2570
Views & Citations1570
Likes & Shares
Objective: The objective of this study is to evaluate
the alterations of the optical coherence tomography angiography (OCTA)
parameters of peripapilary vascular density and perilously vascular perfusion
in patients with glaucomatous optic neuropathy compared to normal.
Methods: Study was conducted with 300 subjects aged 45-65 who were diagnosed
with primary open angle glaucoma. In every subject, Retinal nerve fiber layer
thickness, peripapillary vascular perfusion and peripapillary vessel density
were measured. All were divided in to 4 sub-groups according to the severity of
glaucomatous optic neuropathy based on Optical Coherence Tomography based RNFL thickness
grading section in Hodapp-Parrish-Anderson criteria for glaucoma.
Results: Peripapillary micro vascular density and peripapillary blood
perfusion values were significantly decreased (p>0.05) in every glaucomatous
subgroup compared to normal. Peripapillary micro vascular density and
peripapillary blood perfusion values were directly proportional to the RNFL
thickness which indicates a possible reduction of microvasculature of
peripapillary region in glaucomatous eyes.
Conclusion: Both peripapilary vessel density and perfusion is proportional to
the thickness of Retinal nerve fiber layer and those values correlate with
glaucomatous damage in the peripapillary region.
The use of OCT angiography in glaucomatous
eyes for evaluation of peripapillary perfusion and vessel density gives access
to visualize the defects and quantified the evaluations, with high
repeatability and reproducibility.
Keywords:
Glaucomatous optic neuropathy, Retinal nerve fiber
layer thickness, Peripapillary vessel density, Peripapillary microvascular
perfusion
INTRODUCTION
Glaucoma is rated as the second
leading cause of blindness in the world and the commonest cause of irreversible
blindness affecting almost 60 million people globally [1]. It is characterized
by optic neuropathy associated with retinal ganglion cell death, which then
leads to retinal nerve fiber layer (RNFL) thinning and optic nerve head (ONH)
cupping that leads to a progressive loss of visual field [2,3].
A study conducted in year 2000 at Johns Hopkins
University School of Medicine, Baltimore, USA to estimate the prevalence of
glaucoma among people worldwide, states that the number of people with primary
glaucoma in the world by the year 2000 is estimated nearly 66.8 million, with
6.7 million suffering from bilateral blindness. In developed countries, fewer
than 50% of those with glaucoma are aware of their disease [4].
The Glaucoma Service and the Dana
Center for Preventive Ophthalmology Baltimore, USA states on a review of
published data with use of prevalence models, which was done to estimate the
number of people with open angle and angle closure glaucoma in 2010 and 2020
that 60.5 million people with Open Angle Glaucoma (OAG) and Angle Close
Glaucoma (ACG) in 2010, increasing to 79.6 million by 2020, and of these, 74%
will have OAG. Women will comprise 55% of OAG, 70% of ACG and 59% of all glaucoma
in 2010. Asians will represent 47% of those with glaucoma and 87% of those with
ACG. Bilateral blindness will be present in 4.5 million people with OAG and 3.9
million people with ACG in 2010, rising to 5.9 and 5.3 million people in 2020,
respectively [5].
The epidemiology of glaucoma by Giangiacomo
and Coleman [6] mentioned in 2002, 37 million individuals were blind worldwide,
with glaucoma accounting for 12.3% of these individuals. By the year 2020 it is
estimated that there will be almost 80 million people in the world with
open-angle glaucoma and angle-closure glaucoma. The majority of these
individuals will have open-angle glaucoma. Of those with ACG, it is predicted
that 70% will be women and 87% will be Asian. Bilateral blindness from glaucoma
is projected to affect 8.4 million individuals worldwide by 2010 and greater
than 11 million by 2020 [6].
A review of population-based glaucoma
prevalence studies in Asians, Cho and Kee [7] states that the glaucoma-related
population-based studies from Japan, Mongolia, India, Singapore, Thailand,
China, Bangladesh, Myanmar, Sri Lanka and South Korea show a higher glaucoma
prevalence in Asian patients, including a higher incidence of primary
angle-closure glaucoma, than in white patients, although primary open-angle
glaucoma (POAG) is still the most commonly reported [7].
