Review Article
Phytosomes: A Promising Approach for the Delivery of Bioactive Compounds
3450
Views & Citations2450
Likes & Shares
Phytomedicines and other complex chemical compounds obtained and prepared from plants have been used extensively for the maintenance of health since ages. But the main limitation in using phytomedicines is their effectiveness because they are poorly absorbed when taken orally. The term “phyto” means plant while “some” means cell-like. Phytosomes are little cell like structure which is an advanced form of herbal formulations containing the bioactive phyto-constituents of herb extract surrounding a lipid boundary. Mainly bioactive constituents of phytomedicines are water-soluble compounds like flavonoids, glycosides; terpenoids in which flavonoids are used in a major proportion for their various therapeutic activities. As the phytosomes contain water soluble herbal extract and lipophilic outer layer they elicit better absorption which in turn produces better bioavailability and actions than the conventional herbal dosage forms. Phytosomes are produced by a process where the standardized plant extract or its constituents are bound to phospholipids, mainly phosphatidylcholine producing a lipid compatible molecular complex. This review enlists various aspects of phytosome formulation such as the advantages, methods of preparation, characterization, evaluation and biomedical applications of phytosome formulations.
Keywords: Phytosomes, Herbal medicine, Novel drug delivery system
INTRODUCTION
The conventional systemic drug delivery systems are susceptible to various limitations such as off-target effects, adverse toxic reactions, and quick circulation time leading to undesired side effects. Targeted therapy therefore assembled the research interest widely to the aforementioned dilemma.
Vesicular drug delivery system (VDDS) includes liposomes, niosomes, aquasomes and phytosomes are reported to be an ideal approach which satisfies all the requirements for being an effective drug carrier. VDDS are micelles containing an aqueous core and a lipid bilayer outer shell. The inner aqueous core entraps the hydrophilic drugs, while lipid bilayer encloses the various lipophilic moieties. This dual capability makes the VDDS an effective vehicle for delivery of both categories of drugs [1].
Phytosomes amidst the VDDS are distinctive as they contain the bioactive compounds as their core formulation accompanied with phospholipid. They are generally micelles formulated by colligation of aromatic active phytoconstituents such as flavonoids, terpenoids, and tannins to phospholipids in a nonpolar solvent. The lipid bilayer of the phytosomes assists ‘contact-facilitated drug delivery (CFDD)’ whereby there occurs lipid-lipid interaction between the carrier and the cell membrane followed by diffusion of bioactive compounds into the cell. Indena’s phytosome products and numerous studies conducted on them prove that they are significantly better absorbed and have considerably higher clinical efficacy. The PHYTOSOMETM technology is a breakthrough model for:
- Significantly higher therapeutic benefit.
- Promised delivery to the tissues.
- No question of nutrient safety.
- Efficient enhancement of bioavailability [2].
This review deals with the various aspects of phytosome formulation such as the advantages, methods of preparation, characterization, evaluation and biomedical applications of phytosome formulations.
PRINCIPLE
Phosphatidylcholine (or phosphatidylserine) is a difunctional compound. The phosphatidyl moiety is lipophilic and the choline (serine) moiety is hydrophilic in nature. This duplex solubility profile of the phosphatidylcholine makes it a promising emulsifier. Therefore, the choline head of the phosphatidylcholine molecule coheres to the compounds while the lipid soluble phosphatidyl portion containing the body and tail then encircles the choline bound material. Thereby, the phytoconstituents create a lipid compatible molecular complex with phospholipids (also called as phytophospholipid complex) [3].
Strength of phytosomes
Phytosomes exhibiten enhanced stability as chemical bond is formed between phospholipid molecule and phytoconstituent (s). The dose of phytoconstituents is lowered due to increased bioavailability of the phytoconstituents in the complex form. Duration of action is elevated. Phytoconstituents complex with phospholipids are more stable in gastric sections and resist the action of bacteria present in the gut. The permeability of phytoconstituents across the biological membranes is enhanced. Absorption of lipid insoluble polar phytoconstituents via different routes shows better absorption and significantly higher therapeutic effects.
Phosphatidylcholine used in the formulation of phytosomesalso possess several therapeutic properties contributing to synergistic effect when specific substance is given.
Drug entrapment is not an issue inphytosomes as the complex is biodegradable [4].
