149
Views & Citations10
Likes & Shares
MATERIALS AND METHODS
Calluses were obtained from zygotic embryos extracted from mature soyamiche palm seeds. The seeds were acquired in the community of “El mezón” located in the municipality of Ayutla de Los Libres, Guerrero (latitude 16°50'44“N and longitude 99°3'29”W). The plant material was then transferred to the plant science department intothe tissue culture laboratory at the Autonomous University of Chapingo. The seeds were washed with water, soap, and Tween 20, then disinfected in a laminar flow chamber by immersion in 70% (v/v) ethyl alcohol for three minutes, 10% (v/v) sodium hypochlorite for 15 minutes, followed by triple washing in sterile distilled water. The zygotic embryos were then extracted with a sterile manual stainless steel press and seeded in Murashige and Skoog (1962) MS (Sigma, St. Louis, MO) supplemented with 0.40 mg·L-1 thiamine (ICN Pharmaceuticals, Plainview, N.Y), 100 mg·L-1 myo-inositol (Sigma, St. Louis, MO), 100 mg·L-1 L-cysteine (Sigma, St. Louis, MO) , 50 mg·L-1 polyvinylpyrrolidone, 30 g·L sucrose, and 5.0 mg·L-1thidiazuron to induce callus formation. The explants were incubated in the dark at 25 °C. During the embryo formation phase, in addition to the above supplements (except for thidiazuron), the following were added to the medium: 400 mg· L-1 glutamine, 100 mg·L-1 arginine, 100 mg·L-1 asparagine, 10 g·L-1 hydrolyzed casein, 1 g mg·L-1 activated carbon, kinetin, and 2,4-D. All media were distributed in Gerber-type glass jars with 20 ml of medium per container and placed in a culture room at 25 °C with a light intensity of 12.9 µmol m-2 s-1 and a 16/8 hours light/dark photoperiod. For histological analysis, samples were taken from the in vitro material and fixed using formaldehyde, alcohol, and acetic acid (FAA). Dehydration was then carried out by immersion in graded alcohols. using the 12-station Histokiner Leica TP 1020 automatic tissue processor. The material was left to rest for 6 hours per station. Subsequently, paraffin embedding was performed, and 12 µm sections were made using a manual rotary microtome (Leica®, RM212RT). The in vitro material samples were stained by immersion in xylene for three minutes each, followed by 100%, 96%, 70%, and 50% (v/v) alcohol for three minutes each. They were then stained by immersion for 15 minutes in toluidine blue, followed by a rinse with 96% (v/v) ethyl alcohol (v/v) for ten seconds, then transferred to 96% (v/v) ethyl alcohol for one and a half minutes, followed by a series of immersions in 100% alcohol for 16 minutes and xylene for 12 minutes, and then fixed with liquid resin. They were left to dry for 10 days and photographs were taken in bright field (Axio Scope A1, Carl Zeiss, Oberkochen, Germany). The brightness and contrast of the photographs were adjusted using Zen lite 2012 software.
Rapid safranin-green staining and fluorescence microscopy. The in vitro material samples were stained by immersion in xylene for three minutes each, followed by gradual alcohols at 100%, 96%, 70%, and 50% (v/v) for three minutes each, then stained by immersion for 45 minutes in safranin, followed by rinsing in distilled water for 10 seconds and transferred to 50%, 70%, and 96% ethyl alcohol (v/v) for three minutes, two minutes, and two minutes, respectively. A second staining with fast green was then performed for one minute and rinsed with absolute alcohol, followed by a series of immersions in 100% alcohol for 16 minutes and xylene for 12 minutes, and then fixed with liquid resin. Finally, they were left to dry for 10 days and visualized using the red and green filters of the Carl Zeiss fluorescence microscope, model Axio Scope A1 (Oberkochen, Germany). The micrographs were then processed with Zen lite 2012 software, adjusting only the contrast and brightness.
RESULTS
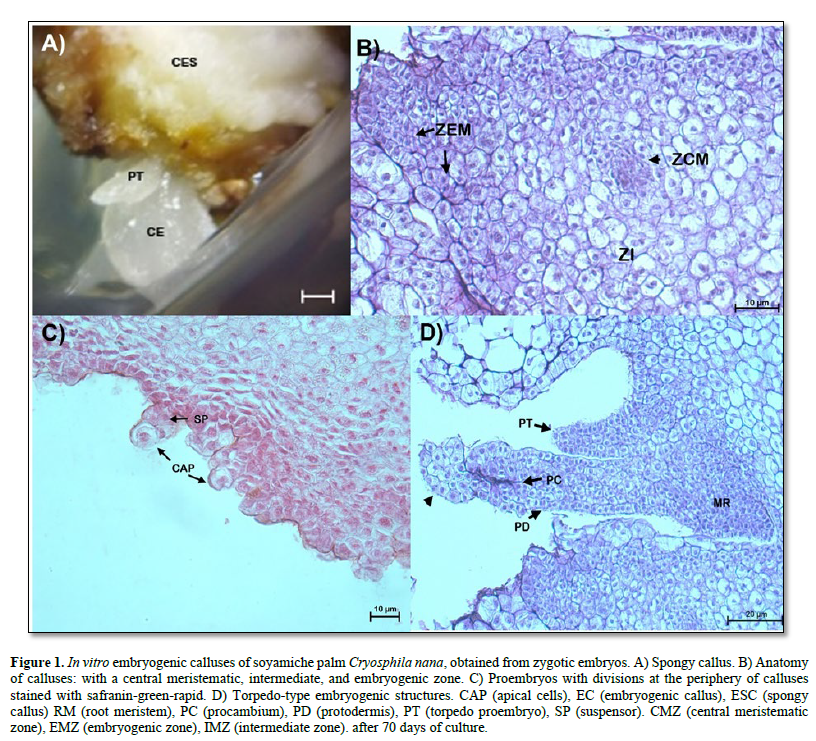
Anatomical description of an embryogenic callus. To better understand the somatic embryogenesis of the soyamiche palm Cryosphila nana, histological analyses were performed on each of the structures obtained during the process of somatic embryogenesis in vitro. In the culture medium used to induce callus formation from soyamiche palm zygotic embryos, spongy calluses ranging in color from white to beige were obtained after 70 days of culture.