Globally, glaucoma is a significant cause of vision loss
that disproportionately affects women and Asians. Risk factors for open-angle
glaucoma include increased age, African and Asian ethnicity, family history,
increased intraocular pressure, myopia and decreased corneal thickness [3,6,7].
Risk factors for angle closure glaucoma include Inuit and
Asian ethnicity, hyperopia, female gender, shallow anterior chamber, short
axial length, small corneal diameter, steep corneal curvature, shallow limbal
chamber depth and thick, relatively anteriorly positioned lens [6,7].
Elevated intraocular pressure (IOP) is a key risk factor
for the development and progression of glaucoma [8,9]. A number of conditions
such as congenital, angle-closure and secondary glaucoma clearly show that
increased IOP is sufficient to lead to glaucomatous optic neuropathy [10].
Although elevated intraocular pressure (IOP) is the main
risk factor for glaucoma, large numbers of patients also develop glaucoma with
normal IOP levels [10]. Therefore, other risk factors and in particular
vascular risk factors have been implicated in the pathogenesis of glaucoma
[3,10].
A study conducted in Pennsylvania, USA in 1999 to find out
the effect of systemic hypertension in optic nerve blood flow in glaucoma,
found that the optic nerve blood flow is reduced in glaucoma patients. Glaucoma
patients without systemic hypertension have lower optic nerve blood flow than
those with hypertension [11].
Another study conducted to observe the Optic Nerve Head in
Glaucomatous Optic Neuropathy at Washington University School of Medicine, St
Louis, Mo in 1997, presents the evidence of specific changes in the
extracellular matrix of the lamina cribrosa and the role of astrocytes in
glaucomatous optic neuropathy [12]. Hernandez [13] and Roth and Foos [14] have
explained the Surface Structure of the Optic Nerve Head in Glaucoma in 1972 and
1973.
It was found that the connective tissue dynamics should
directly and indirectly influence astrocyte and glial metabolism as well as
axonal transport, glaucomatous damage within the ONH may not necessarily occur
at locations with the highest levels of IOP-related connective tissue strain;
the study was conducted to observe the mechanical environment of the optic
nerve head in glaucoma in Portland, Oregon, USA in 2008 [15].
Another study conducted to evaluate the regional
structure-function relationship between visual field sensitivity and retinal
nerve fiber layer thickness and optic nerve head measurements using
spectral-domain optical coherence tomography in 2014 at University Hospital,
CHU Grenoble, Grenoble, France with 142 subjects, states that the
structure-function relationship was significantly stronger with Bruch membrane
opening minimum rim width than other Optic nerve head SD-OCT parameters [16].
Optical coherence tomography (OCT) angiography is a novel
technology that rapidly and conveniently images blood vessels in vivo without
the use of exogenous dyes, providing high-resolution and three-dimensional information
on the morphology of physiological and pathological blood vessels at different
layers of the retina by separating static (tissue) from motion (blood flow)
signals [17,18].
A study conducted at Doheny Eye Institute, Los Angeles,
California, USA to measure the Repeatability of automated vessel density
measurements using optical coherence tomography angiography found that
automated vessel density measurements using OCT-A showed excellent
repeatability in healthy individuals. Although repeatability will also need to
be established in the setting of disease, the level of reproducibility should
be useful for assessing the significance of differences in capillary density
over time or under different conditions [19].
Study conducted in Bangalore, India to evaluate the
diagnostic ability of peripapillary vessel density measurements of optical
coherence tomography angiography (OCTA) in primary open-angle (POAG) and
primary angle-closure glaucoma (PACG) states that the Diagnostic ability of
peripapillary vessel density parameters of OCTA, especially the
inferio-temporal sector measurement, was good in POAG and PACG. Diagnostic
abilities of vessel density measurements were comparable to RNFL measurements
in both POAG and PACG [17].
A pilot study
conducted at Portland, USA in 2012 to obtain Quantitative measurements of OCT
angiography of optic nerve head blood flow, indicates that the OCT angiography
can detect the abnormalities of optic nerve head perfusion and has the
potential to reveal the Optic nerve head blood flow mechanism related to
glaucoma [20].