Difference between phytosome and liposome
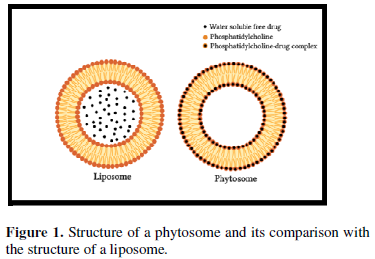
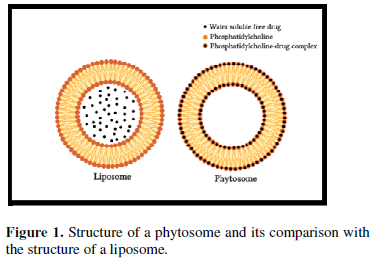
The basic difference between liposomes and phytosomes is that in liposomes the active constituent is dissolved in the medium enclosed in the cavity or in the layers of the membrane, whereas in phytosome, it is a part of the membrane, the molecules anchored through chemical bonds to the polar head of the phospholipid (Figure 1).
Liposomes are used in cosmetic formulations for delivery of water-soluble substances to the skin. A liposome is produced by mixing a water-soluble substance with phosphatidylcholine. No chemical bond is formed; the phosphatidylcholine molecules enclose the water-soluble constituent. There may be hundreds or thousands of phosphatidylcholine molecules enclosing the water-soluble compound. Whereas, in Phytosomes, the phosphatidylcholine and the single herbal compound actually form a 1:1 or a 2:1 complex depending on the substance. This difference results in Phytosomes being much better absorbed than liposomes. Phytosomes are superior to liposomes in skin care products [5].

Advantages of phytosomes
Phytosomes have the following advantages [6]:
- Easy transport from the cell membrane and cellular entry.
- Enhanced bioavailability of drug occurs.
- Ensured increase in duration of action for herbal drugs.
- Enhanced absorption of hydrophilic polar phytoconstituents through oral, topical and other route thereby increased bioavailability.
- Protection of valuable components of herbal extracts from destruction by digestive secretions and gut bacteria.
- Phytosomes ensures proper delivery of drug to the respective tissues.
- The nutrient safety of the herbal extracts does not get compromised by phytosome formulation.
- Reduced dose requirement due to the maximum absorption of active constituents.
- Improved absorption of biologically active constituent.
- Chemical bonds formed between phosphatidylcholine molecule and phytoconstituents provides good stability profile of phytosomes.
- Improved transdermal absorption of phytoconstituents therefore widely used in cosmetic formulations for their enhanced skin penetration and high lipid profile.
- The phytoconstituents in phytosomes can readily permeate the intestinal walls of tissues and better absorbed.
- The complex is biodegradable and drug entrapment is not an issue with phytosome.
- Intensified effect of herbal compounds by improved absorption enhanced biological activity, and delivery to the target tissue; therefore, phytosomes are suitable as targeted delivery system.
- High entrapment efficiency as the drug itself is in conjugation with lipids in the vesicles.
- No problem in drug entrapment while formulating phytosomes.
- Phosphatidylcholine nourishes the skin besides acting as a carrier as it is an integral part of plasma membrane.
- Phytosomes are also better than liposomes in terms of topical delivery.
- Phytosomes have significantly greater therapeutic benefits.
- Phosphatidylcholine also acts as a hepatoprotective and imparts a synergistic effect when hepatoprotective substances are incorporated.
- Low solubility in aqueous media allows the formation of stable semisolid dosage form.
- Promotes liver targeting by increasing the solubility in bile salt.
PROPERTIES OF PHYTOSOMES
Physico-chemical properties
Phytosome is a complex between a natural product and phospholipids obtained by a reaction of stoichiometric amounts of phospholipids and the substrate in an appropriate solvent. On the basis of spectroscopic data, it has been observed that the main phospholipid-substrate interaction is due to the formation of hydrogen bonds between the polar head of phospholipids (i.e., phosphate and ammonium groups) and the polar functionalities of the substrate. On treatment with water, phytosomes acquires a micellar shape forming liposomal structure which can be concluded by comparison of the NMR of the complex with that of the pure precursors. The signals of the fatty chain are almost unchanged. Such evidences referred that the two long aliphatic chains are wrapped around the active content, producing a lipophilic envelope, which shields the polar head of the phospholipid and active constituents [7].
Biological properties
Phytosomes are improved forms of herbal formulations that are better absorbed, utilized and produce better results than conventional herbal extracts. The increased bioavailability of the phytosome over the non-complexed herbal derivatives has been demonstrated by pharmacokinetic studies or by pharmacodynamic tests in experimental animals and in human subjects [8].