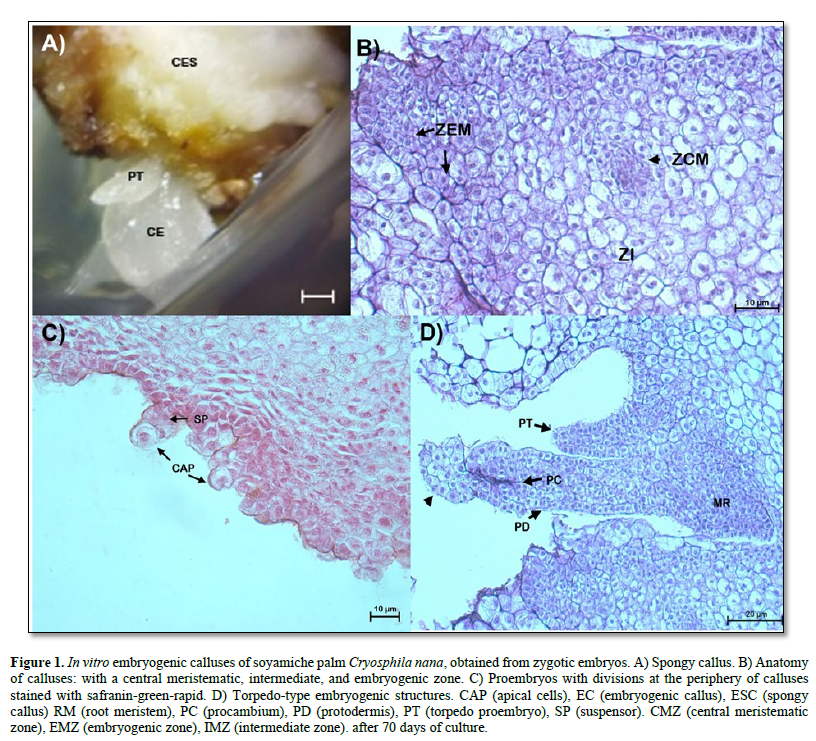
The characteristics of the embryogenic Calli obtained began to differentiate with the formation of more compact translucent tissues, initiating the formation of torpedo-type embryos (Figure 1 A). In Figure 1 B, a potentially embryogenic callus was observed differentiating: three cell layers, a central meristematic zone, formed by dividing cells, that is, showing large nuclei, without vacuoles or small in size, dense cytoplasm, and thin cell walls; the intermediate zone is formed by cells with parenchymatous characteristics: with small nuclei that are compressed toward the cell wall by large vacuoles. At the periphery of the callus, an embryogenic zone was observed, formed by dividing cells with large nuclei, fragmented vacuoles, dense cytoplasm, and visible intracellular spaces. Similarly, multicellular proembryos were visualized at the periphery of the callus. These are formed by an apical cell and a suspensor, which indicates the unicellular origin of somatic embryos (Figure 1 C). It was also observed that some calluses began to differentiate more advanced embryogenic structures, defining a globular (not shown) and torpedo stage, the latter characterized by an elongated structure with protodermis, procambium, and a root meristem (Figure 1 D). In addition, in the distal region of the torpedo-type proembryo, the formation of a spherical structure (arrowhead, Figure 1 D) was observed, consisting of dividing cells with large nuclei, visible nucleoli, and large vacuoles. These structures probably defines a secondary-types omatic embryo.
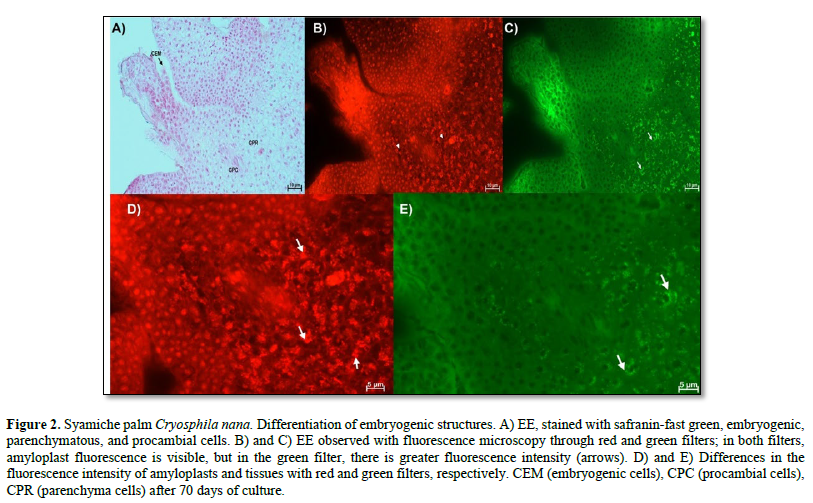
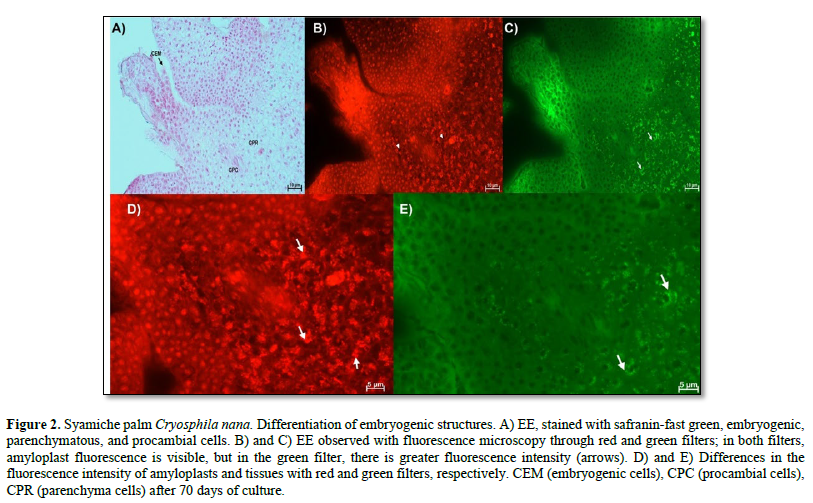
In Figure 2 A, the cell differentiation process highlights the embryogenic structures, made up of cells with large nuclei, visible nucleoli, dense cytoplasm, and no or very few vacuoles. More differentiated tissues were also identified that belong to the characteristics of the explant used, that is, the tissues of the zygotic embryos used, in which parenchyma cells were observed, which had large vacuoles, small nuclei, and amyloplasts, while the procambial cells are elongated with large nuclei. Using fluorescence, it was possible to distinguish the tissue of the embryogenic structure from that of the explant because fluorescence was observed in the latter (Figure 2 B and C). The fluorescence of the amyloplasts (arrows) of the parenchyma was also evident in the red and green filters. Furthermore, in the red filter, the vacuoles of the parenchymatous cells can be distinguished as a black area within the cell (Figure 2 D and E).
