3119
Views & Citations2119
Likes & Shares
Objectives: This study investigated habits of mobile phone use, awareness on health effects of radiofrequency radiation (RFR) and modulation of selected inflammatory humoral markers; immunoglobulin A (IgA), interleukin-33 (IL-33) and myeloperoxidase (MPO) by mobile phone useamong a sample of the Jordanian Yarmouk university students.
Materials & Methods: One hundred volunteers (21.41±2.92 years) were randomly recruited and interviewed to fill questionnaire prior to collection of unstimulated saliva samples. Participants were divided into groups based on daily call time and history of phone use. The immunological response to RFR exposure was recorded by ELISA sandwich technique for quantitating the salivary levels ofIgA. Salivary MPO ODs was measured by colorimetric assay.
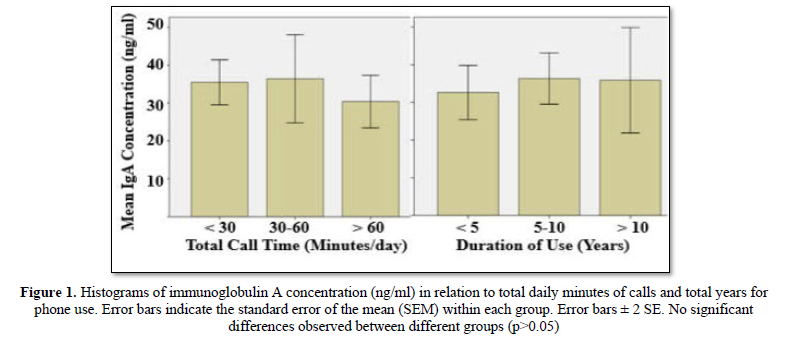
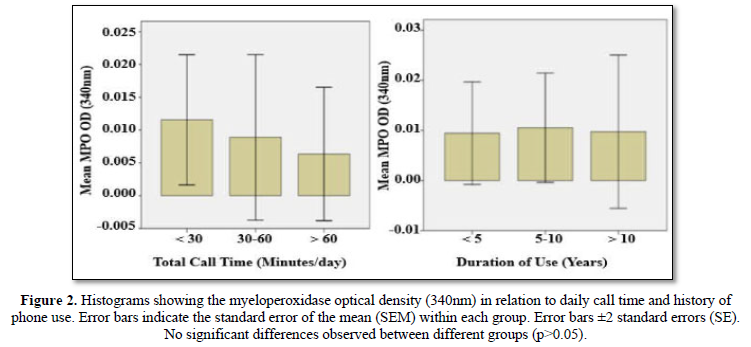
Results: It was revealed that participants were aware about mobile phone/radiation hazards as reflected by the notion that majority of them used precautionary measures and having a habit of putting mobile phone away from body to minimize unwanted effects. No significant correlations were observed between salivaryIgA and MPOlevels on one hand and intensity and duration mobile use on the other hand.
Conclusions: The possibility that prolonged and frequent exposure to RFR from mobile phone use may cause damage in the immune system cannot be excluded.
Keywords:Immunoglobulin A, Mobile phone, Myeloperoxidase, Radiofrequency radiation, Saliva biomarkers
INTRODUCTION
There is a great concern over harmful effects of electromagnetic and radiofrequency waves generated by mobile phones and their telecommunication stations. On average, mobile phones radiate a power in the range of 0.2-0.6 watt/kg, 40% of this energy is absorbed in the head and neck region [1]. Recently, a significant positive correlation between duration of mobile phone use and severity of neck pain has been demonstrated [2]. Parotid gland is the biggest salivary gland and its anatomic location, in front of ear and behind ramus, makes an ideal candidate for influence by exposure to mobile phones [3,4].Increase in the parotid gland volume in the dominant side than the nondominant side of mobile phone usage concurred with the findings of earlier studies [3-6]. One study [7] reported an association between exposure to mobile phone radiation for more than 1 h daily and possible development of parotid tumor. Histopathological changes in the parotid [8, 9] and the thyroid [10] glands of rats were linked to increased exposure duration RFR similar to that emitted by mobile phones.
