100 µL) was added and the mixture was incubated at 37°C for 15 min.
Step 3. A total of 30 µL of monolaurin solution was added, and the mixture was incubated at 37°C for another 5 min.
Step 5 Then, 0.3–0.5 mL of the suspension was injected into egg white and incubated for 7 days at 37°C. Egg white was subsequently recovered and used as DNA (bovine meat) crown cells.
Preparation of blueberry extract
Berries (~50 grains) were ground using a mortar and pestle and suspended in 3 mL of distilled water.
Preparation of powder
- First, 3 mL of blueberry extract was mixed with 3 mL of egg white.
- The mixture was incubated for 5 h at 37°C.
- Then, approximately 25 mL of fresh egg white was added to the mixture.
- The fluid was poured into two Petri dishes and dried for 1–2 days at 37°
- Dried materials were collected and powder was prepared using a mortar and pestle.
- The powder (Fig. 1), named Crown-Bovine-meat-blueberry-ex-P, was stored at room temperature until use.
Preparation of samples for GC-MS/GC analysis
A small amount (~40–50 mg) of powder (Crown-Bovine-meat-blueberry-ex-P) was added to an agar plate and incubated for 2 days at 37°C. Approximately 1.5 mL of 0.1 M monolaurin was dispensed onto a plate, together with two gas assay tubes, and the plate was sealed in a bag. The samples were then incubated for 2 days at 37°C. After incubation, the tubes were sealed and stored in a freezer until gas detection
Gas detection
- The tube was cut open, and the activated carbon was removed. It was transferred to a new brown bottle
- Then, 1 mL of carbon disulfide was added to the bottle and briefly shaken.
- After 2 h, the gasses had been extracted.
- The sample was subjected to GC-MS analysis.
Antibiotic-producing cells:
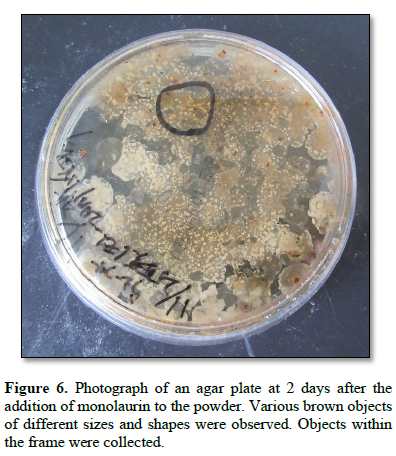
Objects within the frame shown in Fig. 6 that were grown on the plate were suspended in approximately 1 mL of distilled water. Then, approximately 200 μL of the suspension was cultured on an agar plate at 37°C for 1 day.
General observations
Objects on the plates were observed directly with the naked eye and under a light microscope.
RESULTS AND DISCUSSION

Figure 2 shows a photograph of an agar plate immediately after the start of culture with the powder (Crown-Bovine-meat-blueberry-ex-P). Powder particles of various sizes were observed to cover the entire Petri dish.

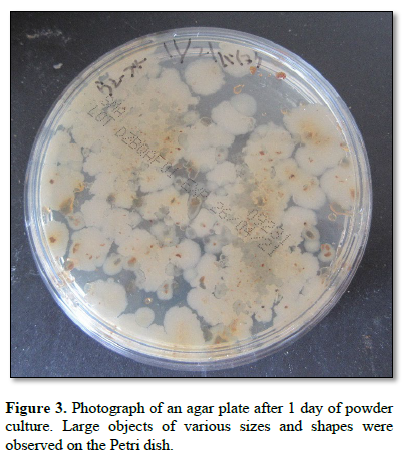
Figure 3 shows a photograph of an agar plate after 1 day of powder culture. Large objects of various sizes and shapes were observed on the Petri dish.
