2795
Views & Citations1795
Likes & Shares
The oncocytic cells that we now consider to be the follicular-derived Hurthle cells were actually described by Askanazy in 1898. The term “Hurthle cells” has remained in the literature to describe follicular-derived epithelial cells with oncocytic cytology. A variety of thyroid non-neoplastic (autoimmune thyroiditis, multinodular goiter) and neoplastic conditions (Hurthle cell adenoma, Hurthle cell carcinoma) are associated with Hurthle cell cytology. We report a case of atypical hurthle cell adenoma in a 35 year old female who presented with a progressively increasing swelling in the right side of the neck for the past 1 year.
INTRODUCTION
The Hurthle cell has consistently been the center of debate in the discussion of non-neoplastic and neoplastic lesions of the thyroid ever since it was first described in 1894 by Hurthle [1]. In fact, the cells originally described by Hurthle are now considered to represent ultimobranchial body-derived parafollicular cells or C cells. The oncocytic cells that we now consider to be the follicular-derived Hurthle cells were actually described by Askanazy in 1898 [2]. Despite the fact that the cells originally described by Hurthle are likely not the Hurthle cells as considered today, the term has remained in the literature to describe follicular-derived epithelial cells with oncocytic cytology. Other terms for the Hurthle cells include oncocytic, eosinophilic and oxyphilic cells [3].
A variety of thyroid non-neoplastic (autoimmune thyroiditis, multinodular goiter) and neoplastic conditions (Hurthle cell adenoma, Hurthle cell carcinoma) are associated with Hurthle cell cytology. In addition, there are several thyroid neoplasms that should be considered when one observes a Hurthle cell neoplasm in the thyroid (oncocytic variant of medullary carcinoma, several variants of papillary thyroid carcinoma) [4].
CASE SUMMARY
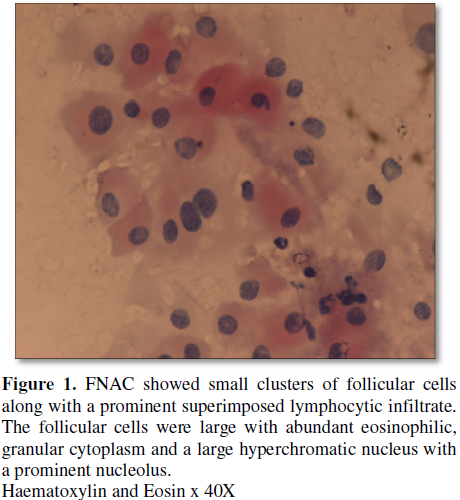
Fine needle aspiration cytology (FNAC) revealed a scant material comprising of thick colloid and small clusters of follicular cells along with a prominent superimposed lymphocytic infiltrate. The follicular cells cytologically were large with abundant eosinophilic, granular cytoplasm and a large hyper chromatic nucleus with a prominent nucleolus (Figure 1). Antibodies to thyroid peroxidase were found to be raised (472 IU/L). Flow cytometric analysis performed was inconclusive. In light of the above observations, a provisional diagnosis of Hashimoto’s thyroiditis with the possibility of a Hurthle cell neoplasm was made.
The patient underwent thyroidectomy. Grossly, the tissue specimen was a single encapsulated firm to hard nodular
DISCUSSION
The Hurthle cell is a follicular-derived cell, which has a cytoplasm characterized as “swollen”. This swelling is due in large part to the presence of numerous mitochondria in the cellular cytoplasm [5,6]. These cells commonly occur in nodular goiters and dominant adenomatous or hyperplastic nodules. In our opinion, the presence of these cells should not engender a diagnosis of Hurthle cell lesion.
By electron microscopy, the cytoplasm of Hurthle cells contains several thousand mitochondria of varying size and shape. The mitochondria often show dense core granules as well as filamentous inclusions [3,7].
Hurthle cells are present in a variety of non-neoplastic conditions involving the thyroid and are not specific for any disease process. Hurthle cell metaplasia is seen in a variety of benign conditions such as autoimmune thyroiditis and multinodular goitre and in thyroids of patients that have been treated with head and neck irradiation and systemic chemotherapy [8,9]. In addition, the thyroids of patients with long-standing hyperthyroidism (Graves’ disease) may show focal or diffuse Hurthle cell change [10]. In fact, Askanazy originally described these cells in thyrotoxicosis [2]. Hurthle cells also increase with aging [11]. In some instances, one can often find an entire nodule composed of oncocytes and the distinction of hyperplasia from neoplasia can be problematic.
Most of the conditions associated with Hurthle cell change are chronic, suggesting the possibility that alteration in the follicular epithelium to take on Hurthle cell histology requires chronic stimulation [9,10].
