2811
Views & Citations1811
Likes & Shares
Gastric cancer (GC) is currently the third leading cause of cancer-related death worldwide. The precise etiology for GC is still obscure. Helicobacter pylori (H. pylori) infection is well-established to be a cause for GC. Obesity is another risk factor. Here, we briefly reviewed the research progression on the crosstalk of H. pylori infection and obesity in induction of gastric cancer and discussed the potential molecular mechanism.
Keywords: Helicobacter pylori, Obesity, Inflammation, Metabolic abnormality, Gastric cancer
INTRODUCTION
GC is closely associated with H. pylori-induced chronic gastritis[1,2]. Epidemiological studies show that persistent H. pylori infection accounts for approximate 75% of confound risk factors for GC [3]. Obesity contributes to tumor development including GC, possibly due to its induction of low systemic inflammation and metabolic abnormality [4,5]. Eradication of H. pylori reduces GC risk by approximately 3-fold. It is interesting that H. pylori infection is reported to be associated with obesity, insulin resistance and metabolic syndrome, which suggests the cooperation of H. pylori and obesity in GC induction [6-8].
H. pylori Infection and GC
H. pylori infects nearly half the world’s population, the infection rate can be even higher than 90% in some developing countries. The bacterium colonizes in the stomach and is usually a persistent infection for the whole lifetime. It causes chronic gastritis and peptic ulcer and is closely associated with the development of GC [1,2].
The clinical consequence of H. pylori infection is determined by the bacterial products and their interaction with host factors. The pathogenicity of H. pylori is primarily mediated by CagA (cytotoxin-associated gene A) and VacA (vacuolating cytotoxin gene) proteins, both of which can trigger severe gastric lesions, causing DNA damage and somatic mutations [9]. These bacterial products can also strongly induce host immune responses by releasing excessive amounts of pro-inflammatory cytokines, including interleukin-8 (IL-8) and IL-1[10-14], which in turn recruit and activate dendritic cells (DCs) and CD4 T cells including Th1, Th2, Th17 and Treg cells [15,16]. A mixed response of Th1 and Th17 cells plays a critical role in H. pylori-induced inflammatory gastric diseases and cancer [17,18]. H. pylorielicits Th1 response to produce interferon-γ and tumor necrosis factor-α (TNFα) causing chronic gastritis andulcers [19]. Th17-derived IL-17A favors angiogenesis and tumor growth through inducing IL-6 that activates STAT3 signaling to promote tumor survival and angiogenesis [20,21]. In contrast, accumulation of Treg and Th2 cells in the infected foci perform anti-inflammatory impacts and allow persistence of the infection and disease progression.
Obesity, Metabolic Deregulation and GC
Obesity is an important public health problem worldwide, which affects more than 300 million people in China. Obesity can lead to a state of chronic low-grade inflammation at multiple sites throughout the body and is mechanistically linked to the metabolic abnormalities with increased incidence of gastric cancer [4,22]. The exact mechanisms responsible for increased incidence of gastric cancer in individuals with obesity are not completely clear. Metabolic endogenous hormones including insulin-like growth factors (IGF-1), ghrelin, and leptin, as well as the incremental pro-inflammatory cytokines such as IL-6 and TNFα may all play a role [4].
Obesity often leads to insulin resistance which reduces IGF binding protein (IGFBP) expression and increases free IGF-1 [23]. IGF-1 plays a vital role in mediating the effects of the growth hormone, which may promote cell growth [24,25]. Leptin is a peptide hormone produced and secreted by the white adipose tissues. Obesity induces abundant leptin production which might be involved in the development of some cancers [26,27]. Ghrelin is a hormone produced by the fundic glands of the stomach and play important roles in gastric cancer [28,29]. Obesity influence ghrelin level and signaling which involved in GC progression [30]. On the other hand, obesity is associated with a chronic systemic inflammation with a mount macrophage accumulated in adipose tissue. Adipose inflammation increases some pro-inflammatory cytokines such as TNFα, IL-6 and monocyte chemo attractant protein1 (MCP-1) [31-33], most of these cytokines are considered oncogenic [34,35].