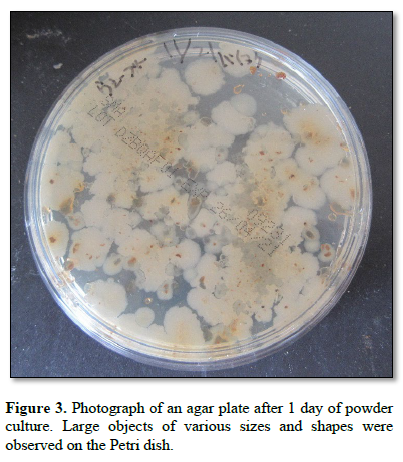
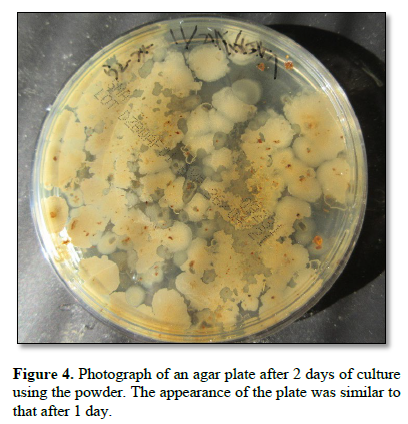
Figure 4 shows a photograph of an agar plate after 2 days of culture using the powder. The appearance was similar to that after 1 day.
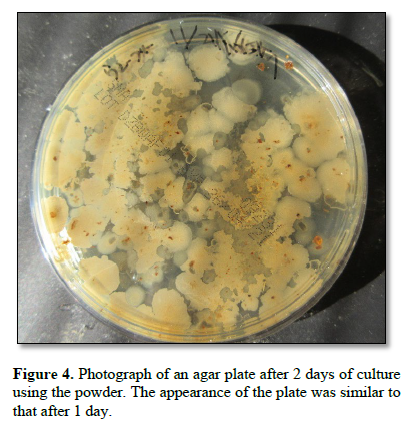
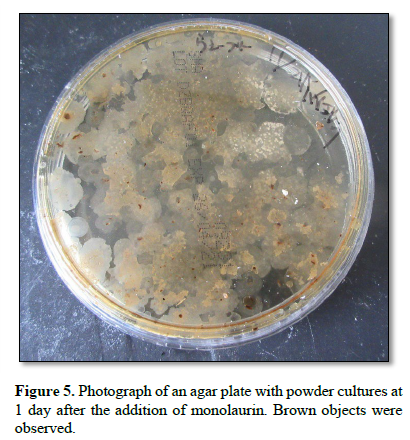
Figure 5 shows a photograph of an agar plate with powder cultures at 1 day after the addition of monolaurin. Brown objects were observed.
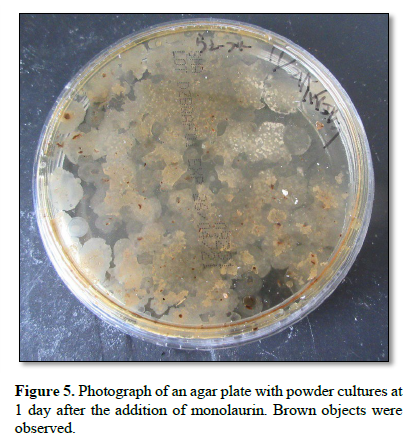
Figure 6 shows a photograph of an agar plate at 2 days after the addition of monolaurin to the powder. Various brown objects of different sizes and shapes were observed. Objects within the frame were collected.
Figure 7 shows the microscopic appearance of objects within the frame shown in Figure 6.

Various amorphous objects were observed (Figure 7a). The approximate size of the objects shown in Fig.7b is 120 μm.
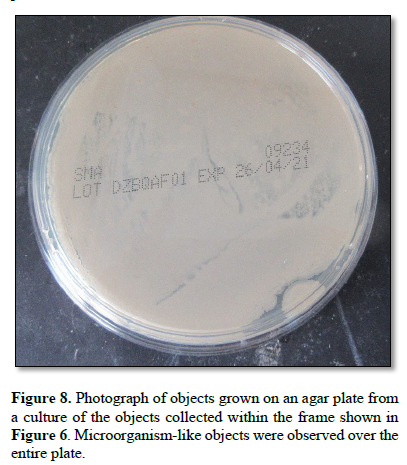
Figure 8 shows the growth obtained on an agar plate after culture of the material within the frame in Figure 6. Microorganism-like objects were observed over the entire plate.