Changes in the saliva and parotid gland as a result of exposure to mobile phone radiations [11]. Dentistry science underlines increasingly the significance of saliva in maintaining oral homeostasis and in protecting oral mucosa mechanically and immunologically [12]. Few in vitro human [13] or in vivo animal [14] studies have focused mostly on effects of exposure to Global System for Mobile communications-radiofrequency electromagnetic fields (GSM-REF) on endocrine responses or immune system. Previous studies showed that a significant decrease in antioxidant profile increases the risk of inflammatory diseases of the oral cavity such as gingivitis, periodontitis, and mucositis in individuals utilizing cell phones for longerdurations [15]. When the integrity of the dental pulp is threatened, pulp cells, especially fibroblasts, produce various pro-inflammatory cytokines and vascular endothelial growth factor (VEGF) [16]. The cytokine expression profile of the salivary gland in heavy cell phone users was investigated [17,18].
In comparison to contralateral parotids in subjects using cell phones for more than 10 years, differences in IL-10 levels in ipsilateral parotids as well asincreases in the salivary flow rate and alteration of the cytokine expression profile were reported [17]. In spite of the increasing popularity of mobile phones, the immune response to RF fields in humans is still unknown [18,19]. Intensive use of mobile phones has negative impact on bladder tissue as well as the other organs [19]. Therefore, minimizing level of mobile phone use makes it easy to be kept under control of diseases in which inflammation is an etiologic factor [19].
Saliva is readily available from most individuals, can be non-invasively collected, easily stored and processed. Recently, markers of inflammation in human saliva have been a subject of active research [20-23]. The potential use of saliva as alternative to frequent serum sampling to study inflammatory biomarkers has been suggested. Due to limited studies conducted in this field, the present research was carried out to explore the degree of awareness of phone users at Yarmouk University (Jordan) and to investigate whether frequency and duration of using mobile phone alter the health status by measuring levels of salivary inflammatory markers; IgA and MPO.
MATERIALS & METHODS
Subjects
This cross-sectional, descriptive-analytical study was approved by the Ethics Committee of Yarmouk University. A random sample of 100 healthy volunteer students from Yarmouk University in the age range of 18-30 years was recruited. Each participant was interviewed by the same person to explain the study’s aimand standardize data collection regarding the knowledge, attitude and habits of using mobile phones by completing a specially constructed questionnaire. The subject was asked to sign a consent form and he/she was assured that the provided data would be confidential and only for scientific purposes with no identifying information. Exclusion criteria consisted of chronic systemic diseases, previous head and neck injuries, and history of chemotherapy or radiotherapy. Participants having any oral lesions, signs of inflammation or infections, and tooth decay were excluded. So that any change in examined immunological parameters will probably be due to exposure to RFR. Participants were divided into three groups based on total time of daily calls; ˂30 min/day, 30-60 min/day and ˃60 min/day. The subjects were further classified in three categories based on the number of years using phone; ˂ 5 years, 5-10 years and ˃10 years.
Collection of saliva
This was done in the university lab settings. Before providing saliva sample, the subject was requested not to eat, drink or brush his/her teeth one hour before collection. All samples were collected daily in the morning between 10 and 12 am. A sample of 1.5-2.0 ml of unstimulated saliva (saliva in rest position without stimulated salivary gland) was collected for 15 min by spitting method into a sterile wide test tube provided by laboratory to the researcher. Salivary samples were kept on ice during and after sample collection. Samples were centrifuged for 20 min at 14000 g at 4 ⁰C to isolate probable debris. Then, the pure sample of saliva for each subject was kept at -80˚C for further analysis within a month period.
Measurement of IgA level
Immunoglobulin A level in saliva was estimated by using enzyme immunoassay (ELISA) technique according to the Human IgA ELISA kit (Abcam, UK), IgA assays were run in duplicates. Saliva samples were added to pre-coated 96 well microplate, followed by addition of antibody cocktail and incubated for 1 h. After washing, TMB substrate was added to each well and incubated for 10 min. Followed by stop solution. OD was obtained at 450 nm using multi scan microplate reader (Thermo, USA).
Interleukin 33 estimation
Interleukin 33 was estimated by ELISA test according to the Human IL-33 ELISA kit (Abcam, UK). IL-33 assays were run in duplicates. Saliva samples were added to pre-coated 96 well microplate, incubated for 2 h, followed by addition of biotin conjugated antibody, incubation for 1 hour, then the streptavidin-HRP was added and followed by TMB substrate. OD was obtained at 450 nm using multi scan microplate reader (Thermo, USA).