Anatomical Description of Globular Embryos
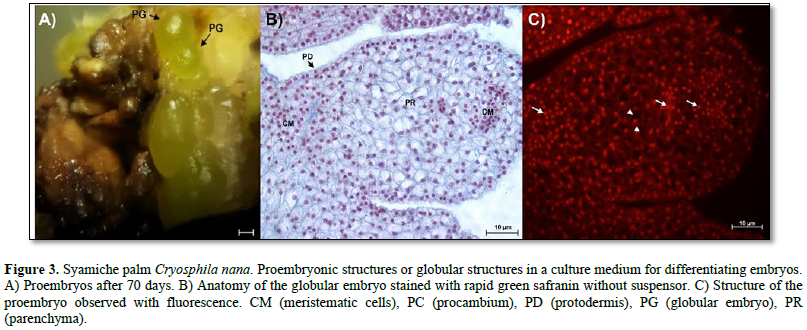
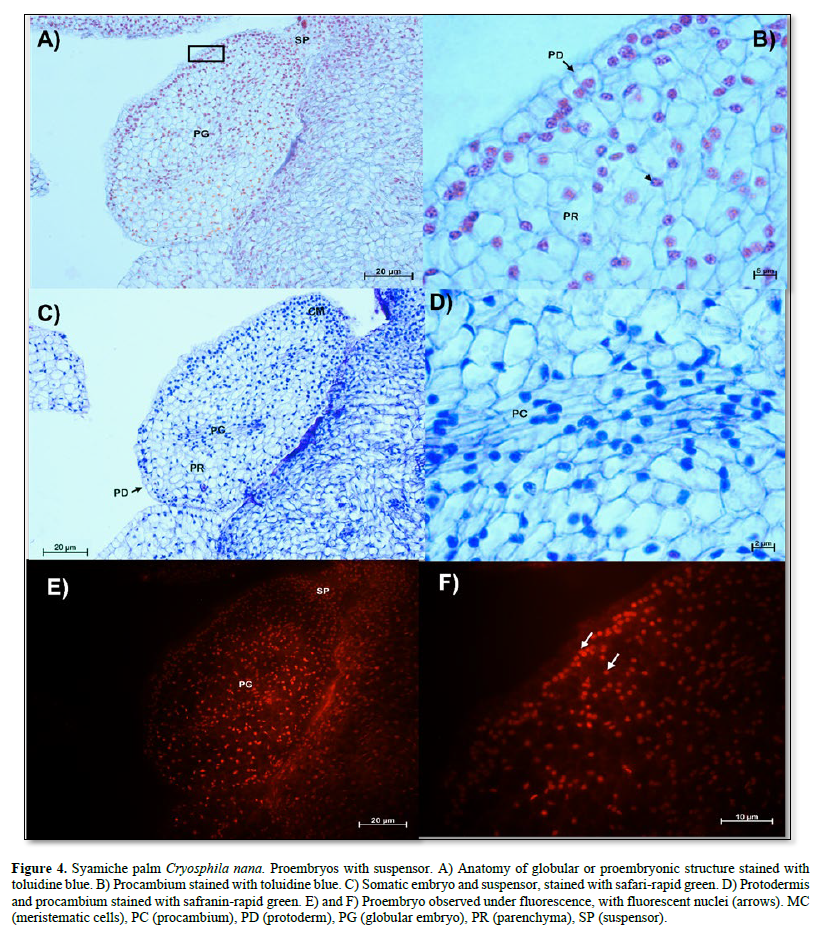
In Figure 4 A, a globular embryo with an evident suspensor is observed, in addition to a protodermis characterized by cells with large, centralized nuclei that form a uniseriate layer (Figure 4 B). On the other hand, an area with meristematic-type cells was observed in the proximal region (Figure 4 C). The characteristics of the cells of the proembryos of the soyamiche palm are the beginning of the formation of procambium cells or vascular tissues, which are characterized by elongated cells with nuclei intensely stained with toluidine blue (Figure 4 D).