The Synergistic Impact of H. pylori infection and obesity on GC Development
There is growing evidence for a potential association between H. pylori infection and obesity. Epidemic studies showed a higher H. pylori prevalence in obese patients give a clue that obesity influence H. pylori infection [36-38]. H. pylori infection can remotely trigger obesity-associated adipose inflammation and insulin resistance [39,40], while obesity accelerates H. felis-induced gastric carcinogenesis by enhancing immature myeloid cell trafficking and Th17 response in mice [40].
H. pylori infection can induce changes in gastric mucosal leptin and ghrelin levels, which influence body weight changes and obesity [41,42]. In return, obesity linked to metabolic deregulation including hyperinsulinemia, hyperlipidemia and hyperglycemia influences leptin and ghrelin signaling [43]. In addition to abundant leptin induced by obesity, increased leptin expression in gastric tissues has also been found in patients with H. pylori infection and gastric cancer [44]. Leptin may promote cell proliferation by activating JAK/STAT signaling pathway to participate in gastric carcinogenesis [45]. H. pylori induced damage to gastric epithelium may alter the hormonal milieu of the stomach, leading to a decreased production of ghrelin [42]. Reduced ghrelin in circulation may enhance activity of TNFα, IL-6 and other major inflammatory cytokines, which have oncogenic function [28,46].
H. pylori infection and obesity can synergistically cause systemic inflammation. A set of inflammatory cytokines and chemokines including IL-6, resisting, PAI-1, leptin, CCL7, CXCL1, IL-17A, and granulocyte-macrophage colony stimulating factor(GM-CSF) produced by the local (gastric) and distant (adipose) sites of inflammation were detected to be elevated in the serum of H. felis-infected obese mice, which are collectively implicated in metabolic disorders [40]. It is worth noting that our recent study in clinical GC specimens suggests that dysregulated lipid metabolism (a manifestation of obesity) may synergize with H. pylori to promote GC development [47]. In this study, we found that high plasma triglycerides and H. pylori had synergy effect on IL-17A expression. Furthermore, RORγt,a key transcription factor for Th17 differentiation, is shown to be associated with H. pylori-related metabolic disorders and GC progression [47]. In agreement with our finding, H. pylori infection and its related gastroduodenal morbidity with metabolic syndrome was also reported in a large cross-sectional study [48]. Furthermore, both obesity surgery and H. pylori eradication inhibit carcinogenesis [49].
CONCLUSION & PERSPECTIVE
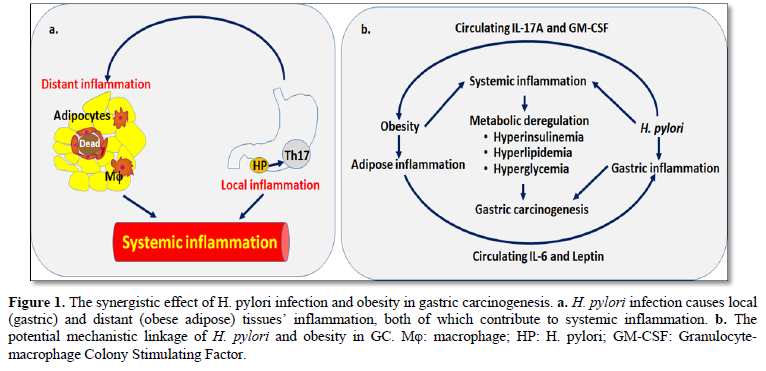
In summary, H. pylori and obesity communicate mutually to influence metabolic processing, inflammation and carcinogenesis. H. pylori infection increases Th17 cells in gastric mucosa causing local inflammation. It is interesting that H. pylori-related Th17 cells in the stomach could exert a remote control of adipose inflammation. Enhanced circulating cytokines and immune cells released from both inflamed gastric and adipose tissues result in systemic inflammation, which collectively cause insulin resistance and metabolic disorders. Adipose-derived adipokines and cytokines like leptin and IL-6 return to H. pylori-infected sites for supporting Th17 expansion and function, thus forming a positive feedback loop for Th17 activation. In this regard, obesity may exacerbate H. pylori-induced gastric lesions and inflammation. Obesity, hyperinsulinemia, and H. pylori synergistically contribute to GC development (Figure 1).
Although the association and crosstalk of H. pylori and obesity have been widely reported, the key factors and mechanism for their regulation in GC progression is still unclear. Inflammatory cytokines and the hormonal milieu of the stomach are believed to involve in the synergic regulation of H. pylori and obesity in induction of GC. More in-depth research on the precise underlying molecular mechanism by which H. pylori infection and obesity impact synergistically on GC development and progression warrant a further investigation.