Determination of MPO level
Salivary MPO OD was measured using 3, 3′-diaminobenzidine (DAB,Bioworld, USA) as a substrate according previous work [24]. Serial dilution of Horse Radish Peroxidase HRP was used as a standard sample (5 mg/ml of HRP was prepared). The optimum absorption of 5 mg/1024 ml was at 340 nm). Each saliva sample (10𝜇L) was pipetted into 100 𝜇L of the 0.5 mM DAB solution (5 mg DAB in 50 mL of 0.1 M potassium dihydrogen phosphate pH 4.5). 50 𝜇L of 6 % H2O2 was added to initiate the reaction. Absorbance was measured at 340 nm.
Statistical Analysis
The collected questionnaires were revised for completeness and logical consistency. The data were analyzed using the Statistical Package for Social Sciences (SPSS) version 12.0 (SPSS; Chicago, Illinois, USA). Descriptive statistics was used to study the samples. One-way Analysis of Variance (ANOVA) test for numerical variables was used to compare between more than two independent groups. Statistical significance was defined as p ≤0.05.
RESULTS
This study included 100 healthy volunteers (45 males and 55 females) with a mean age of 21.41±2.92 years (range of 18-31 years). Sixty-nine of the respondents were between the age group of 18 and 21 years (Table 1). Table 1 summarizes habits of mobile phone usage among the study participants. The most favorite mobile phone brand was Huawei (45%). Mobile phone radiations ranged from 0.29 to 1.16 watt/kg. Ninety percent of students used right ear as the dominant side for mobile phone usage, only 10% used left as the dominant side for mobile phone usage and 8% were bilateral users. Most participants used precautionary measures to keep phone away from theirhead such as earphones (51%) and handheld devices (46%). Results indicated that more female participants used earphone than handheld device; 30 out of 55 (54.55%) and 25 out of 55 (45.45), respectively. Among males, the use of these two modes was equal; 21 out of 45 (46.67%). Very small margin of males; 3 (6.76%) and none of the females used the speaker mode (Gender differences data are not shown). The phone is left in silent position in 64% of the cases. Sixty eight percent of people used to keep phone away from body (near table or bed), 26% kept it in hand and few (6%) had the phone in the trousers/shirts pocket. The majority of the study group (74%) lived near phone base station and they were mostly (68%) nonsmokers.When participants were asked about their knowledge of phone health side effects, 89% responded by yes.The health risks associated with mobile phones include increased chances of anxiety or depression, lack of sleep, brain tumors and low sperm counts, headache, and hearing loss.
In the present study, fifty-seven percent of the study group used mobile phone for less than 30 min/day. Only 16% reported calls longer than 60 min/day. Thirty-seven students used phone less than 5 years, about half of the sample (47%) used mobile phone for a period between 5 and 10 years, the rest (16%) had a history longer than 10 years (Table 2). The results of ANOVA test analysis are presented in Table 2 and depicted in Figure 1.It was found that although shortest time use (˂5years) had lowered levels of salivary IgA, the differences were not statistically significant (P-value = 0.76). The IgA concentrations were 32.63±3.60, 36.34±3.39 and 35.89±7.00ng/ml in an increasing order of phone use; ˂5 years, 5-6 years and ˃10 years, respectively(Table 2andFigure 1). Similarly, total daily speaking time had no significant (P-value = 0.69) effect on IgA concentration; 30.38±3.48 (˃60 min), 36.38±5.80 (30-60 min), and 35.46±2.96 (˂30 min).
In an attempt to correlate between IgA and IL-33 on one hand and the mobile phone use on the other, we were unable of recording any picogram of IL-33 in the saliva of different studied groups. The IL-33 kit was tested with serum of a positive patient known by high score of C reactive protein (CRP) and matched with a healthy individual. Unfortunately, no absorbance readings were obtained at 450nm.This confirmed that the IL-33 kit was not sensitive enough to detect a probably extremely low salivary IL-33 level.
The MPO data are summarized in Table 3 and Figure 2. Results indicate that salivary MPO values were not significantly affected (P-value = 0.84), by the intensity of daily phone.Similarly, duration (year) of phone use had no significant (P-value=0.98) effect on the MPO value.