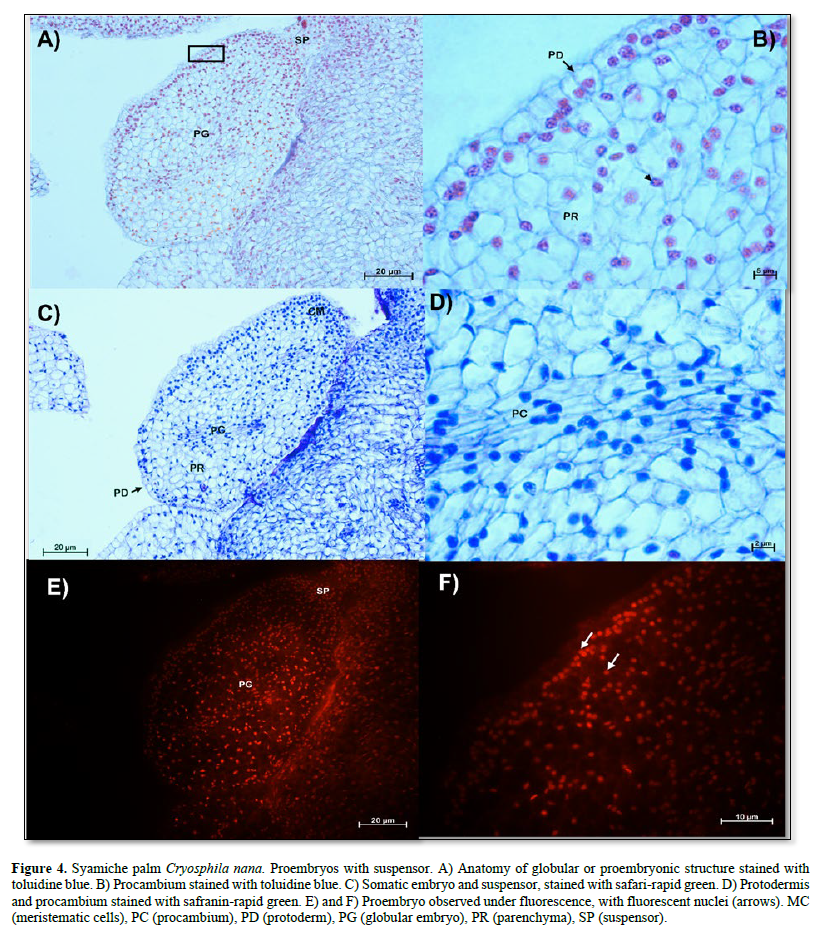
Fluorescence showed that only the nuclei of all cell types emitted intense fluorescence (Figure 4 E and F).
Anatomical description of torpedo-type embryos
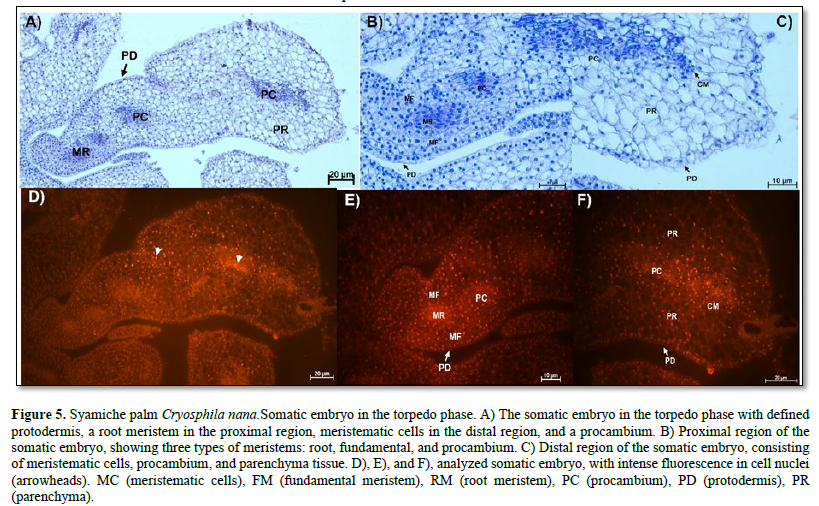
The stage of somatic embryogenesis is the torpedo phase. Figure 5 A shows the anatomy of a torpedo embryo without vascular connection to the maternal tissue. At the proximal pole is the root meristem, formed by cells with meristematic characteristics such as abundant nuclei and cytoplasm, large nuclei-stained bright blue, and visible nucleoli.
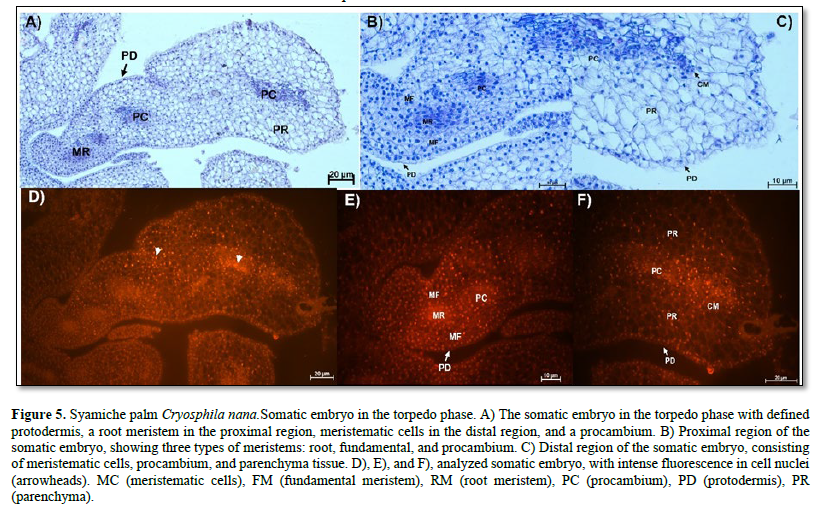
On the sides of the root meristem, fundamental meristem cells with different shapes, visible vacuoles, and centralized nuclei were observed. Similarly, a procambium composed of elongated cells with large nuclei and visible nucleoli was observed (Figure 5 B). At the distal pole, meristematic and parenchymatous cells were present (Figure 5 C). The meristematic regions of the embryo were more evident with the use of fluorescence microscopy because their nuclei emitted intense fluorescence (Figure 5 D, E, F).