Financial Support and Sponsorship
This work was supported by grants from 10th Singapore-China Joint Research Program (S2014GR0448), Natural
Science Foundation of Fujian Province (2019I0002), Natural Science Foundation of China (81673467), Leading Talents in Scientific and Technological Innovation, Double Hundred Talents Program of Fujian Province, and by Open Research Fund of State Key Laboratory of Cellular Stress Biology, Xiamen University.
Conflicts of Interests
There is no conflict of interests.
1. Alfarouk KO, Bashir AHH, Aljarbou AN, Ramadan AM, Muddathir AK, et al. (2019) The possible role of Helicobacter pylori in gastric cancer and its management. Front Oncol 9: 75.
2. Zhang RG, Duan GC, Fan QT, Chen SY (2016) Role of helicobacter pylori infection in pathogenesis of gastric carcinoma. World J Gastrointest Pathophysiol 7: 97-107.
3. de Martel C, Forman D, Plummer M (2013) Gastric cancer: Epidemiology and risk factors. Gastroenterol Clin North Am 42: 219-240.
4. Li Q, Zhang J, Zhou Y, Qiao L (2012) Obesity and gastric cancer. Front Biosci 17: 2383-2390.
5. Yang P, Zhou Y, Chen B, Wan HW, Jia GQ (2009) Overweight, obesity and gastric cancer risk: Results from a meta-analysis of cohort studies. Eur J Cancer 45: 2867-2873.
6. Upala S, Sanguankeo A (2016) Association between Helicobacter pylori infection and insulin resistance: A meta-analysis. Diabetes/metabolism Res Rev 32: 176-177.
7. Wu MS, Lee WJ, Wang HH, Huang SP, Lin JT (2005) A case-control study of association of Helicobacter pylori infection with morbid obesity in Taiwan. Arch Intern Med 165: 1552-1555.
8. Upala S, Jaruvongvanich V, Riangwiwat T, Jaruvongvanich S, Sanguankeo A (2016) Association between Helicobacter pylori infection and metabolic syndrome: A systematic review and meta-analysis. J Dig Dis 17: 433-440.
9. Nejati S, Karkhah A, Darvish H, Validi M, Ebrahimpour S, et al. (2018) Influence of Helicobacter pylori virulence factors CagA and VacA on pathogenesis of gastrointestinal disorders. Microb Pathog 117: 43-48.
10. Hatakeyama M (2006) Helicobacter pylori CagA -- a bacterial intruder conspiring gastric carcinogenesis. Int J Cancer 119: 1217-1223.
11. Ki MR, Hwang M, Kim AY, Lee EM, Lee EJ, et al. (2014) Role of vacuolating cytotoxin VacA and cytotoxin-associated antigen CagA of Helicobacter pylori in the progression of gastric cancer. Mol Cell Biochem 396: 23-32.
12. Dzierzanowska-Fangrat K, Michalkiewicz J, Cielecka-Kuszyk J, Nowak M, Celinska-Cedro D, et al. (2008) Enhanced gastric IL-18 mRNA expression in Helicobacter pylori-infected children is associated with macrophage infiltration, IL-8, and IL-1 beta mRNA expression. Eur J Gastroenterol Hepatol 20: 314-319.
13. Zeng B, Chen C, Yi Q, Zhang X, Wu X, et al. (2020) N-terminal region of Helicobacter pylori CagA induces IL-8 production in gastric epithelial cells via the beta1 integrin receptor. J Med Microbiol 69: 457-464.
14. Outlioua A, Badre W, Desterke C, Echarki Z, El Hammani N, et al. (2020) Gastric IL-1beta, IL-8, and IL-17A expression in Moroccan patients infected with Helicobacter pylori may be a predictive signature of severe pathological stages. Cytokine 126: 154893.
15. Hitzler I, Kohler E, Engler DB, Yazgan AS, Muller A (2012) The role of Th cell subsets in the control of Helicobacter infections and in T cell-driven gastric immunopathology. Front Immunol 3: 142.
16. Ren Z, Pang G, Clancy R, Li LC, Lee CS, et al. (2001) Shift of the gastric T-cell response in gastric carcinoma. J Gastroenterol Hepatol 16: 142-148.
