711
Views & Citations10
Likes & Shares
Objective: To evaluate the
potential pharmacological interactions in a group of adult hospitalized
patients who, due to an epileptic crisis, the present study was carried out.
Methodology: Descriptive-analytical
study, collecting data from hospitalized patients' medical records during the
investigation period.
Results: 937 patients were
enrolled in the study, 143 different drug interactions were detected in 35
patients, which involved 46 medicines administered. The interactions were
classified according to their theoretical relevance in serious (18.88%),
significant (51.04%) and minor (30.07%). 48.57% of patients presented drug
interactions with nutrients.
Conclusion: The intervention of a
multidisciplinary team is necessary for the management of these patients, which
includes nutritionists specially trained in aspects of pharmacokinetics and
pharmacodynamics, in order to minimize the incidence of pharmacological
interactions.
Keywords: Pharmacological interactions, Nutrients, Hospitalization
INTRODUCTION
The term drug
interactions (DI) refer to a clinical situation when the action of a drug is
altered by the presence of another drug or food, with neutral, beneficial or
malefic consequences [1]. The risk of its occurrence and severity is due to
factors related to the patient, the medicines themselves and the medical
prescription [2]; they can ends either in a therapeutic failure or in the
appearance of adverse effects. There are some illnesses that due to their
characteristics or type of medications used have a higher risk of interactions.
This is the case of patients diagnosed with epilepsy. Epilepsy is one of the
most common neurological diseases, with more than 50 million people affected around
the world and of which about 80% come from developing regions. It is estimated
that about 5 million people suffer from the disease in Latin America and the
Caribbean countries [3]. Epilepsy is characterized by recurrent seizures, which
are brief episodes of involuntary movements that can affect a part of the body
(partial seizures) or its entirety (generalized seizures) and are some cases
accompanied by loss of consciousness. Pharmacological treatment for epilepsy
manages to control epileptic seizures in up to 70% of patients, some of them
severe that need hospital admission [3].
The objective of this work is to identify the
possible drug interactions in hospitalized patients due to epileptic seizures;
in order to optimize their health care.
MATERIALS AND
METHODS
Type of study
Descriptive-analytical,
retrospective study.
Institution
The present
work was developed in a Public Hospital Specialized in Burns, located in the
city of Asunción, Republic of Paraguay.
Population
Adult patients
>18 years hospitalized for burn injuries for more than 48 h as a result of
epileptic seizures, who entered the boarding school unit in the period from
January 2015 to December 2017.
Data collection instrument
Review of
medical records of hospitalized patients. Pharmaceutical records of dispense
record.
Instrument for
identification and classification of interactions: Medscape Interaction Checker
(WebMD, LLC).
Variables
Date of
admission, date of discharge, age, sex, origin, number, type, dose of medicines
dispensed and type of administration, type of food and nutritional supplements
administered and type of intake.
STATISTICAL ANALYSIS
The statistical
analysis was performed with the EPI INFO statistical software, each variable
was coded for the programme management, with its description and its
categories. This programme allows expressing the results in frequency and
percentage of each study variable.
ETHICAL ASPECTS
This work was
carried out according to international standards for biomedical research in
human beings proposed by the Council of International Organizations of Medical
Sciences (CIOMS) where the confidentiality of data obtained from patient
records is respected, for this; the project was presented to the Research
Ethics Committee of the Faculty of Chemical Sciences, National University of
Asuncion (UNA) Paraguay Republic and each patient was asked to sign an informed
consent for this purpose.
RESULTS
A total of 937
adult patients entered the Burns boarding room unit during the study period, 35
patients of whom suffered burns due to epileptic seizures, which represent
3.73% of all admissions. 62.86% of the patients belong to the female sex. The
age range is between 18 to 74 years, with an average of 40 years and a standard
deviation of 16.5. 65.71%
of patients come from rural areas (Table
1).
The administration of registered and
dispensed medicines allowed us to identify that the patients enrolled in the
study received between 5 and 28 medicines on the whole day of hospitalization
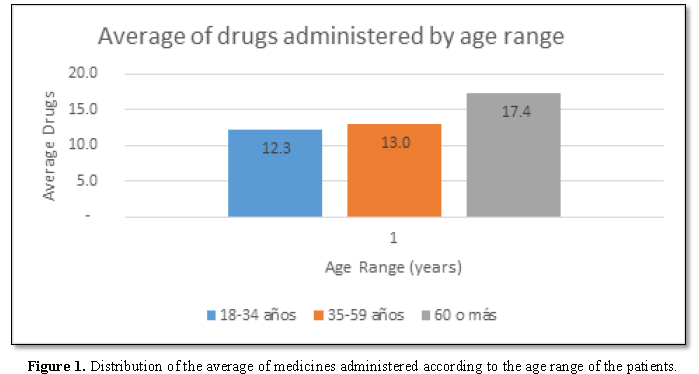
(29 ± 8), with an average of 12 medicines per patient (Figure 1).