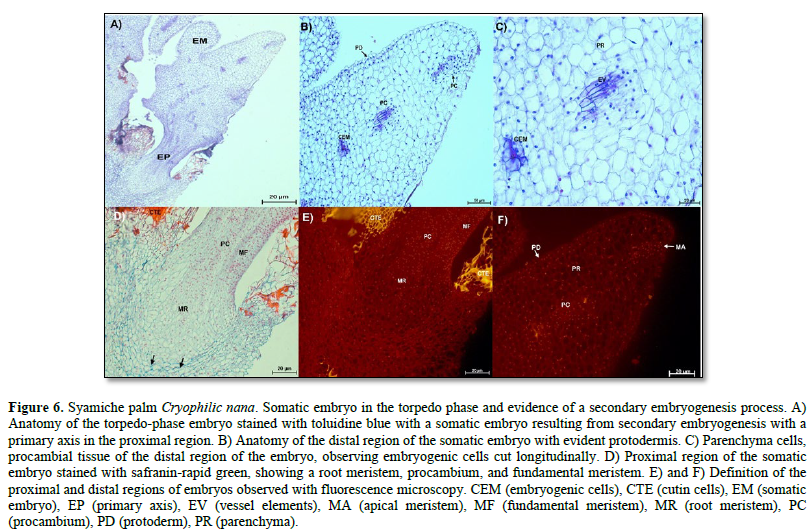
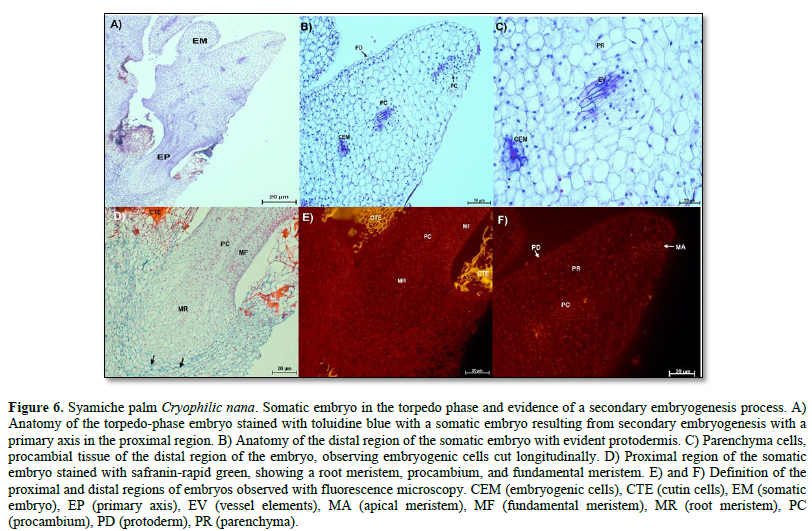
In the torpedo-stage embryos, the formation of a primary axis was observed in the proximal region of the embryo (Figure 6 A). This axis consists of a root merited, a fundamental meristem, and procambium. Here, the most developed cells were characterized by a thick cell wall composed mainly of cellulose (Figure 6 D, arrows). The formation of a secondary somatic embryo was also evident, which was observed on the lateral side of the torpedo-type embryo (Figure 6 A). Longitudinally cut embryogenic cells are present in the distal region. Parenchymatous, protodermal, and procambial cells, specifically vessel elements, were observed in this region (Figure 6 B and C).
Meanwhile, the nuclei of these cell types showed fluorescence, and thanks to this technique, it was possible to visualize the cuticle of some cells belonging to the maternal tissue, whose fluorescence appeared as an intense yellow color (Figure 6 E and F).
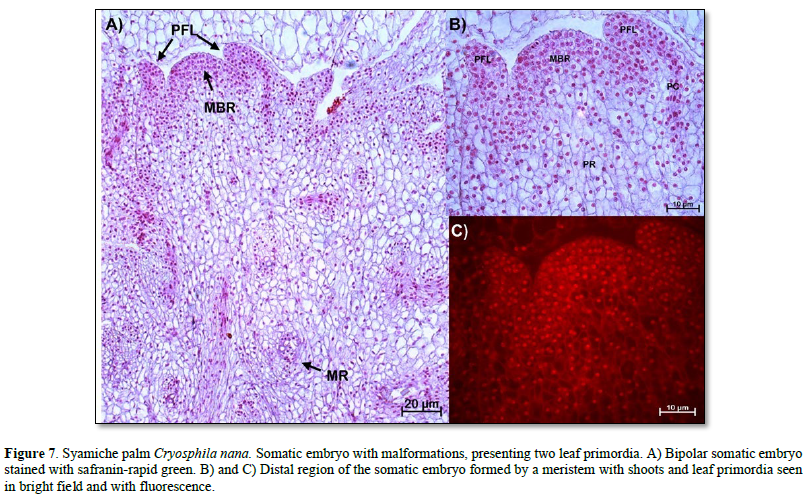
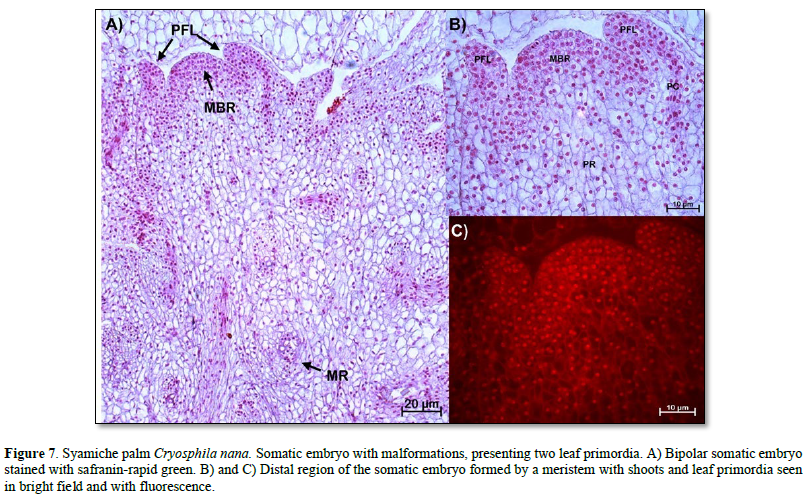
Anatomical description of mature somatic embryos (mutants). Once the somatic embryos were obtained, they were subcultured in culture media to reach maturity. However, it was evident that there were embryogenic structures with malformations, as shown in Figure 7. Here, an embryo with two leaf primordia and a root meristem was observed. This distal region was characterized by meristematic cells with nuclei intensely stained with safranin and intense fluorescence (Figure 7 B and C).