METHODS OF PREPARATION FOR PHYTOSOMES
Anti-solvent precipitation technique
The specific amount of plant extract and phospholipid were taken into a 100 ml round bottom flask and refluxed with 20 ml of dichloromethane at a temperature not exceeding 60°C for 2 h. The mixture was then concentrated to 5-10 ml. Hexane (20 ml) was added carefully with continuous stirring to obtain the precipitate which was filtered and collected and stored in desiccators overnight. The dried precipitate was crushed in mortar and sieved through #100 mesh. Powdered complex was placed in amber colored glass bottle and stored at room temperature [9].
Rotary evaporation technique
The specific amount of plant material and phospholipid were dissolved in 30 ml of tetrahydrofuran in a rotary round bottom flask followed by stirring for 3 hours at a temperature not exceeding 40°C. Thin film of the sample was obtained to which n-hexane was added and continuously stirred using a magnetic stirrer. The precipitate obtained was collected, placed in amber colored glass bottle and stored at room temperature.
Solvent evaporation technique
The specific amount of plant material and phospholipids were taken into a 100ml round bottom flask and refluxed with 20ml of acetone at a temperature 50-60°C for 2h. The mixture was concentrated to 5-10 ml to obtain the precipitate which was filtered and collected. The dried precipitate phytosome complex was placed in amber colored glass bottle and stored at room temperature.
Ether-injection technique
In this technique, the drug lipid complex is dissolved in an organic solvent. The mixture is then slowly injected into a heated aqueous agent, resulting in the formation of vesicles. The state of amphiphiles depends on the concentration. When the concentration is less, amphiphiles introduce a monomer state but as the concentration is increased, variety of structures may be formed, that is, round, cylindrical, disc, cubic, or hexagon type [10].
CHARACTERIZATION OF PHYTOSOMES
Physical attributes
The following are the characterization techniques used for Phytosomes [11]:
- Visualization: Visualization of phytosomes can be done using transmission electron microscopy (TEM) which provides details about internal composition such as morphology, crystallization, stress or even magnetic domains. Scanning electron microscopy (SEM) reveals the surface and its composition and provides morphological details of the phytosomes.
- Particle size and zeta potential: The particle size and zeta potential can be determined by dynamic light scattering (DLS) using a computerized inspection system and photon correlation spectroscopy (PCS).
- Entrapment efficiency: The entrapment efficiency is determined by the ultracentrifugation technique. It gives an idea about the % drug that is successfully entrapped inside the phytosomes.
- Transition temperature: The transition temperature of the phytosomes is determined by differential scanning calorimetry (DSC).
- Surface tension activity measurement: The surface tension activity of the drug in aqueous solution can be measured by the ring method in a Du Nouy ring tensiometer.
- Vesicle stability: The stability of vesicles can be determined by assessing the size and structure of the vesicles over time. The mean size is measured by DLS and structural changes are monitored by TEM.
- Drug content: The drug content can be estimated by modified high performance liquid chromatographic method or by a suitable spectroscopic method.
Spectroscopic Evaluation
The spectroscopic evaluations are conducted to confirm the formation of complex between phytoconstituents and the phospholipids moiety and also to study the corresponding interaction between the two [4].
- 1H-NMR: Using this method, the complex formation between the active phytoconstituents and the phosphatidylcholine molecule can be estimated.
- 13C-NMR: Using this method, the phytoconstituents and the stoichiometric complex with the phosphatidylcholine when recorded the phytoconstituents carbons were invisible. The signals corresponding to the glycerol and choline portion are broadened and some are shifted, while most of the resonance of the fatty acid chains retains their original sharp line shape.
- FTIR: The formation of the complex can be confirmed by IR spectroscopy by comparing the spectrum of the complex with the spectrum of the individual components and their mechanical mixtures.
In-Vitro and In-Vivo Evaluations
Models of in-vitro and in-vivo evaluations are selected on the basis of the expected therapeutic activity of the biologically active phytoconstituents present in the phytosomes [12].
APPLICATIONS OF PHYTOSOME AND COMMERCIALLY AVAILABLE PHYTOSOME TECHNOLOGY
Silymarin phytosomes
Most of the Phytosomal studies are concentrated on Silybummarianum (milk thistles) which carriesexclusive liver protectant flavonoids. Yanyu [13] formulated silymarin phytosome and evaluated its pharmacokinetic activity in rats. In the experiment, the bioavailability of silybin in rat was increased significantly after oral administration of silybin-phospholipid complex due to magnificent improvement of the lipophilic properties of silybin-phospholipid complex and improvement of biological effect of silybin. Tedesco et al (2004) reported Silymarin phytosome exhibit better anti-hepatotoxic activity than silymarin alone and can provide protection against the toxic effects of aflatoxin B1 on performance of broiler chicks [13].