Also, a study
done to observe the optical coherence tomography angiography of optic disc
perfusion in Glaucoma in Oregon Health & Science University, Portland
suggest that the optical coherence tomography angiography, repeatedly measures
optic disc perfusion and may be useful in the evaluation of glaucoma and
glaucoma progression [21].
Optical coherence
tomography angiography vessel density had similar diagnostic accuracy to RNFL
thickness measurements for differentiating between healthy and glaucoma eyes. A
study done in California United States in 2016, with 164 eyes suggests that
OCT-A measurements reflect damage to tissues relevant to the pathophysiology of
OAG [22].
Vessel Density is
defined as the total length of perfused vasculature per unit area in a region
of measurement. It is measured in units of inverse millimeters. Vessel density
can be thought of as Untangling all the vasculature in a region of tissue. Vascular density was defined as
the percentage of the sample area occupied by vessel lumens following binary
reconstruction of images. Vascular density equals to the Proration of the
flowing vessel divided by total scan area [23].
Perfusion density
is defined as the total area of perfused vasculature per unit area in a region
of measurement. This metric is calculated by summing up the number of pixels
which contain perfused vasculature and dividing the sum by the total number of
pixels in the considered region [23].
Optical coherence
tomography has become an essential tool for evaluation of Retinal nerve fiber
layer. The Cirrus HD-OCT (Carl Zeiss Meditec, Inc. Dublin, CA) is a
spectral-domain OCT that provides visualization of the distribution pattern and
measurement of RNFL abnormalities in a 200 × 200 µ area. Analysis of the RNFL
thickness deviation maps—a color-coded map displaying areas of RNFL
abnormalities—detects glaucoma with high sensitivity and specificity.
OBJECTIVE
The objective of
this study is to evaluate the alterations of the optical coherence tomography
angiography (OCTA) parameters of peripapillary vascular density and peripapillary
vascular perfusion in patients with glaucomatous optic neuropathy compared to
normal.
There are many
studies that have been investigated in microvasculature of the peripapillary
area in glaucomatous optic neuropathy on different types of ethnic groups
[15,16,20,21].
Each study was
found relations as well as differences in the peripapillary microvasculature in
glaucomatous eyes.
There is no any
research or studies have been found in the literature regarding a Sri Lankan
sample and very few with south Asian samples regarding this topic.
METHODS
Participants
A retrospective
cross sectional study according to convenience sampling was carried out at
Vision Care Optical Services Pvt. Ltd., No 6 ward place Colombo 7, form 2017
March to 2019 April. Participants were 150 Sri Lankan adults (300 eyes), 150
males and 150 females’ age range from 45 to 65 years who were diagnosed with
Primary Open Angle Glaucoma (POAG). Subjects who presented with any type of
secondary glaucoma, angle closed glaucoma, any other ocular pathology and those
who presented with systemic vascular disorders were excluded from the study.
All participants were given a full explanation of investigation procedures with
the option to withdraw from the study at any time.
Materials
Data were recorded in each subject by
carrying out a questionnaire. Questionnaire was divided into three parts; first
part is for social demographic data which include age, gender, marital status
and area of residence. Second part is for personal health and medical status
which include level of blood sugar, blood pressure levels, body mass index,
cholesterol levels, family history of ocular or other diseases, past
ocular/systemic medical and surgical history, current ocular/systemic
medications, history of habits- alcohol consumption/smoking, etc. Third part is
for optical coherence tomography assessment which includes retinal nerve fiber
layer thickness, peripapillary vessel density and peripapillary perfusion.
Retinal nerve fiber layer thickness,
peripapillary vessel density and peripapillary perfusion was measured by the
Cirrus 5000 spectral domain Optical Coherence Tomography by Carl Zeiss Meditec
with acquired 200 × 200 Optic disc cube scans for RNFL thickness measurements
and 6 × 6 angioplex scanning strategy on the Optic nerve head for the
measurements of peripapillary vessel density and peripapillary perfusion and
all the acquired images were analyzed by Cirrus 5000 software version 5000.10.1
and 5000.10.1 angioplex matrix. Reports with signal strength less than 6 were
excluded.