DISCUSSION
Anatomical description of embryogenic calluses. The physical characteristics of the embryogenic calluses of the soyamiche palm were similar to those reported by Silva-Cardoso et al. (2022) and Ferreira et al. (2022a), who reported observing embryogenic cells in the periphery of the spongy callus, as in the calluses of the soyamiche palm, so this type of callus can be considered potentially embryogenic for this species. In addition, the definition of three growth zones (central meristematic, intermediate, and embryogenic) found in soyamiche palm calluses coincides with the embryogenic calluses obtained from leaf explants and zygotic embryos of the Euterpe precatoria palm (Ferreira et al., 2022b; Ferreira et al., 2022a) and the species Syagrus oleracea (Silva-Cardoso et al., 2022) with tissues taken from inflorescences as explants. In addition, it is recognized that the central meristematic zone is strongly related to callus growth (Ferreira et al., 2022a).
The embryos developed in the periphery of the callus may have a unicellular development due to the presence of a suspensor, which, together with the apical cell, is evidence of asymmetric cell division (Luis and Scherwinski-Pereira, 2014).
They also reported obtaining embryos in the torpedo phase and the formation of embryogenic structures during the callus induction stage, which has been reported in species such as Euterpe precatoria and Syagrus oleracea (Ferreira et al., 2022a; Silva-Cardoso et al., 2022). According to the hypothesis of Silva-Cardoso et al. (2022), these developed proembryonic structures are somatic embryos formed from cell clusters and are of multicellular origin.
In soyamiche palm, evidence of cellular behavior was found that possibly indicates the onset of secondary embryogenesis. This process has also been reported in other palm species such as: Syagrus oleracea (De Araújo Silva-Cardoso et al., 2020), Bactris gasipaes (Steinmacher et al., 2011), and Euterpeprecatoria (Ferreira et al., 2022b). In species such as Bactris gasipaes, attempts have been made to promote secondary embryogenesis due to its embryogenic potential despite several subcultures, thereby obtaining a greater number of somatic embryos (Steinmacher et al., 2011). Therefore, the results in the formation of embryogenic calluses, proembryonic phases, and somatic embryos, the treatments used, the culture media, and the participation of growth regulators in soyamiche palm may not only help accelerate the process of cell differentiation in the embryogenic calluses obtained, but it is also increase yields in the obtaining of somatic embryos. However, further studies are needed to explain the processes of secondary somatic embryogenesis in this species of soyamiche palm.
Using fluorescence, it was possible to observe the presence of amyloplasts in parenchymatous cells. This type of plastid is mainly found in reserve parenchyma cells. It has been reported that callus formation begins in parenchyma cells and that carbohydrate reserves in palms during somatic embryogenesis are related to the differentiation of embryogenic cells (Meira et al., 2019). In addition, these starch reserves serve as an energy source for the intense cellular activity of the embryogenic cell process (Ferreira et al., 2022b).
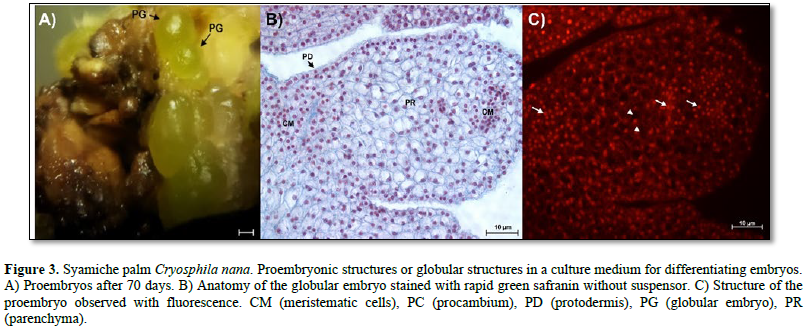
Anatomical description of globular embryos. The precise origin of somatic embryos should be discussed, recognizing that they may have two origins: both unicellular and multicellular (Silva-Cardoso et al., 2022; Ferreira et al., 2022a). The unicellular origin can be confirmed by the development of a suspensor, while embryos of multicellular origin do not develop one (Ferreira et al., 2022a). Figures 2 and 3 A show that the two types of somatic embryos were formed from calluses obtained from zygotic embryos of the soyamiche palm. According to Luis and Scherwinski-Pereira (2014), somatic embryos of multicellular origin are formed from the internal tissue of nodular calluses, while unicellular embryos are formed from external tissue, as reported for the Acrocomia aculeata palm.
On the other hand, the protodermis is a meristematic type that will give rise to the epidermis, and it is the first tissue that was histologically identified in the globular embryos of species such as: Euterpe precatoria (Ferreira et al., 2022b), Acrocomia aculeata (Luis and Scherwinski-Pereira, 2014) , Syagrus oleracea (De Araújo Silva-Cardoso et al., 2020), and Elaeis guineensis (Vilela et al., 2019). In the latter, the formation of procambial tissue was also observed, like the embryos with suspensor of the soyamiche palm. Also, the meristematic cells located in the proximal regions of the globular embryos are indicative of meristematic tissue formation (Silva-Cardoso et al., 2022), as shown in the globular or embryogenic embryos of the species analyzed. Using fluorescence microscopy, it was observed that the somatic embryos of soyamiche palm presented red fluorescence in their nuclei when using safranin as a fluorophore. This is because the excitation spectrum of the red filter is 530-555 nm and, according to Guo et al. (1998), safranin has a maximum excitation spectrum of 533 nm in the presence of nucleic acids and a maximum emission spectrum of 578 nm. On the other hand, with the green filter (excitation spectrum = 450-490 nm), it did not exhibit fluorescence.