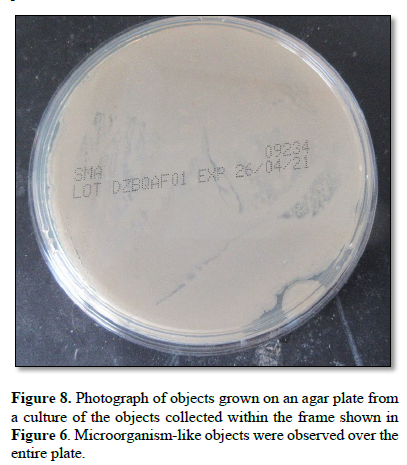
Figure 9 shows a photograph of the experimental setup for the gas assay. After gas collection, the tubes were sealed.

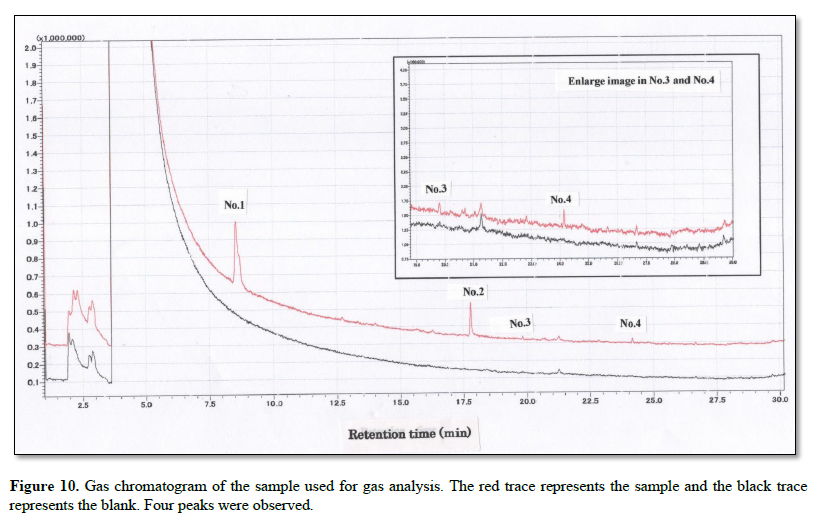
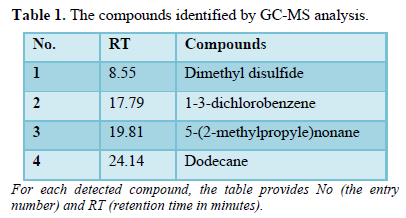
Figure 10 shows a gas chromatogram of the sample used for gas analysis. The red trace represents the sample, and the black trace represents the blank. Four peaks were observed.
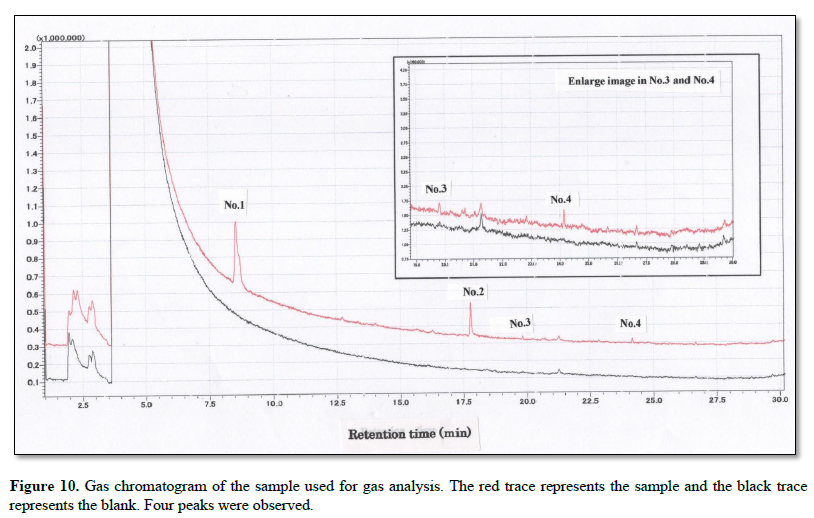
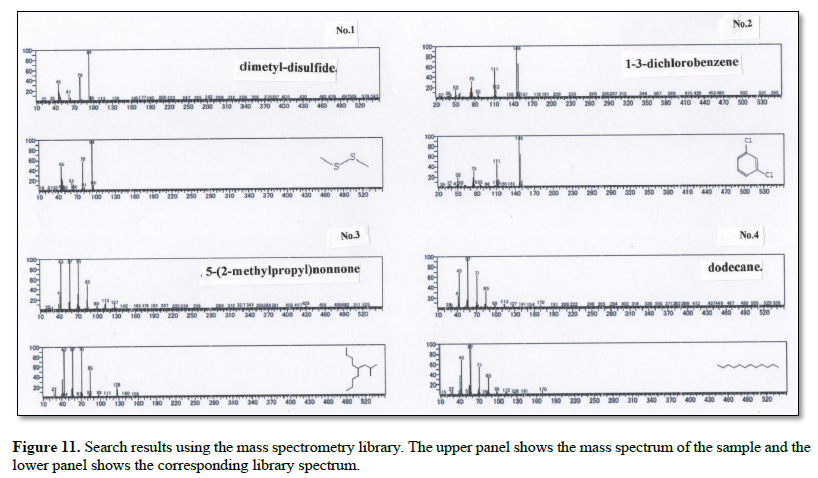
Figure 11 shows the search results obtained using the mass spectrometry library. For each entry (No.), the upper panel shows the mass spectrum of the sample, and the lower panel shows the corresponding library spectrum.
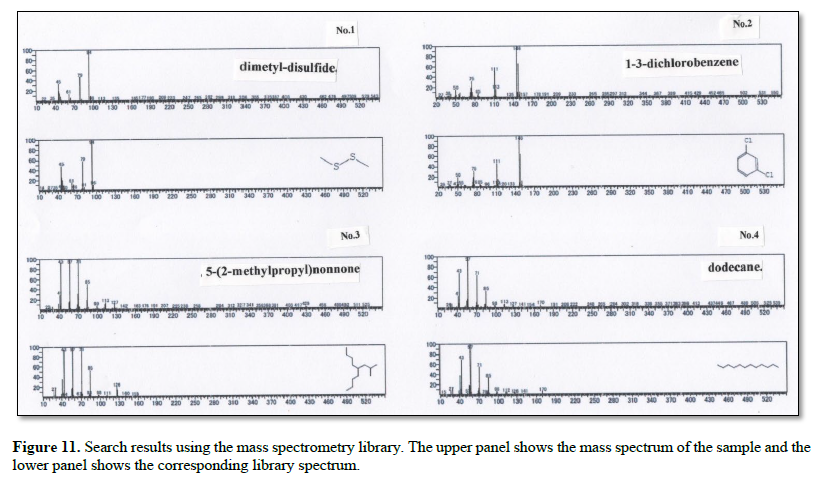
Table 1 summarizes the compounds identified by GC-MS analysis.
In previous studies, it was demonstrated that antibiotic-producing cells were generated through the combination of DNA crown cells and partners (22–27). Importantly, the generation process was found to common across all combinations tested.
At the initial stage of the experiments, the powder was spread onto agar culture medium and the plates were incubated (Figures 2, 3 and 4). Following incubation, monolaurin was added to the culture. Two days after the addition of monolaurin, the developing objects exhibited a slightly brownish coloration (Figures 5 and 6), appearing amorphous under microscopic examination (Figure 7)
When the monolaurin-treated objects were further cultured, numerous self-regenerating antibiotic-producing cells subsequently emerged (Figure 8).
Therefore, the observed increase in objects appear to be stimulated by monolaurin.
However, no conclusions can be drawn at present as slight increases in object number have also been observed in the absence of monolaurin. It is possible that such increases may be attributed to the egg white-derived lipids in powder, some of which may exhibit monolaurin-like activity.
Since the process underlying the generation of antibiotic-producing cells is shared across all combinations of DNA crown cells and partners tested, the use of powder prepared from the combination of DNA crown cells (bovine meat) and blueberry extract in this study is not particular unique. If anything, it merely demonstrates that this specific combination can also be used to produce DNA crown cells.