Procedure
Grading of the subjects was done according
to the basis of RNFL thickness of each subject. The Optical Coherence Tomography
based RNFL thickness grading section in Hodapp-Parrish-Anderson criteria for
Glaucoma was used for grading of Glaucoma stages.
All subjects were divided in to 3 stages of
glaucoma and in to an age matched normal group. Stage 3 includes the subjects
with average RNFL thickness of 37 ± 8.77, Stage 2 includes the average RNFL
thickness of 47 ± 8.77 µ, Stage 1 includes the average RNFL thickness of 67 ± 8.77
µ while the controlled group indicate average RNFL thickness more than 83.77 µ.
The changes in peripapillary vessel density and
peripapillary perfusion in the disease groups was analyzed using one-way ANOVA.
All statistical analyses were performed
using the Statistical Package for the Social Sciences by IBM Company, Chicago,
IL.
RESULTS
In the study population peripapillary
perfusion and vessel density was studied in 50 normal eyes and 200 glaucomatous
eyes. Glaucomatous eyes were divided in to 3 stages based on and each group
contends 50 subjects which were randomly selected and the rest was excluded.
The mean age of the study group was 56.6 ± 7.53 years of age. Each group
contained 50 subjects. The Average RNFL thickness in controlled group was
94.6+2.6 µ, in stage 1 glaucoma group 76.3+3.1 µ, Stage 2 glaucoma groups
46.8+2.7 µ and in stage 3 glaucoma group 38.5+1.8 µ. One way ANOVA Statistical
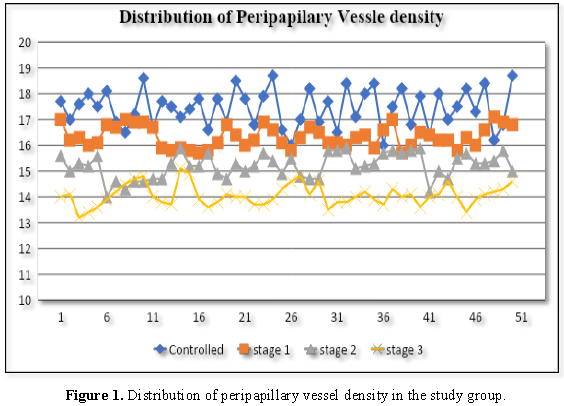
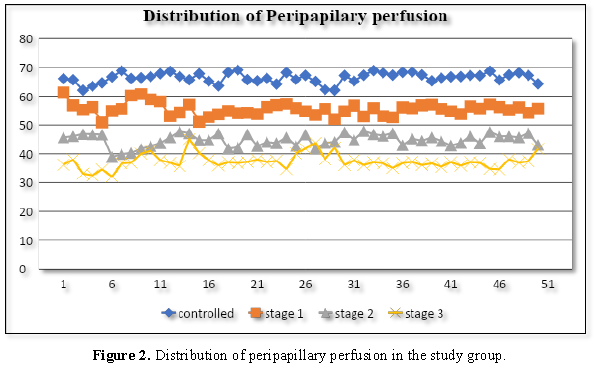
test was performed in analysis of data. In controlled group the average PVD was
17.44 ± 0.53 and average PMP was 66.4 ± 3.12. In glaucoma stage 1 group the
average PVD was 16.33 ± 0.16 and average PMP was 55.43 ± 4.71. In glaucoma
stage 2 group the average PVD was 15.20 ± 0.23 and average PMP was 44.77 ± 4.82
and in glaucoma stage 3 group the average PVD was 14.06 ± 0.17 and average PMP
was 37.33 ± 6.61.
Test of homogeneity of variances in vessel
density levene statistics based on mean was 11.049, based on median was 10.320,
based on median and with adjusted df was 10.320 and based on trimmed mean was
10.966, P=0.0001. One way ANOVA test shows 382.563 F Value and P=0.001. Test of
homogeneity of variances in peripapillary perfusion levene statistics based on
mean was 0.896, based on median was 0.826, based on median and with adjusted df
was 0.826 and based on trimmed mean was 0.854, P=0.044. One way ANOVA test
shows 1669.107 F value and P=0.001.