This coincides with what was observed in flax cells with lignified walls under 488 nm excitation, determining that their emission spectrum depends on the amount of lignin in the walls, whose values ranged between 570–600 nm, which corresponds to the red and orange region. They also observed that the green region remained without fluorescence (Baldacci-Cresp et al., 2020). Based on the above, it is recommended that the red filter be used for the development of somatic embryos, because with the green filter, other cellular components such as walls cannot be distinguished, as young tissues do not yet contain lignin.
Anatomical description of torpedo-type embryos. The bipolarity characteristic of the embryos obtained, which became evident during the torpedo phase or stage, was also reported in somatic embryos of the palm Acrocomia aculeata (Luis and Scherwinski-Pereira, 2014). The differentiation and observation of tissues such as the protodermis and fundamental meristem, as well as the root meristem with procambium, in addition to other characteristics in calluses according to their origin, have been characteristics of torpedo-type embryos in other palm species such as: Eateries precatoria (Ferreira et al., 2022B), Syagrus oleracea (Silva-Cardoso et al., 2022), Elaeis guineensis (Vilela et al., 2019), and Euterpe oleracea (De Olivera Freitas et al., 2016). With the results reported above, it was possible to identify somatic embryos in the torpedo stage in soyamiche palm.
Similarly, somatic embryos in the torpedo stage, it was possible to visualize some embryos of secondary origin. Studies have shown that secondary embryogenesis can occur in the early stages of primary embryogenesis (Steinmacher et al., 2011) . For example, in Syagrus oleracea (De Araújo Silva-Cardoso et al., 2020), Bactrisgasipaes (Steinmacher et al., 2011) and Euterpe oleracea (De Olivera Freitas et al., 2016), the formation of secondary embryos occurred in the proembryonic or globular embryo stages, while in Euterpe precatoria it occurred in the embryo maturation stage (Ferreira et al., 2022a). However, it is in the early stages of development that there is greater embryogenic capacity because there is greater expression of transcription factors such as SERK and LEC, which are related to somatic embryogenesis (Steinmacher et al., 2011).
On the other hand, with the helpof fluorescence microscopy, it was possible to identify the cuticle of the cells corresponding to the maternal tissue, which presented an intense yellow color, as reported by De Micco and Aronne (2007) in epidermal cells of Rhamnus alaternus L. stained with safranin.
In addition, it was observed that the torpedo-type somatic embryo was made up of a primary axis. However, the development of this structure has not been reported before, but based on the location of the root meristem, it is hypothesized that it may be due to abnormal hypocotyl development.
Anatomical description of mature somatic embryos (mutants). In the soyamiche palm, embryos with abnormal development were found. The appearance of this type of embryo has been reported in other species such as Butiaodorata (Fior et al., 2018) and Euterpe oleracea (De Olivera Freitas et al., 2016), Euterpe precatoria (Ferreira et al., 2022a), noting that the abnormal somatic embryos obtained were fused to meristematic tissue or fused. However, in the case of those obtained from soyamiche palm, these had two primordia. According to García et al. (2019), the most common abnormalities in somatic embryos are the fusion of two or more embryos, the lack of apical or radicle meristems, and the appearance of multiple cotyledons, as observed in the analyzed species. In Paeonia spp., embryos with multiple cotyledons, a single cotyledon, and a tubular cotyledon were reported. These abnormalities were possibly caused by disruptions in cell division in vitro, which was also induced by the high concentrations of growth regulators added to the culture media. However, these embryos did not germinate or produce any radicle (Du et al., 2020).
CONCLUSION
The anatomical and cellular characteristics in the differentiation of the proembryonic structures and somatic embryos obtained in vitro in this soyamiche palm, locating each of the phases found during the process of somatic embryogenesis allowed to identify the development of cellular behavior until somatic embryos were obtained, successfully corroborating with histological work and fluorescence microscopy in these processes. This allowed the differentiation of somatic embryos of direct origin, multicellular, unicellular, and embryos with specific disorders.
- Baldacci‐Cresp F., Spriet, C., Twyffels L., Blervacq A. S., Neutelings G., Baucher, M. y Hawkins, S. (2020). A rapid and quantitative safranin‐based fluorescent microscopy method to evaluate cell wall lignification. The Plant Journal, 102(5), 1074-1089. https://doi.org/10.1111/tpj.14675
- De Araújo Silva‐Cardoso I. M., Meira, F. S., Gomes, A. C. y Czerwinski‐Pereira, J. E. (2020). Histology, histochemistry and ultrastructure of pre‐embryogenic cells determined for direct somatic embryogenesis in the palm tree Syagrus oleracea. Physiologia plantarum, 168(4), 845-875. https://doi.org/10.1111/ppl.13026
- De Micco, V. y Aronne, G. (2007). Combined histochemistry and autofluorescence for identifying lignin distribution in cell walls. Biotechnic & Histochemistry, 82(4-5), 209-216. https://doi.org/10.1080/10520290701713981
- De Olivera Freitas, E., Monteiro, T. R., Nogueira, G. F. y Scherwinski-Pereira, J. E. (2016). Somatic embryogenesis from immature and mature zygotic embryos of the açaí palm (Euterpe oleracea): Induction of embryogenic cultures, morphoanatomy and its morphological characteristics. Scientia Horticulturae, 212, 126-135.