DISCUSSION
The present study is useful to the general population particularly to the students as the perceived health risk did not significantly deter students from using mobile phone. It showed that 89% students (mean age 21.41±2.924 years) reported a very good knowledge of adverse health impact of mobile phone radiation on their heads. Therefore, in addition to keeping their phones away from their bodies, they followed precautionary measures such as use of handheld sets or earphones. The third method was to reduce mobile using frequency to less than 30 min a day (57%). A study in Malaysia [25] reported an overall 62% perception of mobile phone hazard among 200 Medical School students.Another study [26] found that 50% of 400 Saudi final year medical students and medical interns had poor awareness of cell phone use and its health hazards. An average knowledge about physical hazards related to mobile phone usage; about 72% of an Indian studied group [27]. About half of 145 higher secondary school students in Nepal showed low level of knowledge on the mobile phone hazards [28]. More recently it was revealed[29] that only 60.7% of a sample of 150 Egyptian nursing students had knowledge of mobile phone cancer hazards.
Placing the phone at a distance of about 0.05m from the body during conversion has been highly recommended [30]. The latter study revealed that the intensity of measured field strength is about three times higher while dialing the network compared to when the call has been established in the network. As a consequence, it is advisable for a call to be established on the network before placing the phone to the ear.
In our study, no relationship was observed between age or gender and preference side of mobile phone use;90% used
right ear as the dominant side. This is in agreement with a previous study [6], where 39 out of 50 (78%) used right dominant.
Previous studies correlated EMF exposure to disturbance in the immune system including increased oxidative stress, enhanced phagocytic activity and increased production of chemokines [31-35]. This increases risk of inflammation and can predispose mobile phones users for longer durations to oral diseases such as gingivitis, periodontitis, and mucositis [15,35,36]. Decrease in salivary flow rate has been proposed to be partly responsible for the increase in salivary biochemical constituents in diseases [37]. Conversely, increase of the salivary flow rate in mobile-phone users was suggested to have a diluting effect on the saliva components [4].
In the present study, all confounding factors that could cause immune toxicity were excluded (tobacco, alcohol, recent medication, systemic factor etc.). Therefore, any change observed in the examined immunological parameters was expected to be the immediate result of phone use. To make sure that the observed alterations are attributed to radiation induced effects, people with gingivitis or people with periodontitis were not included in the study. We found that about 5 years of mobile use and more than 60 min of close exposure to EMF emitted by the phone had lowered, but not significantly, the concentrations of salivary IgA. Critical analysis of the data with the inflammation may relate the absence of the positive effect to the fact that most of the participants were using earphone (51%) or handheld set (46%) as a precautionary measure i.e. they were not keeping their phones near the ear while calling.
These results do not depart from those reported in literature, where levels of salivary flow, concentration of protein and of IgA in saliva [6,13,38-40] and blood [41] of people were not significantly affected by exposure to RF radiation. Contrary to this, speaking on mobile phone over an hour decreased total antioxidant capacity of saliva and salivary IgA [42]. Three studies on the effect of use of mobile phones on the level of salivary anti-inflammatory cytokine (IL-10) reported three different results; decrease [17], increase [43] and no change [44]. The proinflammatory cytokine IL-1β values in subjects who used mobile phones for more than 10 years presented higher differences between ipsilateral versus contralateral parotids [17].
Likewise, in vitro human studies gave no convincing evidence that exposure to RF field initiate adverse modifications in immune cells or cytokines characteristic of human disease [12,45-47]. It was proposed [48] that pulse-modulated microwaves may represent the potential of immunotropic influence, stimulating preferentially the immunogenic and proinflammatory activity of monocytes of cultured human blood at relatively low levels of exposure.A small but significant downregulation of expression of CD95 gene, which regulates immunologic response in lymphocytes, was found [49] in cells taken from older (88 ± 2 years), but not younger (26 ± 5 years) donors. Results from experiments with RFF exposure at 2.45GHz SAR at 10 W/kg have shown very little or no effects on either chemotaxis or phagocytosis in neutrophil-like human HL-60 cells [50].