In vessel density comparisons TUKEY HSD
analysis shows a mean deference between groups 1 and 2=1.10 (P=0.001) between 1
and 3=2.23 (P=0.001) between 1 and controlled=3.37 (P=0.001); mean deference
between groups 2 and 3=1.13 (P=0.001) 2 and controlled=2.27 (P=0.001) and mean
difference between groups 3 and controlled=1.14 (P=0.001). In every group mean
deference is significant at the 0.05 level.
In peripapillary perfusion multiple
comparisons TUKEY HSD analysis shows a mean deference between groups 1 and
2=10.97 (P=0.001), between 1 and 3=21.63 (P=0.001), between 1 and
controlled=29.07 (P=0.001); mean deference between groups 2 and 3=10.66
(P=0.001), 2 and control=18.10 (P=0.001) and mean deference between group 3 and
control=7.43 (P=0.001). In every group mean deference is significant at the
0.05 level.
According to the results of our study it
was noted in every group, peripapillary microvascular perfusion and
peripapillary vessel density significantly decreased when the severity of
glaucomatous optic neuropathy increased (Figures
1 and 2).
DISCUSSION
We
found that the peripapillary vessel density and perfusion was lower in
glaucomatous eyes when compared with aged-matched normal eyes and the vessel
density progressively decreased with the severity of glaucoma stage. The
correlation between the reduction in RNFL thickness and vessel density and
perfusion was proportional and this was significant in every group.
A study to compare optic disc perfusion between normal
subjects and subjects with glaucoma using optical coherence tomography
angiography and to detect optic disc perfusion changes in glaucoma with 24
normal subjects and 11 patients with glaucoma was conducted in Oregon Health
& Science University, Portland, Oregon. It shows that the disc flow index
was reduced by 25% in the glaucoma group; In our study we have found that the vessel density
was reduced in 13.2% in stage 1 glaucoma group, 14% in stage 2 glaucoma group
and 15.7% in stage 3
glaucoma group, while the peripapillary perfusion reduced in 32.1% in stage 1
glaucoma group, 38.9 % in stage 2 glaucoma group and 50.9% in stage 3 glaucoma
group.
A study on
diagnostic agreement with Heidelberg Retinal Tomography Retinal nerve fiber
layer imaging with spectral-domain optical coherence tomography done at Hong Kong, China in 2010 with 79
glaucoma and 76 normal subjects states that the optics disc blood flow was
significantly decreased in glaucomatous patients compared to age matched normal
group [21]. Even
though we have used the Cirrus 5000 Optical coherence tomography for
measurements of ONH microvasculature every glaucoma group showed a reduction of
the density and the perfusion values in peripapillary microvasculature.
Microvascular
density in glaucomatous eyes with hemifield visual field defects was evaluated
in a study conducted in Kyoto, Japan in 2016 with 60 eyes with primary open
angle glaucoma, 41 with superior and 19 with inferior hemifield visual field
defect. They have found that the microvascular reduction was associated with VF
defects in a region-specific manner. Even though we have not measured any
functional aspects in the study group, we suggest that the functional
evaluation with structural changes happens in peripaillary area deserves
further study [24].
The
sample that we used was selected on Sri Lankan population and subjects of any
other ethnicity were not included in the study. Studies that carried out to
evaluate the Peripapillary microvasculature based on the population samples in
United States of America, Africa, China, Japan and other parts of the world
indicates that there is a co-relation between the reductions of peripapillary
microvascular parameters in development of glaucoma [25-29].
However,
a prospective study conducted for a 2 year time period in Budapest, Hungar with
individuals who use anti-glaucoma eye drops states that the peripapillary
vessel density measurement did not support the detection of glaucomatous
progression. But in our study the behavior of vessel density measurements
strongly agreed for understanding the changes in the peripapillary area in the
development of glaucoma [30].