Phytosomes of gingko biloba leaves
Studies have revealed that gingko phytosomes (prepared from standardized extract of Ginkgo bilobaleaves) exhibited better results compared to the conventional standardized extract from plant (GBE, 24% ginkgo flavones glycoside and 6% terpenes lactones). In a bioavailability study conducted with healthy human volunteers the level of GBE constituents (flavonoids and terpenes) from the Phytosomal form peaked after 3 hours and sustained longer for at least 5 hours after oral administration. It was observed that the Phytosomal GBE produced a 2-4 times higher plasma concentration of terpenes than the non-Phytosomal GBE. Its major indication is cerebral insufficiency and peripheral vascular disorders and it can also ameliorate reduced cerebral circulations. Its improved oral bioavailability and high tolerability makes it the ideal gingko formulation even for long term treatment. Studies have also showed the improved efficacy of ginkgo phytosomes over the conventional standardized extract in protecting rat isolated hearts against ischemia [14].
Curcumin phytosomes
Maiti [14] formulated the phytosomes of curcumin (flavonoid from Curcuma longa, turmeric) and naringenin (flavonoid from grape fruit, Vitisvinifera) in two different studies. The antioxidant activity of the complex was markedly greater than pure curcumin in all dose level tested. In the second study, the formulated phytosome of naringenin exhibited better antioxidant activity than the free compound with a sustained duration of action, which may be due to decrease in the rapid elimination of the molecule from body [14].
Phytosomes of grape seed extract
Phytosomes of grape seed extract is made of oligomeric polyphenols (grape proanthocyanidins or Procyanidin from grape seed extract, Vitisvinifera) of different molecular sizes complexed with phospholipids. The main properties of Procyanidin flavonoids of grape seed include high total antioxidant capacity and stimulation of immune response of plasma, protection from ischemia/reperfusion induced damage in the heart, protective effects against atherosclerosis hence displaying enhanced protection for the cardiovascular system and other organs through a network of mechanism that extend beyond their antioxidant effect [15].
Phytosome of olive (Olea Europaea) oil
A commercially available phytosome- Oleaselect phytosome is present in the market based on olive oil polyphenols [16]. It is a strong antioxidant, anti-inflammatory and anti-hyper-lipidemic. It inhibits the oxidation of LDL cholesterol and is cardioprotective.
Phytosomes of green tea
Green tea leaves (Theasinensis) are famous for the presence of a polyphenolic compound epigallocatechin 3-O-gallate as the key compound. These compounds are potent modulators of various biochemical processes linked to the breakdown of homeostasis in serious chronic-degenerative diseases such as cancer and atherosclerosis. Green tea exhibits various beneficial activities such as antioxidant, anticarcinogenic, antimutagenic, hypocholesterolemic, and cardioprotective effects [17].
Quercetin-phospholipid phytosomal complex
Maiti [14] formulated the quercetin-phospholipid Phytosomal complex by a simple and reproducible method and also claimed that the formulation exhibited better therapeutic efficacy than the molecule in rat liver injury induced by carbon tetrachloride [17].
Some Patented Technologies Related to Phytosomes
Following are the innovative processes and formulation research studies in the field of phytosomes carried out by a number of academic scientists as well as by industrial laboratories. Some patents for phytosomes and other related technologies along with their applications and innovations are listed in Table 1 [8].

Cost-Benefit Analysis of Phytosome Technology
Aside from the modern developments in industrial-scale production of vesicular systems, comprising extruding methods, which offers hopeful prospective for the commercial production of these vesicular systems, the high cost of raw materials viz., PEGylated soy phosphatidylcholine may endanger this advancement. In spite of the endangering limitations ushering the formulation of phytosomes, this concept and technology bears a promising potential in its applications. In the forthcoming future, phytosomal delivery systems will stand to advance and grow for clinical therapy. It is obvious that the engineering of herbal and synthetic agents into phytosomal complexes can remarkably enhance oral bioavailability. Although there are some limitations in the manufacturing process, phytosomes have sufficient advantages to qualify for industrial production (at the pharmaceutical and food industrials) for eliciting therapeutic effects along with developing medicinal-functional foods (Table 2) [18].