Although it is better to limit the discussion on one model organism as the findings may be totally different (or to an extent) in two different species, it may deserve mentioning some of the similar studies. Animal studies reached no definite conclusions regarding the immunologic effects of mobile phone and microwave radiation; no change was detected in humoral response of young rats exposed in utero and postnatal to non-ionizing radiofrequency field regardless of the types of biomarker and SAR levels [51]. In contrast, exposure of rats to EMF resulted in significant decrease in immunoglobulin levels (IgA, IgE, IgM, and IgG); total leukocyte, lymphocyte, eosinophil and basophil counts [52]. The presence of more inflammatory cells especially large and small lymphocytes, which are characteristic of chronic inflammation, has been shown recently in gingival tissues of rabbits exposed to mobile phone radiation [53].
In the present work, the salivary IL-33 concentration was measured by ELISA, however, no absorbance was detected. This may due to the fact that IL-33 is detected in serum of people with chronic diseases, or that the IL-33 kit was not sensitive enough to detect a probably extremely low salivary IL-33 level [54]. The salivary MPO ODs values measured in this study were not significantly affected neither by time of call per day, nor by the duration (year) of phone use. Up-to-date, no experimental human studies describing changes in the salivary MPO levels due to the exposure RFR have been encountered in the published literature. The available literature on the effect of RF fields on MPO using laboratory animals is scarce. In line with our data are those found by others, who demonstrated no significant (p>0.05) alterations in MPO concentrations in livers [55]and submandibular glands [56] of rats exposed to 100 and 500 µT extremely low frequency magnetic field (ELF-MF) (2 h/day, 7 days/week, for 10 months) corresponding to the safety standards for public and occupational exposure [56,57]. In contrast, significant increases in MPO were observed in various organs, such as rat kidney and guinea pig’s liver after RFR exposure [58,59].
One point of strength of the present study is the fact it was conducted on both sexes. However, it is limited by small sample size and only with saliva from relatively young volunteers. The disadvantage of epidemiological studies; small sample size and a lack of prospective data acquisition should be kept in mind. For this reason, it is difficult to evaluate the results on a person basis. Another methodological issue concerns the different mobile phone types assessed in the study. Therefore, it is recommended that future studies should plan to examine whether or not the reported results herein represent adaptive response to radiation stress and to evaluate effect of frequent mobile phone use on salivary flow rate and other immunological parameters. To arrive at a more confirmatory conclusions sample size should be larger and with people of various age groups from different geographical regions. A sample of deaf people may serve as negative control for the mobile phone users.
CONCLUSION
Salivary IgA and MPO levels were lower, but not significantly, in saliva from people whose daily use exceeded 60min as compared with those observed in shorter period callers. Likewise, duration of phone use had no significant effect on the IgA and MPO values. However, higher IgA concentrations were noticed in saliva of subjects who used phone longer than 5 years as compared to scores of the shortest time less than 1 year. Whether or not the slight alterations in the immune system relevant to RFR have any clinical implications deserves further investigation.
ACKNOWLEDGEMENT
Authors would like to thank the Deanship of Scientific Research and Graduate Studies at Yarmouk University/Jordan for financially supporting this research (Grant number: 35/2018). Special thanks are extended to Dr. Mohamad Al Qaderi and Dr. AymanRawashdeh for their assistance in statistical analysis.
1. Gandhi G, Singh P (2005) Cytogenetic damage of mobile phone users: preliminary data. Int J Hum Genet 5: 259-265.
2. Hadidi FA, Bsisu I, Ryalat SAA, Zu'bi BA, Bsisu R, et al. (2019) Association between mobile phone use and neck pain in university students: A cross-sectional study using numeric rating scale for evaluation of neck pain. PLoS One 14: e0217231.
3. Goldwein O, Aframian DJ (2010) The influence of handheld mobile phones on human parotid gland secretion. Oral Dis 16: 146-150.
4. Hamzany Y, Feinmesser R, Shpitzer T, Mizrachi A, Hilly O, et al. (2013) Is human saliva an indicator of the adverse health effects of using mobile phones? Antioxid Redox Signal 18: 622-627.
5. Bhargava S, Motwani MB, Patni VM (2012) Effect of handheld mobile phone use on parotid gland salivary flow rate and volume. Oral Surg Oral Med Oral Pathol Oral Radiol 114: 200-206.
6. Ranjitha GE, Austin RD, Ramasamy S, Bharathi CS, Angeline D, et al. (2017) Influence of handheld mobiles on parotid: A cohort study. J Indian Acad Oral Med Radiol 29: 254-258.