Grossly, Hurthle cell neoplasms are usually solitary and show at least partial encapsulation. They are distinctly mahogany brown due the abundant mitochondrial cytochrome content. Lesions may vary significantly in size from one to several centimeters. Similar to oncocytic neoplasms in other organs, a central scar may be evident. Hemorrhage and necrosis may be grossly seen especially in lesions that have undergone preoperative fine-needle aspiration biopsy; massive infarction either spontaneously or following fine-needle aspiration biopsy has been reported in Hurthle cell tumors [7,9]. The infarction can be extensive, leading to severe hampering of the histologic assessment. The presence of spontaneous infarction does not equate with malignancy.
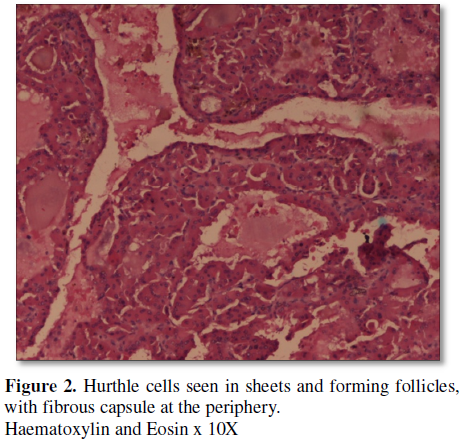
Microscopically, Hurthle cell neoplasms should be composed of at least 75% Hurthle cells. A variety of patterns of growth may be seen such as macrofollicular, follicular, trabecular, solid and pseudopapillary [11]. Many tumors will show a variety of these patterns of growth. The pattern of growth is usually follicular, but it also can be trabecular or solid. Hurthle cell neoplasms, both benign and malignant, have a tendency to show pseudopapillary change, which is felt to be an artifact from fixation and tissue processing.
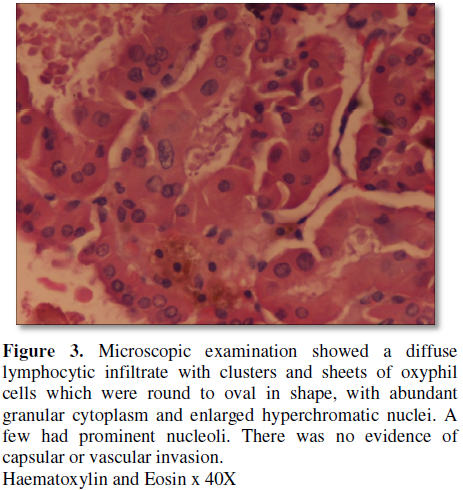
One of the most difficult aspects of examining Hurthle cell neoplasms is to determine whether the lesion is benign or malignant. Size in and of itself is not predictive of behavior, although larger lesions have a higher incidence of malignancy [11,12]. It is quite clear that pathologic criteria for malignancy, such as vascular and capsular invasion, can predict the clinical behavior of these tumors [13]. Nuclear atypia, multinucleation, cellular pleomorphism, mitoses or histologic pattern of the lesion are not determinants of malignancy [14]. The main criteria for malignancy include the presence of capsular and/or vascular invasion.
Hurthle cells of the thyroid in cytologic specimens appear as large polygonal cells with ample eosinophilic granular cytoplasm and central or occasionally eccentrically placed nuclei containing prominent nucleoli. The cell borders appear sharply demarcated and one may see empty space between 2 Hurthle cells similar to “windows” seen in mesothelial cells. Intranuclear grooves can be seen in Hurthle cells in Papanicolaou-stained smears and monolayer preparations [8,9]. Hurthle cells can be a prominent component in fine needle aspiration (FNA) specimens from Hurthle cell adenomas and carcinomas, Hurthle cell nodules in Hashimoto thyroiditis, adenomatoid nodules with Hurthle cell metaplasia, and nodules arising in a background of Graves’ disease [11]. Because Hurthle cells can be present in both nonneoplastic and neoplastic thyroid lesions it can be diagnostically challenging to differentiate between these two in thyroid FNA specimens [10,11].
Fine-needle aspiration cytology cannot differentiate between Hurthle cell adenoma and carcinoma because this distinction is dependent on demonstration of capsular and/or vascular invasion by histopathologic examination. Therefore, cytopathologists can only render a diagnosis of Hurthle cell lesion/neoplasm. These specimens demonstrate a monotonous population of Hurthle cells comprising more than 90% of the specimen. The cells are seen arranged in sheets, follicular groups, and singly scattered cells. Some authors have suggested that on the basis of nuclear atypia such as prominent nucleoli, high nuclearcytoplasmic ratio, and nuclear pleomorphism one can differentiate between Hurthle cell adenoma and carcinoma; however, others have refuted these observations. It has been shown that transgressing vessels and intracytoplasmic lumina are more commonly seen in neoplastic rather than non-neoplastic lesions [12,13]. Others have shown that transgressing vessels are a more reliable criterion for the diagnosis of Hurthle cell neoplasm as compared with intracytoplasmic lumina [13]. In our experience, monotonous population of Hurthle cells comprising more than 90% of the specimen is a reproducible and reliable criterion for the diagnosis of Hurthle cell neoplasm.