CONCLUSION
The absorption of phytosomes in gastro intestinal tract is substantially higher resulting in the increased plasma level of the phytoconstituent than the conventional formulation. Complex formation ratio of component and phospholipids is 1:1 and 2:1. Phytosomes are widely used as a medicament and have wide scope in cosmeticology. A wide area of phytosome technology is yet to be revealed in future in the prospect of various pharmaceutical applications. Phytosomes forms an appreciable bridge between the conventional delivery systems and novel delivery systems.
ACKNOWLEDGEMENT
The authors wholeheartedly acknowledge the laboratory facilities and guidance provided by. The Director of Regional Institute of Paramedical and Nursing Sciences, Aizawl, India for carrying out this work and giving such a great opportunity to explore the recent technology and development in the field of Pharmaceutical Sciences.
- Azeez NA, Deepa VS, Sivapriya V (2018) Phytosomes: Emergent promising nano vesicular drug delivery system for targeted tumor therapy. Adv Nat Sci: Nanosci Nanotechnol 9: 033001.
- Shivanand P, Kinjal P (2010) Phytosomes: Technical revolution in phytomedicine. Int J PharmTech Res 2: 627-631.
- Gao L, Liu G, Wang X, Liu F, Xu Y, et al. (2011) Preparation of a chemically stable quercetin formulation using nanosuspension technology. Int J Pharm 404: 231-237.
- Pawar HA, Bhangale BD, Deshpande PK (2016) Phytosomes : A Noval Drug Delivery System for Phytoconstituents Phytosomes : A Noval Drug Delivery System for Phytoconstituents. J Chem Pharm Res 7: 144-160.
- Manglani N, Shilpa V (2012) Phytosomes : A Novel Herbal Drug Delivery System. J Pharm Sci Innov 1: 35-40.
- Thurapati PR, Reddy MS, Veerareddy PR (2011) Phyto-complexs, a novel phyto-phospholipid carriers for herbal drug delivery. Int Res J Pharm 2: 28-33.
- Singh RP, Narke R (2015) Preparation and Evaluation of Phytosome of Lawsone. Int J Pharm Sci Res 6: 5217-5226.
- Saha S, Sarma A, Saikia P, Chakrabarty T (2013) Phytosome : A Brief Overview. Sch Acad J Pharm 2: 12-20.
- Rathore P, Swami S (2012) Planterosomes: A potential phyto-phospholipid carriers for the bioavailability enhancement of herbal extracts. Int J Pharm Sci Res 3: 737-755.
- Saini V, Rani B, Nagpal M, Arora S (2013) Phytosomes: Potential carriers for herbal drugs. Am J PharmTech Res 3: 250-260.
- Changediya V, Khadke M, Devdhe S (2011) Phytosomes: New approach for delivering herbal drug with improved bioavailability. Res J Pharm Biol Chem Sci 2: 57-68.
- Awasthi R, Kulkarni GT, Pawar VK (2011) Phytosomes: An approach to increase the bioavailability of plant extracts. Int J Pharm Pharm Sci 3: 1-3.
- Yanyu X, Yunmei S, Zhipeng C, Qineng P (2006) The preparation of silybin-phospholipid complex and the study on its pharmacokinetics in rats. Int J Pharm 307: 77-82.
- Maiti K, Mukherjee K, Gantait A, Saha BP, Mukherjee PK (2007) Curcumin-phospholipid complex: Preparation, therapeutic evaluation and pharmacokinetic study in rats. Int J Pharm 330: 155-163.
- Kidd PM (2009) Bioavailability and activity of phytosome complexes from botanical polyphenols: The silymarin, curcumin, green tea, and grape seed extracts. Alt Med Rev 14: 226-246.
- Patel J, Patel R, Khambholja K, Patel N (2009) An overview of phytosomes as an advanced herbal drug delivery system. Asian J Pharm Sci 4: 363-371.
- Maiti K, Mukherjee K, Gantait A, Ahamed HN, Saha BP, et al. (2005) Enhanced therapeutic benefit of quercetin– phospholipid complex in carbon tetrachloride-induced acute liver injury in rats: A comparative study. Iran J Pharmacol Ther Res 405: 1735-2657.
- Babazadeh A, Jafari SM, Shi B (2019) Encapsulation of food ingredients by nanophytosomes: Lipid-Based Nanostructures for Food Encapsulation Purposes 2: 405-443.
- Sravanthi M, Krishna JS (2013) Phytosomes: A Novel Drug Delivery for Herbal Extracts. Int J Pharm Sci Res 4: 949-959.