- Du, Y., Cheng, F. y Zhong, Y. (2020). Induction of direct somatic embryogenesis and shoot organogenesis and histological study in tree peony (Paeonia sect. Moutan). Plant Cell, Tissue and Organ Culture (PCTOC), 141, 557-570. https://doi.org/10.1007/s11240-020-01815-4
- Ferreira, J. C. B., de Araújo Silva-Cardoso, I. M., de Oliveira Meira, R., y Scherwinski-Pereira, J. E. (2022A). Somatic embryogenesis and plant regeneration from zygotic embryos of the palm tree Euterpe precatoria Mart. Plant Cell, Tissue and Organ Culture (PCTOC), 148(3), 667-686. https://doi.org/10.1007/s11240-022-02227-2
- Ferreira, J. C. B., de Araújo Silva-Cardoso, I. M., Meira, R. O., da Silva Costa, F. H. y Scherwinski-Pereira, J. E. (2022B). Towards development of an efficient somatic embryogenesis protocol for the palm tree Euterpe precatoria (Mart.) from leaf tissues of adult plants. In Vitro Cellular & Developmental Biology-Plant, 58(5), 750-768. https://doi.org/10.1007/s11627-022-10310-8
- Fior, C., Costa-Calil, A., Dutra de Souza, P. y Schwarz, S. (2018). Impacto de las concentraciones de ANA y sacarosa en el enraizamiento in vitro de Butiaodorata (Barb. Rodr.) Noblick. Iheringia, 73(1), 13-21. DOI:10.21826/2446-8231201873102
- García, C., Furtado de Almeida, A. A., Costa, M., Britto, D., Valle, R., Royaert, S. y Marelli, J. P. (2019). Abnormalities in somatic embryogenesis caused by 2, 4-D: an overview. Plant Cell, Tissue and Organ Culture (PCTOC), 137, 193-212. https://doi.org/10.1007/s11240-019-01569-8
- Guo, X. Q., Li, F., y Zhao, Y. B. (1998). Fluorescence investigation of the interaction of safranine T with DNA. Analytical letters, 31(6), 991-1005.
- Hassan, M. M., Allam, M. A., Shams El Din, I. M., Malhat, M. H., y Taha, R. A. (2021). High-frequency direct somatic embryogenesis and plantlet regeneration from date palm immature inflorescences using picloram. Journal of Genetic Engineering and Biotechnology, 19, 1-11. https://doi.org/10.1186/s43141-021-00129-y
- Liu, Y., Wei, C., Wang, H., Ma, X., Shen, H., y Yang, L. (2021). Indirect somatic embryogenesis and regeneration of Fraxinus mandshurica plants via callus tissue. Journal of Forestry Research, 32(4), 1613-1625. https://doi.org/10.1007/s11676-020-01199-3
- Luis, Z. G. y Scherwinski-Pereira, J. E. (2014). An improved protocol for somatic embryogenesis and plant regeneration in macaw palm (Acrocomia aculeata) from mature zygotic embryos. Plant Cell, Tissue and Organ Culture (PCTOC), 118, 485-496. https://doi.org/10.1007/s11240-014-0500-x
- Meira, F. S., Luis, Z. G., de Araújo Silva-Cardoso, I. M. y Scherwinski-Pereira, J. E. (2019). Developmental pathway of somatic embryogenesis from leaf tissues of macaw palm (Acrocomia aculeata) revealed by histological events. Flora, 250, 59-67. https://doi.org/10.1016/j.flora.2018.11.011
- Oliveira, L. B., de Mello, T., de Araujo, C. P., de Oliveira, J. P. B., Ferreira, A., Zanardo, T. É. C., Vieira, L.M., Otoni, W. O., Alexandre, R.S. y Carvalho, V. S. (2022). Morphoanatomical aspects of auxin herbicides-induced somatic embryogenesis in Euterpe edulis Martius, a symbol and threatened species of the Atlantic Forest. ScientiaHorticulturae, 299, 111051. https://doi.org/10.1016/j.scienta.2022.111051
- Ree, J. F., y Guerra, M. P. (2015). Palm (Arecaceae) somatic embryogenesis. In Vitro Cellular & Developmental Biology-Plant, 51, 589-602. https://doi.org/10.1007/s11627-015-9722-9
- Silva-Cardoso, I. M. D. A., Meira, F. S., y Scherwinski-Pereira, J. E. (2022). The maturity level of explant plays a key role in somatic embryogenesis of the palm tree Syagrus oleracea [Mart.] Becc. Acta PhysiologiaPlantarum, 44(8), 87. https://doi.org/10.1007/s11738-022-03420-8
- Steinmacher, D. A., Guerra, M. P., Saare-Surminski, K. y Lieberei, R. (2011). A temporary immersion system improves in vitro regeneration of peach palm through secondary somatic embryogenesis. Annals of Botany, 108(8), 1463-1475. https://doi.org/10.1093/aob/mcr033
- Vilela M. S. P., Santos, R. S., Stein, V. C. y Paiva, L. V. (2019). Histological analysis of indirect somatic embryogenesis induced from root explats of oil palm (Elaeis guineensis Jacq). Revista árvore, 43. https://doi.org/10.1590/1806-90882019000100006
- Winkel mann, T. (2016). Somatic versus zygotic embryogenesis: learning from seeds. Invitro embryogenesis in higher plants, 25-46. https://doi.org/10.1007/978-1-4939-3061-6_2
- Yan R., Sun Y., Sun, H. (2020). Current status and future perspectives of somatic embryogenesis in Lilium. Plant Cell, Tissue and Organ Culture (PCTOC), 143, 229-240. https://doi.org/10.1007/s11240-020-01916-0
- Zhang M., Wang A., Qin M., Qin X., Yang S., Su, S., Sun, Y., y Zhang, L. (2021). Direct and indirect somatic embryogenesis induction in Camellia oleifera Abel. Frontiers in Plant Science, 12, 644389. https://doi.org/10.3389/fpls.2021.644389