7. Qahtani KA (2016) Mobile phone use and the risk of parotid gland tumors: A Retrospective case-control study. Gulf J Oncol 1: 71-78.
8. Aydogan F, Unlu I, Aydin E, Yumusak N, Devrim E, et al. (2015) The effect of 2100 MHz radiofrequency radiation of a 3G mobile phone on the parotid gland of rats. Am J Otolaryngol 36: 39-46.
9. Ghoneim FM, Arafat EA (2016) Histological and histochemical study of the protective role of rosemary extract against harmful effect of cell phone electromagnetic radiation on the parotid glands. ActaHistochem 118: 478-485.
10. Eşmekaya MA, Seyhan N, Omeroğlu S (2010) Pulse modulated 900 MHz radiation induces hypothyroidism and apoptosis in thyroid cells: A light, electron microscopy and immunohistochemical study. Int J Rad Biol 86: 1106-1116.
11. Dagli R, Hans R (2015) Effect of mobile phone radiations on oral health. J Int Oral Health 7: i-ii.
12. Augner C, Hacker GW, Oberfeld G, Florian M, Hitzl W, et al. (2010) Effects of exposure to GSM mobile phone base station signals on salivary cortisol, alpha-amylase and immunoglobulin A. Biomed Environ Sci 23: 199-207.
13. Tuschl H, Novak W, Djafari HM (2006) In vitro effects of GSM modulated radiofrequency fields on human immune cells. Bioelectromagnetics 273: 188-196.
14. Nasta F, Prisco MG, Pinto R, Lovisolo GA, Marino C, et al. (2006) Effects of GSM-modulated radiofrequency electromagnetic fields on B-cell peripheral differentiation and antibody production. Radiat Res 165: 664-670.
15. Dodwad R, Betigeri AV, Preeti B (2011) Estimation of total antioxidant capacity levels in saliva of caries-free and caries-active children. ContempClin Dent 2: 17-20.
16. Turner MD, Nedjai B, Hurst T, Pennington DJ (2014) Cytokines and chemokines: At the crossroads of cell signaling and inflammatory disease. BiochimBiophysActa 1843: 2563-2582.
17. Siqueira EC, Souza FTD, Ferreira E, Souza RP, Macedo SC, et al. (2016) Cell phone use is associated with an inflammatory cytokine profile of parotid gland saliva. J Oral Pathol Med 45: 682-686.
18. Mishra SK, Chowdhary R, Kumar S, Rao SB (2017) Effect of cell phone radiations on orofacial structures: A systematic review. J Clin Diagn Res 11: ZE01-ZE05.
19. Koca O, Gokce AM, Akyuz M, Ercan F, Yurdakul N, et al. (2014) A new problem in inflammatory bladder diseases: Use of mobile phones! IntBraz J Urol 40: 520-525.
20. Khurshid Z, Zafar MS, Khan RS, Najeeb S, Slowey PD, et al. (2018) Role of salivary biomarkers in oral cancer detection. Adv Clin Chem 86: 23-70.
21. Smith M, Sharma P, Dahiya S (2018) The validation of salivary C-reactive protein as a marker of inflammation in humans. Rheumatology 57: key075.479.
22. Nascimento GG, Baelum V, Sorsa T, Tervahartiala T, Skottrup PD, et al. (2019) Salivary levels of MPO, MMP-8 and TIMP-1 are associated with gingival inflammation response patterns during experimental gingivitis. Cytokine 115: 135-141.
23. Pay JB, Shaw AM (2019) Towards salivary C-reactive protein as a viable biomarker of systemic inflammation. ClinBiochem 68: 1-8.
24. Klangprapan S, Chaiyarit P, Hormdee D, Kampichai A, Khampitak T, et al. (2016) Salivary myeloperoxidase, assessed by 3, 3-Diaminobenzidine colorimetry, can differentiate periodontal patients from nonperiodontal subjects. Enzyme Res 2016: 7517928.
25. Kumar LR, Chii KD, Way LC, Jetly Y, Rajendaran V (2011) Awareness of mobile phone hazards among university students in a Malaysian medical school. Health 3: 406-415.