17. Maruyama T, Kono K, Mizukami Y, Kawaguchi Y, Mimura K, et al. (2010) Distribution of Th17 cells and FoxP3(+) regulatory T cells in tumor-infiltrating lymphocytes, tumor-draining lymph nodes and peripheral blood lymphocytes in patients with gastric cancer. Cancer Sci 101: 1947-1954.
18. Bhuiyan TR, Islam MM, Uddin T, Chowdhury MI, Janzon A, et al. (2014) Th1 and Th17 responses to Helicobacter pylori in Bangladeshi infants, children and adults. PLoS One 9: e93943.
19. Liang JK, Hu L, Zheng XF (2012) [Study of Th1/Th2 balance in peripheral blood of chronic gastritis patients with Pi-Wei damp-heat syndrome]. Zhongguo Zhong xi yi jie he za zhi Zhongguo Zhongxiyi jiehe zazhi = Chinese journal of integrated traditional and Western medicine 32: 322-324, 328.
20. Kryczek I, Banerjee M, Cheng P, Vatan L, Szeliga W, et al. (2009) Phenotype, distribution, generation and functional and clinical relevance of Th17 cells in the human tumor environments. Blood 114: 1141-1149.
21. Pinchuk IV, Morris KT, Nofchissey RA, Earley RB, Wu JY, et al. (2013) Stromal cells induce Th17 during Helicobacter pylori infection and in the gastric tumor microenvironment. PLoS One 8: e53798.
22. Song M, Choi JY, Yang JJ, Sung H, Lee Y, et al. (2015) Obesity at adolescence and gastric cancer risk. CCC 26: 247-256.
23. Garten A, Schuster S, Kiess W (2012) The insulin-like growth factors in adipogenesis and obesity. Endocrinol Metab Clin North Am 41: 283-295.
24. Khandwala HM, McCutcheon IE, Flyvbjerg A, Friend KE (2000) The effects of insulin-like growth factors on tumorigenesis and neoplastic growth. Endocr Rev 21: 215-244.
25. Vasas P, Winslet MC, Shi YY (2009) The role of insulin-like growth factors (IGF) in cell division processes and in malignancy. Orvosi Hetilap 150: 2308-2312.
26. Lautenbach A, Budde A, Wrann CD, Teichmann B, Vieten G, et al. (2009) Obesity and the associated mediators leptin, estrogen and IGF-I enhance the cell proliferation and early tumorigenesis of breast cancer cells. Nutr Cancer 61: 484-491.
27. Garofalo C, Koda M, Cascio S, Sulkowska M, Kanczuga-Koda L, et al. (2006) Increased expression of leptin and the leptin receptor as a marker of breast cancer progression: Possible role of obesity-related stimuli. Clin Cancer Res 12: 1447-1453.
28. Tian C, Zhang L, Hu D, Ji J (2013) Ghrelin induces gastric cancer cell proliferation, migration and invasion through GHS-R/NF-kappaB signaling pathway. Mol Cell Biochem 382: 163-172.
29. Seto Y, Miura H, Wada I, Shimizu N, Nomura S (2010) [What is ghrelin for gastric cancer?]. Gan to kagaku ryoho Cancer Chemother 37: 1429-1435.
30. Au CC, Furness JB, Brown KA (2016) Ghrelin and breast cancer: Emerging roles in obesity, estrogen regulation. Cancer Front Oncol 6: 265.
31. La Vignera S, Condorelli R, Bellanca S, La Rosa B, Mousavi A, et al. (2011) Obesity is associated with a higher level of pro-inflammatory cytokines in follicular fluid of women undergoing medically assisted procreation (PMA) programs. Eur Rev Med Pharmacol Sci 15: 267-273.
32. Song M, Ahn JH, Kim H, Kim DW, Lee TK, et al. (2018) Chronic high-fat diet-induced obesity in gerbils increases pro-inflammatory cytokine and mTOR activation and elicits neuronal death in the striatum following brief transient ischemia. Neurochem Int 121: 75-85.
33. Hernandez JC, Cervantes MIM, Reyes JP, Marin NP, Cervantes EM, et al, (2017) Obesity is the main determinant of insulin resistance more than the circulating pro-inflammatory cytokines levels in rheumatoid arthritis patients. Rev Bras Reumatol 57: 320-329.