CONCLUSION
According
to our study peripaillary vessels density and peripapillary vessel perfusion
shows decreased values in glaucomatous optic neuropathy in the observation of
patients with primary open angle glaucoma.
Both
peripapillary vessel density and perfusion is proportional to the thickness of
retinal nerve fiber layer and those values correlates with glaucomatous damage
in the peripapillary region.
Using
OCT angiography in glaucomatous eyes for evaluation of peripapillary perfusion
and vessel density will give access to visualize the defects and quantified the
evaluations, with high repeatability and reproducibility. Quantitative OCT
angiography measurements may have a value in future studies to determine its
potential usefulness in glaucoma evaluation.
LIMITATIONS
There
could be a potential effect of anti0glaucoma drugs on microvasculature of the
peripapillary area [31,32]. 143 eyes that were included in the study which was
categorized into any of glaucoma stages had been receiving different anti-glaucoma
eye drops for different time period. Some studies states that there is no
decrease in blood circulation found with anti-glaucoma eye drops [33].
Nevertheless, the effect of anti-glaucoma eye drops on perfusion and vessel density
require further investigations.
However,
the focal nature of the eyes with decreased vessel density and perfusion
correlates with glaucomatous damage which indicates with an RNFL defects.
1.
Khaw
PT, Elkington AR (2004) Glaucoma. ABC Eyes 328: 97-99.
2.
Newell
FW (1989) Intraocular pressure and glaucoma. Am J Ophthalmol 107: 186-188.
3.
Grieshaber
MC, Flammer J (2005) Blood flow in glaucoma. Curr Opin Ophthalmol 16: 79-83.
4.
Quigley
HA (1996) Number of people with glaucoma worldwide. Br J Ophthalmol 80:
385-386.
5.
Quigley
HA, Broman AT (2006) The number of people with glaucoma worldwide in 2010 and
2020. Br J Ophthalmol 90: 262-267.
6.
Giangiacomo
A, Coleman AL (2009) The epidemiology of glaucoma. Essentials in Ophthalmology,
pp: 13-21.
7.
Cho HK, Kee C (2014) Population-based glaucoma
prevalence studies in Asians. Surv Ophthalmol 59: 434-447.
8.
Gardiner
SK, Fortune B, Wang L, Downs JC, Burgoyne CF (2012) Intraocular pressure
magnitude and variability as predictors of rates of structural change in
non-human primate experimental glaucoma. Exp Eye Res 10: 1-8.
9.
Awadalla
MS, Fingert JH, Roos BE, Chen S, Holmes R, et al. (2015) Copy number variations
of TBK1 in Australian patients with primary open-angle glaucoma. Am J
Ophthalmol 159: 124-130.
10.
Flammer
J, Orgul S, Costa VP, Orzalesi N, Krieglstein GK, et al. (2002) The impact of
ocular blood flow in glaucoma. Prog Retin Eye Res 21: 359-393.
11.
Grunwald
JE, Maguire MG (1999) Optic nerve blood flow in glaucoma: Effect of systemic
hypertension. Am J Ophthalmol 127: 516-522.
12.
Hernandez
MR, Pena JDO (1997) The optic nerve head in glaucomatous optic neuropathy. Arch
Ophthalmol 115: 389-395.
13.
Hernandez
MR (2000) The optic nerve head in glaucoma: Role of astrocytes in tissue
remodeling. Prog Retin Eye Res 19: 297-321.
14.
Foos
RY, Roth AM (1973) Surface structure of the optic nerve. Am J Ophthalmol 76:
662-671.
15.
Crawford
DJ, Michael DR, Claude FB (2008) Mechanical environment of the optic nerve head
in glaucoma. Optom Vis Sci 85: 425-435.
16.
Villard
FC, Chiquet C, Romanet JP, Noel C, Aptel F (2014) Structure-function
relationships with spectral-domain optical coherence tomography retinal nerve
fiber layer and optic nerve head measurements. Invest Ophthalmol Vis Sci 55:
2953-2962.
17.
Spaide
RF, Klancnik JM, Cooney MJ (2015) Retinal vascular layer imaged by fluorescein
angiography and optical coherence tomography angiography JAMA Ophthalmol 133:
45-50.