As noted earlier, numerous antibiotic-producing cells emerged during the culture period following the addition of monolaurin. However, the reason such a phenomenon occurred and the processes involved during the course of cultivation, remain unclear.
To clarify this point, gas production was investigated during cultivation. Gas was collected over two days of cultivation after the addition of monolaurin using the setup shown in Figure 9.
The collected gas was analyzed by GS-MS and the resulting gas chromatograms showed four distinct peaks (Figure 10). The MS spectrum of each distinct peak, along with the corresponding-library search results, are shown in Figure 11. The library results identified the peaks as DMS (No. 1), 5-(2-methylpropyl) nonane (No. 2), 1-3-dichlorobenzene (No. 3), and dodecane (No.4) (Table 1).
How gases were generated and whether they have any relationship to the proliferation of antibiotic-producing cells remains unknown. Nevertheless, the discovery that gas is produced in this type of culture system is considered significant when the nature of the detected gases is considered.
Several of these gasses are commonly associated with biological activity, which raises important questions about the chemical processes occurring within the culture systems.
In particular, DMS is used in a variety of fields, including the food industry
It is also produced by marine microorganisms and is associated with biological processes in sulfur-rich environments.
Moreover, it is considered one of the most significant and promising gases in astrobiology and is regarded as a potential indicator of extraterrestrial life. Indeed, its presence on some planets, has been interpreted as a possible indicator of life.
Likewise, the presence of methyl-containing compounds is often interpreted as an indication of biological or pre-biological chemical processes. Similarly, the gases detected during the generation of antibiotic-producing cells are considered to be suggestive evidence of potential biological activity.
Further studies should clarify whether gas production is directly linked to the emergence of antibiotic-producing cells, to the formation of the unique cellular structures, or to the synthesis of antibiotics themselves.
In addition, it will be important to clarify how these gases are produced and which gases are generated under different combinations of DNA crown cells and partners.
ACKNOWLEDGMENTS
I would like to thank Riho Katagiri, (Materials Analysis Center, JAPAN TESTING LABORATORIES Co. Ltd., Gifu, Japan) for assistance with gas analysis and for useful discussions.
-
Inooka S., Preparation and cultivation of artificial cells. App. Cell Biol.25.15-18.2012. ISSN 1881-0772
-
Inooka S., Investigation of the chemical composition of artificial cell seeds-Sphingosine-DNA bound components from extracts of the meat from adult Ascidiara, LICRLS 534-540, 2016
-
Inooka S., Preparation of Artificial Cells Using Eggs with Sphingoshine-DNA. J. Chem. Eng. Process Technol. 17. 277. 2016. Doi.1110.4172/2157-77048.1000277
-
Inooka S., Aggregation of sphingosine-DNA and cell construction using components from egg white. Integrative Molecular Medicine. 3 (6). 1-5, 2016. doi 10.157761/imm.1000256
-
Inooka S., Systematic Preparation of Artificial Cells (DNA Crown Cells). J. Chem. Eng. Process Technol.,8,2017. Doi.104172/2157-2048.100027
-
Inooka S., Systematic Preparation of Bovine meat DNA Crown Cells. App Cell Biol. Japan 30 13-16, 2017
-
Inooka S., Preparation of Artificial Cells for Yogurt Production. App Cell Biol., 26,13-17,2013
-
Inooka S., Preparation of Artificial Placental Cells. App Cell Biol.,27, 4-49, 2014
-
Inooka S., Prepartion of DNA (Nannochloropsis species) crown cells (artificial cells) using eggs, sphingoshin, and DNA, and subsequent cell recovery. App Cell Biol., 32, 55-64, 2019, ISSN: 2433-5800