It was found that patients with ages over 60 years have a tendency to
receive a greater quantity of medicines (Figure
2), which is directly related to the risk of possible pharmacological
interactions in patients with that age range.
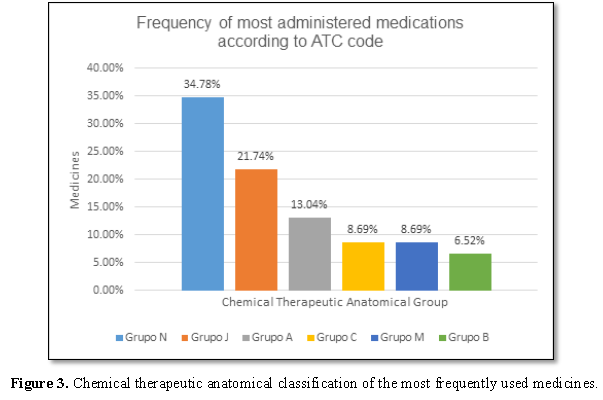
Of a total of 46 medicines that were detected in the interactions, the
most prescribed correspond to group N (Drugs that act on the Nervous System)
according to ATC. Phenytoin was the most prescribed and administered medicines,
with a frequency of 71.43% of the total patients. Then they are followed by
those of group J, anti-infective drugs for systemic use, in which ciprofloxacin
corresponds to the most administered medicines within the group, with a
frequency of 45.71% (Table 2 and Figure 3).
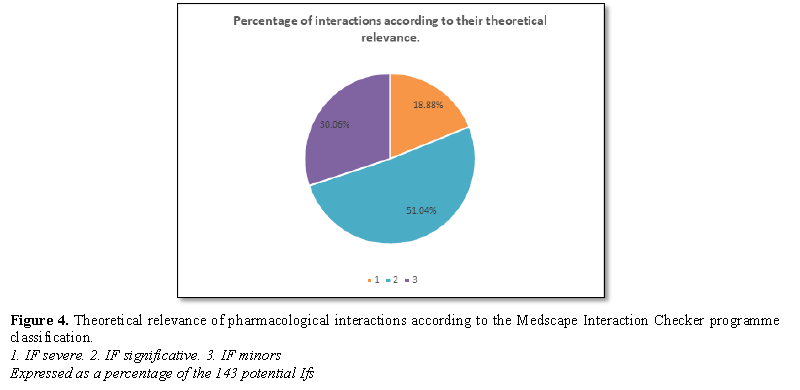
The patients included in this study presented a total of 143
pharmacological interactions, which were classified according to their
theoretical relevance in serious; which represent 18.88% of the interactions;
51.04% significant and 30.07% lower (Figure
4). The lowest number of potential interactions presented by the patients
was 4 and the highest was 44 interactions (Tables
3-5).
48.57% of the patients in the study had drug-nutrient type interactions (Table 6), corresponding to 3.50% of
the total drug interactions. The most frequent potential interaction was that
of Phenytoin with Calcium (8.39%).
DISCUSSION
The prevalence of interactions observed in
the present study was directly proportional to the increase in the age of the
patients, suggesting that older patients are more vulnerable due of their
greater number of prescribed drugs, the complexity in treatment and a reduction
in renal function [2,4].
Pharmacological interactions themselves are a
cause of hospital admission, but in practice this is relatively unusual. In
daily practice only some of them have relevant clinical consequences that
warrant hospitalization. Although it is very difficult to know the real
frequency and clinical relevance of the interactions, it is currently known
that many of them do not pose a risk to the patient and those that are
clinically significant only occur in a small proportion of patients.
In this work 143 interactions in 937 patients
were detected, they are significantly smaller than the data provided from other
published studies. Several authors found 329 theoretical interactions in 412
patients, although it should be noted that these data cannot be directly
compared with our study, due to the methodological difference, population and
design, which contributes considerably for the variation of the observed
frequencies [4].
The interactions found in our work were
classified into 3 levels: serious, significant and minor; the “significant”
ones were the most prevalent (51.04%), a fact that matches with other
observations made by various authors [2].
Co-morbidities and the amount of drugs
co-administered can increase the incidence of drug-related adverse effects and
therefore, increase the risk of injury. For example, drowsiness is a common
side effect of antiepileptic drugs and could be enhanced due to their
association with hypnotic drugs or with psychiatric problems [5].