26. Muhayawi SA, Eldeek B, Abubakr H, Kuddah RB, Zahid A, et al. (2012) The impact of medical education on Saudi medical students’ awareness of cell phone use and its health hazards. Life Sci J 9: 1143-1148.
27. Pendse N, Zagade T (2014) Knowledge and attitude regarding health hazards of mobile phone users among the junior college students. Int J Sci Res 3: 554-561.
28. Gautam S, Shakya J (2016) Knowledge regarding harmful effects on cell phone use among higher secondary school students. BharatpurChitwan, Nepal. J Chitwan Med Coll 6: 47-53.
29. Nasser S, Amer NM, Ghobashi MM, Morcos G, Hafez SF, et al. (2018) Knowledge, attitude, and practices (KAP) study and antioxidant status among mobile phone users. Biosci Res 15: 3658-3664.
30. Isabona J, Srivastava VM (2017) Cellular mobile phone - A technical assessment on electromagnetic radiation intensity on human safety. IEEE 3rd International Conference on Electro-Technology for National Development (NIGERCON). 2017: 271-274.
31. Johansson O (2009) Disturbance of the immune system by electromagnetic fields: A potentially underlying cause for cellular damage and tissue repair reduction which could lead to disease and impairment. Pathophysiology 16: 157-177.
32. Bilgici B, Akar A, Avci B, Tuncel OK (2013) Effect of 900-MHz radiofrequency radiation on oxidative stress in rat brain and serum. ElectromagnBiol Med 32: 20-29.
33. Khadra KMA, Khalil AM, Samak MA, Aljaberi A (2015) Evaluation of selected biochemical parameters in the saliva of young males using mobile phones. ElectromagnBiol Med 34: 72-76.
34. Guerriero F, Ricevuti G (2016) Extremely low frequency electromagnetic fields stimulation modulates autoimmunity and immune responses: A possible immuno-modulatory therapeutic effect in neurodegenerative diseases. Neural Regen Res 11: 1888-1895.
35. Khan A, Naheed S, Alam M, Salahuddin S (2019) Saliva-A new horizon for estimating antioxidant profile of mobile phone user. RADS J Biol Res Appl Sci 10: 1-7.
36. Kolanjiappan K, Ramachandran C, Manoharan S (2003) Biochemical changes in tumor tissues of oral cancer patients. ClinBiochem 36: 61-65.
37. Navazesh M (2003) Methods for collecting saliva. Ann NY AcadSci 20: 72-74.
38. Radon K, Parera D, Rose DM, Jung D, Vollrath L (2001) No effects of pulsed radio frequency electromagnetic fields on melatonin, cortisol, and selected markers of the immune system in man. Bioelectromagnetics 22: 280-287.
39. Souza FTD, Silva JFC, Ferreira EF, Siqueira EC, Duarte AP, et al. (2014) Cell phone use and parotid salivary gland alterations: No molecular evidence. Cancer EpidemiolBiomarkPrev 23: 1428-1431.
40. Hashemipour MS, Yarbakht M, Gholamhosseinian A, Famori H (2014) Effect of mobile phone use on salivary concentrations of protein, amylase, lipase, immunoglobulin A, lysozyme, lactoferrin, peroxidase and C-reactive protein of the parotid gland. J LaryngolOtol 128: 454-462.
41. Yuan ZQ, Li F, Wang DG, Wang Y, Zhang P (2004) Effect of low intensity and very high frequency electromagnetic radiation on occupationally exposed personnel. Zhonghua Lao Dong Wei Sheng Zhi Ye Bing ZaZhi 22: 267-269.
42. Kalati FA, Salimi S, Rabiee AV, Noraeei M (2014) Effect of mobile phone usage time on total antioxidant capacity of saliva and salivary immunoglobulin A. Iran J Public Health 43: 480-484.
43. Pedrami A, Kalti FA (2019) Evaluation of the effect of cellular phones on salivary levels of IL-10. Iran J Pub Health 48: 367-368.
44. Taheri M, Roshanaei G, Ghaffari J, Rahimnejad S, Khosroshahi BN, et al. (2017) The effect of base transceiver station waves on some immunological and hematological factors in exposed persons. Hum Antibod 25: 31-37.
45. Selmaoui B, Bogdan A, Auzeby A, Lambrozo J, Touitou Y (1996) Acute exposure to 50 Hz magnetic field does not affect hematologic or immunologic functions in healthy young men: a circadian study. Bioelectromagnetics 17: 364-372.