-
Inooka S., Systematic Preparation of Generated DNA (Akoya pear oyster) Crown Cells.App Cell Biol. Japan 31, 21-34, 2018, ISSN: 2433-5800
-
Inooka S., Preparation of a DNA (E. coli) Crown Cell line in Vitro-Microscopic Appearance of Cells. Annals of Reviews & Research 2022.8(1) ARR.MS.ID.555728. DOI.10.19080/ARR.2022.08.555726
-
Inooka S., Preparation and Microscopic Appearance of a DNA (Human Placenta) Crown Cell Line. Journal of Biotechnology & Bioresearch 5(1). JBB 000603. DOI:10.1031/JBB.2023.05.000603
-
Inooka S. Preparation of a DNA (Akoya pearl oyster) crown cell line. Applied Cell Biology Japan. 36.2023. ISSN 2033-5800
-
Inooka S. Preparation of a DNA (HepG2 is a hepatoblastorma-derived cell line) Crown Cell Line. Journal of Tumor Medicine & Prevention. 2023.4(2) ISSN 2575-89OX. DOI 10.19080/JTMP.2023.04.555675016/55760
-
Inooka S., Microscopic observation of assemblies formed in mixtures of DNA (Human placenta) crown cells with Bacillus subtilis. An archive of organic and inorganic science 2021. 2637-46-09
-
Inooka S., Assembly Formation of Synthetic DNA (E. coli) crown cells with Salt “Reconstruction and Regeneration of Synthetic DNA Crown Cells”. American Journal of Biomedical Science. 2020 Vol.16 ISSN 2642-1747
-
Inooka S., Cell Proliferation from the Assembly of Synthetic DNA (E. coli) Crown Cells with Stimulation by Monolaurin Twice. Current Trends on Biotechnology & Microbiology. 2022. DOI.10.2474/CTBM2022.02.000149
-
Inooka S. Microscopic appearance of Cell Assembly and Proliferation in Agar Cultures of Synthetic DNA (E. coli) Crown Cells with Monolaurin Chemical & Pharmaceutical Research 2024, 6(1), 1-4, ISSN 2684-1050
-
Inooka S., Synthesis and Microscopic Appearance of Thalus-like Objects tin Synthetic DNA (E. coli) Crown Cells Created Using Monolaurin and Egg white. Open Access Journal of Reproductive System and Sexual Disorder. 3(2), 2024. DOI;10.32474/OAIRSD.2024.03.000157
-
Inooka S., Double Cell-Like Objects Created from synthetic DNA (Human Placenta) Crown Cells using Monolaurin. American. Journal of Medical and Clinical Science Vol.9 No.2. 1-3,2024
-
Inooka S. Separation of Antibiotic-Producing Cells from Beer Produced in Co-cultures of DNA (Streptomyces) Crown Cells with Yeast. Annals of Reviews and Research 12(3) ARR.MS.ID. 555839 (2025)
-
Inooka S., Isolation of Antibiotic Producing Cells from Plate Culture of Egg Powder Enclosing DNA (Bovine Meat) Crown Cells and Beef Extract. Annals of Reviews and Research 12 (5) ARR. MS.ID 555850 (2025)
-
Inooka S., Separation of antibiotic producing cells from culture fluids of egg white powder enclosed DNA (Streptomyces) crown cells and yeast. International Journal of Bioprocess & Biotechnological Advancements. 10(1) 499-504, 2025. ISSN: 2380-0259
-
Inooka S., Separation of Antibiotic-Producing Cells from Agar Cultures of Egg Powder-Enclosed DNA (hepato-blastoma cell Line: HepG2) Crown Cell and Yeast. American Journal of Biomedical Science & Research 2025 26(2). DOI;10.34297/AJBSR 2025.26.003417. ISSN: 2642-1747
-
Inooka S., Isolation of Antibiotic-Producing Cells from Cultures of Egg Cultures of Egg Powder Enclosing DNA (Hepg2) Crown Cells and Salmon Roe. Trends in General Medicine 2025, 3(1):1-5, ISSN:2996-3893
-
Inooka S., Antibiotic-Producing Cells Produced from A Plate Culture of Egg Powder Enclosing DNA (Nannochloropsis Species) Crown Cells and Yeast Scientific Journal Biology & Life Sciences Vol.2, 2 (2025). ISSN 2694-166X, DOI;10.3552/SJBLS.2025.03.000582
-
Inooka S., Antibiotic-Producing Cells in Milk Cultures of Egg Powder-Enclosed DNA (Ascidians Sea Squirt) Crown Cells Cultured with Peptide (Glu-Glu) Partners. International Journal of Bioprocess & Biotechnological Advancements 10(1), 505-511, 2025. ISSN: 2380-0259