34. Mosaffa F, Kalalinia F, Lage H, Afshari JT, Behravan J (2012) Pro-inflammatory cytokines interleukin-1 beta, interleukin 6, and tumor necrosis factor-alpha alter the expression and function of ABCG2 in cervix and gastric cancer cells. Mol Cell Biochem 363: 385-393.
35. Smirnova MG, Kiselev SL, Gnuchev NV, Birchall JP, Pearson JP (2002) Role of the pro-inflammatory cytokines tumor necrosis factor-alpha, interleukin-1 beta, interleukin-6 and interleukin-8 in the pathogenesis of the otitis media with effusion. Eur Cytokine Netw 13: 161-172.
36. Xu X, Li W, Qin L, Yang W, Yu G, et al. (2019) Wei Q: Relationship between Helicobacter pylori infection and obesity in Chinese adults: A systematic review with meta-analysis. PLoS One 14: e0221076.
37. Chen LW, Kuo SF, Chen CH, Chien CH, Lin CL, et al. (2018) A community-based study on the association between Helicobacter pylori infection and obesity. Sci Rep 8: 10746.
38. Yang GH, Wu JS, Yang YC, Huang YH, Lu FH, et al. (2014) Obesity associated with increased risk of gastric Helicobacter pylori infection in an elderly Chinese population. JAGS 62: 190-192.
39. Lender N, Talley NJ, Enck P, Haag S, Zipfel S, et al. (2014) Review article: Associations between Helicobacter pylori and obesity--An ecological study. Aliment Pharmacol Ther 40: 24-31.
40. Ericksen RE, Rose S, Westphalen CB, Shibata W, Muthupalani S, et al. (2014) Obesity accelerates Helicobacter felis-induced gastric carcinogenesis by enhancing immature myeloid cell trafficking and TH17 response. Gut 63: 385-394.
41. Kiewiet RM, van Vliet AC (2006) Helicobacter pylori, obesity and gastro-oesophageal reflux disease: Is there a relation? Neth J Med 64: 131-132.
42. Isomoto H, Ueno H, Saenko VA, Mondal MS, Nishi Y, et al. (2005) Impact of Helicobacter pylori infection on gastric and plasma ghrelin dynamics in humans. Am J Gastroenterol 100: 1711-1720.
43. Ritze Y, Schollenberger A, Sinno MH, Buhler N, Bohle M, et al. (2016) Gastric ghrelin, GOAT, leptin and leptinR expression as well as peripheral serotonin are dysregulated in humans with obesity. J Neurogastroenterol Motil 28: 806-815.
44. Howard JM, Pidgeon GP, Reynolds JV (2010) Leptin and gastro-intestinal malignancies. Obes Reviews 11: 863-874.
45. Zhao L, Shen ZX, Luo HS, Shen L (2005) Possible involvement of leptin and leptin receptor in developing gastric adenocarcinoma. World J Gastroenterol 11: 7666-7670.
46. Tian PY, Fan XM (2012) The proliferative effects of ghrelin on human gastric cancer AGS cells. J Dig Dis 13: 453-458.
47. Liu J, Wang H, Chen G, Yang M, Wu ZX, et al. (2017) The synergy of Helicobacter pylori and lipid metabolic disorders in induction of Th17-related cytokines in human gastric cancer. J Cancer Metastasis Treat 3: 169-176.
48. Refaeli R, Chodick G, Haj S, Goren S, Shalev V, et al. (2018) Relationships of H. pylori infection and its related gastroduodenal morbidity with metabolic syndrome: A large cross-sectional study. Sci Rep 8: 4088.
49. Kassir R, Lointier P, Phelip JM, Denneval A, Williet N, et al. (2017) Barrett's oesophagus and oesophageal carcinogenesis following obesity surgery: Helicobacter Pylori must be eradicated? Obes Surg 27: 2464-2465.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Chemotherapy Research Journal (ISSN:2642-0236)
- Journal of Pathology and Toxicology Research
- Journal of Nursing and Occupational Health (ISSN: 2640-0845)
- Archive of Obstetrics Gynecology and Reproductive Medicine (ISSN:2640-2297)
- International Journal of Internal Medicine and Geriatrics (ISSN: 2689-7687)
- Journal of Ageing and Restorative Medicine (ISSN:2637-7403)
- Journal of Allergy Research (ISSN:2642-326X)