Flow cytometric analysis of hurthle cell tumors indicates that this technique cannot discriminate between benign and malignant hurthle cell neoplasms because adenomas may show aneuploidy and carcinomas may be diploid [14]. Though oncocytic follicular variants of papillary carcinomas showed RET/PTC rearrangements, none of the hurthle cells neoplasms showed any molecular genetic defects [15].
CONCLUSION
1. Kumar V, Abbas AK, Fausto N (2014) Robbins and Cotran- Pathologic Basis of Disease. 7th Edn. New Delhi: Elsevier, pp: 1169-1170.
2. Franssila KO (2010) Thyroid gland. In: Kisane JM, Anderson WAD, editors. Anderson’s Pathology. 8th Edn. St. Louis: CV Mosby Company, pp: 1399-1419.
3. Sah SP, Agarwal CS, Mishra A, Rani S (2010) Latent papillary carcinoma of thyroid with Hashimoto’s thyroiditis: Diagnosis by fine needle aspiration cytology. J Nephrol Med Assoc 39: 222-225.
4. Stojadinovic A, Hoos A, Ghossein RA, Urist MJ, Leung DHY, et al. (2012) Hurthle cell carcinoma: A 60 year experience. Ann Surg Oncol 9: 197-203.
5. Baloch ZW, Livolsi VA (2010) Pathology of thyroid and parathyroid disease. In: Mills SE, Carter D, Reuter VE, Greenson JK, Oberman HA, Stoler MH, editors. Sternberg’s Diagnostic Surgical Pathology. 4th Edn. Philadelphia: Lippincott Williams and Wilkins, pp: 578-579.
6. Rosai J (2012) Rosai and Ackermann’s Surgical Pathology. 9th Edn. New Delhi: Elsevier, pp: 544-547.
7. LiVolsi VA. Surgical pathology of the thyroid. In: Bennington JL, editor. Major problems in Pathology. Philadelphia: W.B. Saunders 22: 357-363.
8. Muller-Hocker J, Jacob U, Seibel P (1998) Hashimoto’s thyroiditis is associated with defects of cytochrome c oxidase in oxyphil Askanazy cells and with the common deletion (4,977) of mitochondrial DNA. Ultrastruct Pathol 22: 91-100.
9. Maximo V, Soares P, Lima J, Cameslle-Teijeiro J, Sobrinho-Simoes M (2002) Mitochondrial DNA somatic mutations (point mutations and large deletions) and mitochondrial DNA variants in human thyroid pathology - A study with emphasis on Hurthle cell tumours. Am J Pathol 160: 1857-1865.
10. Katoh R, Harach HR, Williams ED (1998) Solitary, multiple and familial oxyphil tumours of the thyroid gland. J Pathol 186: 292-299.
11. Angell JE, Lindner DJ, Shapiro PS, Hoffman ER, Kalvakolanu DU (2000) Identification of GRIM-19, a novel cell death-regulatory gene induced by the interferon P and retinoic acid combination, using a genetic approach. J Biol Chem 275: 33416-33426.
12. Maximo V, Botelho T, Capela J, Soares P, Lima J, et al. (2005) Somatic and germline mutations in GRIM-19, a dual function gene involved in mitochondrial metabolism and cell death is linked to mitochondrion rich (Hurthle cell) tumour of the thyroid. Br J Cancer 92: 1892-1898.
13. Gonzalez JL, Wang HH, Ducatman BS (1993) Fine needle aspiration of Hurthle cell lesions. A cytomorphologic approach to diagnosis. Am J Clin Pathol 100: 231-235.
14. Bronner MP, Clevenger C, Volsi VA, Edmonds PR, Lowell DM, et al. (1988) Flow cytometric analysis of DNA content in Hurthle cell adenomas and carcinomas of the thyroid. Am J Clin Pathol 89: 764-769.
15. Chiapetta G, Totti P, Cetta F, Giuliano A, Pentimalli F, et al. (2002) The RET/PTC oncogene is frequently activated in oncocytic thyroid tumors but not in oncocytic hyperplastic lesions. J Clin Endocrinol Metab 87: 364-369.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- International Journal of AIDS (ISSN: 2644-3023)
- Ophthalmology Clinics and Research (ISSN:2638-115X)
- International Journal of Clinical Case Studies and Reports (ISSN:2641-5771)
- Journal of Immunology Research and Therapy (ISSN:2472-727X)
- Journal of Cardiology and Diagnostics Research (ISSN:2639-4634)
- Journal of Forensic Research and Criminal Investigation (ISSN: 2640-0846)
- Journal of Cell Signaling & Damage-Associated Molecular Patterns