46. Capri M, Salvioli S, Altilia S, Sevini F, Remondini D, et al. (2006) Age-dependent effects of in vitro radiofrequency exposure (mobile phone) on CD95+ T helper human lymphocytes. Ann NY AcadSci 1067: 493-499.
47. Selmaoui B, Lambrozo J, Lundeen LS, Haus E, Touitou Y (2011) Acute exposure to 50-Hz magnetic fields increases interleukin-6 in young healthy men. J Clin Immunol 31: 1105-1111.
48. Dabrowski MP, Stankiewicz W, Kubacki R, Sobiczewska E, Szmigielski S (2003) Immunotropic effects in cultured human blood mononuclear cells pre-exposed to low-Level 1300 MHz pulse-modulated microwave field. ElectromagBiol Med 22: 1-13.
49. Kazemi E, Mortazavi SMJ, Ali-Ghanbari A, Sharifzadeh S, Ranjbaran R, et al. (2015) Effect of 900 MHz electromagnetic radiation on the induction of ROS in human peripheral blood mononuclear cells. J Biomed PhysEng 5: 105-114.
50. Koyama S, Narita E, Suzuki Y, Taki M, Shinohara N (2015) Effect of a 2.45-GHz radiofrequency electromagnetic field on neutrophil chemotaxis and phagocytosis in differentiated human HL-60 cells. J Radiat Res 56: 30-36.
51. Aïssa SA, Billaudel B, Gannes FPD, Ruffié G, Duleu S, et al. (2012) In utero and early-life exposure of rats to a Wi-Fi signal: Screening of immune markers in sera and gestational outcome. Bioelectromagnetics 33: 410-420.
52. Gohary OAE, Said MA (2017) Effect of electromagnetic waves from mobile phone on immune status of male rats: Possible protective role of vitamin D. Can J PhysiolPharmacol 95: 151-156.
53. Sirajuddin S, Kripal K, Chandrasekaran K, Anuroopa P (2018) Effects of electromagnetic radiations from mobile phone on gingiva in the era of 4g Lte-An in vivo study in rabbits. Dentistry 8: 10.
54. Tawfik MS, Elnasr MS, Elkady AA, Alkady MM, Hawas AM (2018) Protective role of ferulic acid against the damaging effect induced by electromagnetic waves on rat liver and intestine tissues. Int J Radiat Res 16: 421-430.
55. Dasdag S, Bilgin HM, Akdag MZ, Celik H, Aksen F (2014) Effect of long-term mobile phone exposure on oxidative-antioxidative processes and nitric oxide in rats. Biotechnol Equip 22: 992-997.
56. Akdag M, Akdag MZ, Dasdag S, Erel O (2014) Oxidative and antioxidative responses in submandibular and parotid glands of rats exposed to long-term extremely low frequency magnetic field. J Clin Exp Invest 5: 219-225.
57. Dasdag S, Akdag MZ, Meric F, Uzunlar AK, Celik MS, et al. (2011) Effect of extremely low frequency magnetic field and mobile phone exposure on nasal mucosa and nose skin. BiotechnolBiotechnol Equip 25: 2273-2278.
58. Oktem F, Ozguner F, Mollaoglu H, Koyu A, Uz E (2005) Oxidative damage in the kidney induced by 900-MHz-emitted mobile phone: protection by melatonin. Arch Med Res 36: 350-355.
59. Ozgur E, Güler G, Seyhan N (2010) Mobile phone radiation-induced free radical damage in the liver is inhibited by the antioxidants N-acetyl cysteine and epigallocatechin-gallate. Int J RadiatBiol 6: 935-945.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Veterinary and Marine Sciences (ISSN: 2689-7830)
- Food and Nutrition-Current Research (ISSN:2638-1095)
- Journal of Agriculture and Forest Meteorology Research (ISSN:2642-0449)
- Journal of Microbiology and Microbial Infections (ISSN: 2689-7660)
- Proteomics and Bioinformatics (ISSN:2641-7561)
- Advances in Nanomedicine and Nanotechnology Research (ISSN: 2688-5476)
- Journal of Genomic Medicine and Pharmacogenomics (ISSN:2474-4670)