Regarding the number of medicines
administered and their indication, it was observed that there was a higher
frequency of medicines with action on the nervous system, being phenytoin
(anti-epileptic) the most administered drug (54.34%) and therefore the medicine
most involved in possible interactions. In addition, carbamazepine and
clonazepam, also used to control epileptic seizures, were found within the
group.
Some of the anti-epileptics bind extensively
to plasma proteins. Only the unbound (free) fraction of the drug is able to
pass through membranes and have pharmacological activity, hence many drug-drug
interactions may occur as a result of competition for protein binding sites as
observed in the case of phenytoin-ibuprofen or phenytoin-diazepam in our
results.
From these data it is inferred that it might
be appropriate to monitor the levels of the free fraction of drugs only in
those cases where drug interactions are important and have sufficient risk to
the health of patients [6].
Although monitoring would not be useful in all
cases, it should be performed for drugs with the greatest potential to produce
interactions are those with a narrow therapeutic index, which require precise
control of plasma concentrations, those with dose-response curves with a broad
slope, saturable metabolism or in case there is an indication of chronic
treatment [7].
In the second place in the ranking of the
interactions, we identified drugs from therapeutic group called “anti-infective
for systemic use” (Group J), in 34.78% interaction of the cases, being
ciprofloxacin the most prevalent in the group.
Microbial use was associated with age,
co-morbidities and toxic habits such as smoking and alcoholism of patients [8].
It was detected that 48.57% of the patients
under study presented an interaction of the drug-nutrient type. The most
frequent case of this type was that phenytoin with calcium (8.39%), an
essential component of nutritional supplements. This fact is clinically
relevant, since anticonvulsants have a higher risk of producing bone demineralization
and fracture on pathological bone, events that are facilitated by risk factors
such as vitamin D deficiency and hypocalcaemia [9].
To assess the safety of concomitant use of
drugs, the different characteristics of the interaction must be studied.
In this context, the presence of the clinical
pharmacist in each of the hospital units is essential to be able to monitor the
pharmacotherapeutic treatment and taking into account other variables inherent
to the patient, which go unnoticed by other disciplines and professions; in
order to minimize the incidence of preventable drug interactions [2].
CONCLUSION
The present
study 48.57% of hospitalized patients with epilepsy, a drug-nutrient or drug
interactions were detected. The drugs involved belong predominantly to the
group acting on the nervous system, being phenytoin the medicine that presented
the greatest interaction risk. The presence of a clinical pharmacist could help
to prevent and reduce the incidence of drug interactions, optimizing health
care practice.
1.
Santibañez C, Roque J, Morales G, Corrales R. (2014)
Characteristics of drug interactions in a pediatric intensive care unit.
Chilean Journal of Pediatrics 85.
2.
Caribe RA, Chaves GR, Pocognoni JD, Souza IA (2013)
Potential drug interactions in patients with sepsis admitted to the intensive
care unit. Hosp Pharm 37.
3.
Orozco J, Quintero J, Marin Medina D, Castaño J,
Hernández P, et al. (2016) Clinical and sociodemographic profile of adult
epilepsy from a reference center in Colombia. Science Direct 31.
4.
López Vazquez P, Rodríguez Moreno C, Durán Parrondo C,
Tato Herrero F, Rodríguez López I, et al. (2005) Interactions between
medications prescribed for discharge in an internal medicine service. Ann
Intern Med 22.
5.
Asadi Pooya A, Nikseresht A, Yaghoubid E, Nei M (2012)
Physical injuries in patients with epilepsy and their associated risk factors.
Elsevier 21: 165-168.
6.
López González R (2016) Epilepsy, pharmacological
treatment and its monitoring. Dome 30.
7.
Miquet RLM, Rodriguez GR, Llorente BN, Hernández CM,
González RH (2015) Local infection of the burn and nutritional status. Cuban
Magazine of Nutrition and Nutrition 25: 301-313.
8.
Vildoso Fernández M (2009) Nutritional effects of
anticonvulsants. MedWave 9.
9.
Piñeiro Corrales G (2009) Drug-nutrient interactions
in neurological pathologies. Hosp Nutr Suppl 2.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Alcoholism Clinical Research
- Journal of Clinical Trials and Research (ISSN:2637-7373)
- Journal of Immunology Research and Therapy (ISSN:2472-727X)
- Oncology Clinics and Research (ISSN: 2643-055X)
- Journal of Cell Signaling & Damage-Associated Molecular Patterns
- Journal of Forensic Research and Criminal Investigation (ISSN: 2640-0846)
- International Journal of Clinical Case Studies and Reports (ISSN:2641-5771)