18.
Flocks
M, Miller J, Chao P (1959) Retinal circulation time with the aid of fundus
cinephotography. Am J Ophthalmol 48: 3-6.
19.
Al-Sheikh
M, Tepelus TC, Nazikyan T, Sadda SR (2017) Repeatability of automated vessel
density measurements using optical coherence tomography angiography. Br J
Ophthalmol 101: 449-452.
20.
Jia Y,
Morrison JC, Tokayer J, Tan O, Lombardi L, et al. (2012) Quantitative OCT
angiography of optic nerve head blood flow. Biomed Opt Express 3: 3127-3137.
21.
Jia Y,
Wei E, Wang X, Zhang X, Morrison JC (2014) Optical coherence tomography
angiography of optic disc perfusion in glaucoma. Ophthalmology 121: 1322-1332.
22.
Yarmohammadi
A, Zangwill LM, Filho AD, Suh MH, Manalastas PI, et al. (2016) Optical
coherence tomography angiography vessel density in healthy, glaucoma suspect
and glaucoma eyes. Invest Ophthalmol Vis Sci 57: 451-459.
23.
(2012)
CIRRUS HD-OCT User Manual – Models 500, 5000 2660021169012 Rev. A 2017-2012.
24.
Akagi
T, Lida Y, Nakanishi H, Terada N, Morooka S, et al. (2016) Microvascular
density in glaucomatous eyes with hemifield visual field defects an optical
coherence tomography angiography study. Am J Ophthalmol 168: 237-249.
25.
Yarmohammadi
A, Zangwill LM, Filho AD, Saunders LJ, Suh MH, et al. (2017) Peripapillary and
macular vessel density in patients with glaucoma and single hemifield visual
field defect. Ophthalmology 124: 109-719.
26.
Caprioli
J, Coleman AL (2010) Blood pressure, perfusion pressure and glaucoma. Am J
Ophthalmol 149: 704-712.
27.
Yarmohammadi
A, Zangwill LM, Diniz Filho A, Suh MH, Yousefi S, et al. (2016) Relationship
between optical coherence tomography angiography vessel density and severity of
visual field loss in glaucoma. Ophthalmology 123: 2498-2508.
28.
Arend
O, Remky A, Cantor L, Harris A (2000) Altitudinal visual field asymmetry is
coupled with altered retinal circulation in patients with normal pressure
glaucoma. Br J Ophthalmol 84: 1008-1012.
29.
Yu J,
Jiang C, Wang X, Zhu Li, Gu R, et al. (2015) Macular perfusion in healthy
Chinese: An optical coherence tomography angiogram study. Invest Ophthalmol Vis
Sci 56: 3212-3217.
30.
Hollo
G (2018) Comparison of peripapillary OCT angiography vessel density and retinal
nerve fiber layer thickness measurements for their ability to detect
progression in glaucoma. J Glaucoma 27: 302-305.
31.
Liu L,
Jia Y, Takusagawa HL, Pechauer AD, Edmunds B, et al. (2015) Optical coherence
tomography angiography of the peripapillary retina in glaucoma; JAMA Ophthalmol
133: 1045-1052.
32.
Ohguro
I, Ohguro H (2012) The effects of a fixed combination of 0.5% timolol and 1%
dorzolammide on optic nerve head blood circulation. J Ocul Pharmacol Ther 28:
392-396.
33.
Mayama
C, Araie M (2013) Effects of anti-glaucoma drugs on blood flow of optic nerve
heads and related structures. Jpn J Ophthalmol 57: 133-149.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Stem Cell Research and Therapeutics (ISSN:2474-4646)
- International Journal of Surgery and Invasive Procedures (ISSN:2640-0820)
- Journal of Cell Signaling & Damage-Associated Molecular Patterns
- Journal of Cardiology and Diagnostics Research (ISSN:2639-4634)
- International Journal of Clinical Case Studies and Reports (ISSN:2641-5771)
- Journal of Spine Diseases
- Journal of Renal Transplantation Science (ISSN:2640-0847)